Cancer risks and Vitamin D Receptors – association is unclear – 2017
Vitamin D Receptor Polymorphism and Cancer: An Update
Anticancer Research 37: 3991-4003 (2017), doi: 10.21873/anticanres .11784
📄 Download the PDF from VitaminDWiki
VIKRANT RAI, JOE ABDO, SWATI AGRAWAL and DEVENDRA K. AGRAWAL Dkagr@creighton.edu
Department of Clinical and Translational Science, Creighton University School of Medicine, Omaha, NE, U.S.A.
Figure 1
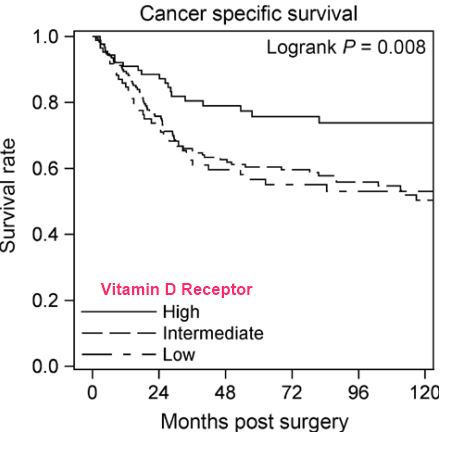
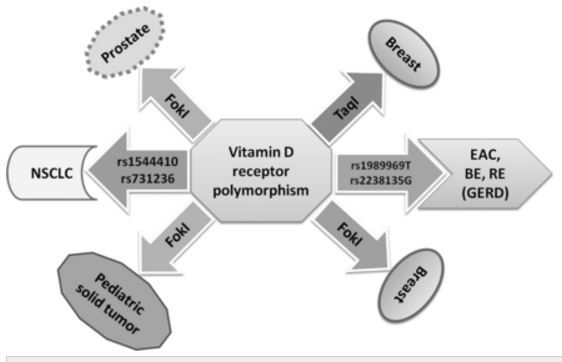
Figure 2
Note: Tables and references are in the PDF
Background: Vitamin D mediates its action via vitamin D receptor (VDR) and is involved in a wide variety of biological processes including regulation of cell proliferation and differentiation in normal tissue and apoptosis, and cell adhesion in tumor cells. The study of genetic variations in VDR may elucidate the association of vitamin D levels, its metabolism, and VDR polymorphism with various diseases and cancer. The association of VDR polymorphism with cancer has been reported; however, the literature lacks critical analyses of the studies in last 3 years.
Materials and Methods: A systematic search of PubMed database (2015 through mid-2017) was conducted to provide a comprehensive overview of this clinical arena. Results: Studies on the association of VDR polymorphisms FokI, BsmI, TaqI, and ApaI and cancer have suggested the involvement of VDR polymorphism in tumorigenesis.
Conclusion: The inconsistent results and lack of the studies in some cancer types warrant additional research.
Introduction
Vitamin D is involved in a wide variety of biological processes including bone metabolism, modulation of the immune response, and regulation of cell proliferation and differentiation. Vitamin D has a substantial function in overall human health, including cancer occurrence (1). Vitamin D regulates cellular differentiation and proliferation in normal tissue and regulates proliferation, apoptosis, and cell adhesion at the tumor cell level. It also modifies tumor angiogenesis, invasion, and metastasis along with decreasing oxidative DNA damage (2). Vitamin D mediates its biological action via the vitamin D receptor (VDR) (3). VDR is a steroid intracellular hormone receptor which binds to 1,25(OH)2 D and interacts with vitamin D receptor response elements (VDRE) of the target genes to produce various biological effects (4). The VDR gene lies on the long arm of chromosome 12 (12q12-14) and has approximately 200 single nucleotide polymorphisms (SNPs) (5). The most common allelic variants studied includes a start codon polymorphism FokI (T/C) in exon II, BsmI (A/G) and ApaI (C/A) polymorphisms in the intron between exon VII and IX and a TaqI (T/C) variant in exon IX (6). The most commonly studied VDR polymorphisms include Fok1 (rs2228570), Apa1 (rs7975232), Bsm1 (rs1544410), Bgl1 (rs739837), and restriction fragment length polymorphisms rs7975232 (G/T substitution), rs1544410 (A/G substitution), and rs739837 (G/T substitution) (7). Since vitamin D acts through VDR, studying genetic variations in VDR may elucidate the association of altered vitamin D levels and metabolism, VDR polymorphism, and cancer. This systemic review focused on pooling the case-control studies and metaanalyses performed in the past 3 years (2015-mid 2017) for the association of VDR polymorphism and cancer.
Materials and Methods
A systematic search of the PubMed database (January 2015 through mid-2017) of the National Library of Medicine was performed using the following Medical Subject Headings: cancer, vitamin D, VDR polymorphism, tumor, breast cancer, female reproductive tract cancer, prostate cancer, skin cancer, gastrointestinal tract, esophageal cancer, colorectal cancer, urinary tract cancer, pediatric tumors, hematologic cancer. We included articles that examined and described the VDR polymorphisms in relation to risk and incidence of cancer. Articles that were not written in English, conference abstracts and studies not performed on humans were excluded.
VDR Polymorphism and Cancer
VDR SNPs within the gene coding for VDR may influence cancer risk (8). Numerous investigations have analyzed VDR- affiliated SNPs; however, their general association with carcinogenesis remains controversial. Significant associations between VDR polymorphisms and breast (Fok1, Bsm1, Apal), prostate (Fok1, Bsm1, TaqV), colorectal (Fok1, Bsm1, Taq1) and skin cancer (Fok1, Bsm1, TaqV) have been reported (9). For instance, the CDX2 polymorphism (SNP - rs11568820) of the VDR gene has been associated with cancer susceptibility amid varied results. Various researchers have conducted the meta-analysis to establish the viable association between VDR polymorphism and cancer risk (10-22). A meta-analysis including 25 separate studies totaling 34,018 individuals published prior to March 2015 for CDX2 polymorphism association with cancer conducted by Dai et al. demonstrated that in the homozygous model, CDX2 mutations were significantly associated with an increased cancer risk of colorectal and ovarian cancer, particularly in African- American but not in Caucasians or Asians (10). Data-analysis of dominant, homozygous and recessive comparison models yielded a significant association between cancer risk and CDX2 mutations in African-Americans. Stratifying the data by cancer types showed a strong association between VDR CDX2 polymorphisms and an amplified risk of colorectal and ovarian cancer (10). Gandini et al. analysed the association between VDR polymorphisms (Fok1, Bsm1, Taq1, Apa1, and Cdx2) and various types of cancer such as ovarian cancer, renal cell carcinoma, thyroid carcinoma, hepatocellular carcinoma (HCC), sarcoma, skin (melanoma and nonmelanoma skin cancer), bladder cancer, non-Hodgkin’s lymphoma, leukaemia, oesophageal adenocarcinoma (EAC), head and neck and oral squamous cell carcinoma including 79 independent studies, for a total of 52427 cases and 62225 controls (9). The study concluded that significant associations exist between VDR polymorphisms and prostate (Fok1, Bsm1, Taq1), breast (Fok1, Bsm1, Apa1), colorectal (Fok1, Bsm1, Taq1) and skin cancer (Fok1, Bsm1, Taq1) (Figures 1 and 2).
Figure 2. Vitamin D receptor (VDR) polymorphisms associated with reduced risk of cancer.
The presence of these polymorphisms is associated with a reduced risk of cancer. However, these relationships may vary with age, ethnicity, and race as discussed in the text. BE: Barrett's oesophagus, EAC: oesophageal adenocarcinoma; GERD: gastro-oesophageal reflux disease, RE: reflux oesophagitis; NSCLC: non-small cell lung cancer.
A meta-analysis between VDR polymorphism including Cdx2 (rs11568820), FokI (rs2228570), BsmI (rs1544410), ApaI (rs7975232), and TaqI (rs731236) and the risks for female reproductive cancer such as breast, ovarian, cervical, endometrial, uterine and vaginal cancer, found an increased risk of developing breast and ovarian cancers with FokI polymorphism and reduced risk with BsmI polymorphism (19). A meta-analysis for the association between VDR polymorphism (TaqI, ApaI, and Cdx2) and the risk of basal cell carcinoma, colorectal cancer, EAC, hepatocellular carcinoma, head and neck cancer, non-Hodgkin lymphoma, oral squamous cell carcinoma, renal cell carcinoma, squamous cell carcinoma, and thyroid carcinoma found Cdx2 gg versus GG to be associated with a significantly increased risk for all cancer types and TaqI was associated with significantly increased risk for colorectal cancer (21) (Figures 1 and 2). Significant variations in frequency were also existent between diverse ethnic groups. VDR genotypes may have more significance when grouped with specific haplotypes, or when linked to additional gene polymorphisms involved in the vitamin D pathway, or when stratified by different tumor type or patient background (21). Nevertheless, there are clear intersections between the ability of VDR to function properly and carcinogenesis in a number of indications. This section focuses on the effect of VDR polymorphisms in the entire gastrointestinal tract - where vitamin D-related patient management strategies hold much promise.
Female Reproductive Tract Cancer
VDR polymorphism has been associated with cancer of the female reproductive including breast, ovarian, cervical, endometrial, uterine, and vaginal cancer (19). Increased expression of VDR has been found in breast cancer and inversely related to aggressive tumor characteristics, including large tumor size, hormonal receptor (HR) negativity, triplenegative subtype [estrogen-receptor (ER) negative, progesterone receptor (PR) negative, and human epidermal growth factor receptor 2 (HER2) negative], and expression of proliferative marker Ki-67 (23). Similarly, various case- control studies suggested that the association between VDR polymorphism and risk of breast cancer may depend on race and ethnicity. Furthermore, these studies also showed inconsistent results, some suggesting the association of VDR polymorphism with breast cancer, while others suggested a lack of association (3, 19, 24-31). No association between VDR polymorphisms and breast cancer risk was found, however, modification of this relationship was observed with the menopausal status in Europeans (VDR rs4328262 and rs11168292) and East Asians (VDR rs11168287). Furthermore, heterogeneity by tumor subtype was also seen for rs1544410, rs7967152 and rs2239186 VDR polymorphisms in Europeans (30). These case-control and meta-analysis studies suggest that VDR polymorphism may be associated with increased risk of breast cancer (Table I). However, Shaikh et al. reviewed the impact of VDR gene polymorphisms Fok1, Bsm1, Taq1, Apa1 and poly(A) on development of breast cancer from 1996 to 2015 and suggested that due to the inconsistent results of various studie, no conclusive evidence associates the risk of breast cancer with VDR polymorphism (32). Similarly, the inconsistent results of the studies shown in Table I suggest the need for further research.
Table I
Table II. Vitamin D receptor (VDR) polymorphisms and prostate cancer.
Prostate Cancer
Prostate cancer is the most common malignant tumor in older men across the globe. The incidence of prostate cancer is increasing worldwide while ethnic origin, family history, hormonal status, dietary structure, age and low vitamin D levels are risk factors for prostate cancer (36, 37). Furthermore, studies have reported the association between VDR polymorphism with an increased risk of prostate cancer (36, 38). However, the results of various studies were disputable and contradictory (39, 40). Most of the studies found no association between VDR polymorphism and prostate cancer (13, 22), however, Nunes et al. found correlation between CC genotype (TaqI) with age (>58 years old), and GG (BsmI) with lower prostate-specific antigen (PSA) levels (<10 ng/ml) (41). Aassociation between a significantly increased estimated chance of PSA <10 ng/ml with G allele (Bsml) and 9.75-fold increased chance of patients with prostate cancer having a lower PSA level with GG/GG genotype (BsmI/Apal) was also found. Recently, various metaanalysis and case-control studies have been conducted to find a relation between VDR polymorphism (TaqI, FokI, Cdx2, Apal, Bsml, rs731236, rs7975232, rs731236, rs1544410, rs3782905, rs1544410, and rs2239185) and risk of prostate cancer (11, 13, 18, 22, 41-44). VDR polymorphism FolkI, Cdx2, and ApaI were not found to have any association with an increased risk of prostate cancer. VDR polymorphism TaqI, BsmI, rs731236, rs7975232, rs731236, rs1544410, rs3782905, rs1544410, and rs2239185 were found associated with increased risk of prostate cancer (Table II). However, the association varies with race (Asian, Caucasian, and African-American) and the genotype- phenotype-based relationship of VDR. Recently, a case-control study investigating the possible associations of VDR polymorphism (FokI, Bsml, Apal and Taqal), and the cytochrome P-45017 alpha (CYP17) gene (the MspA1I locus) with prostate cancer among Lebanese men suggested that the overall polymorphism profile of every gene involved in prostate physiology is a better indicator than polymorphisms in individual genes (45).
Cancer of the Gastrointestinal Tract
An array of epidemiological studies have shown that vitamin D protects cells from carcinogenesis in several solid tumors; however, recent elucidation concerning the possible deleterious effect of high vitamin D levels in the development of EAC has raised concern in this field. VDR polymorphism (rs10735810) representing to a T-to-C transition (ATG to ACG) in exon 2 of the VDR gene has been associated with gastric cancer in Chinese Han population (46). The role of VDR polymorphisms in relation to the risk of Barrett’s oesophagus and EAC has been robustly investigated. Barrett’s oesophagus is characterized by highly dysplastic columnar squamous epithelium near the gastro-oesophageal junction and has been deemed a precursor to EAC. Janmaat et al. in a meta-analysis found that VDR haplotype rs2238135G allele is associated with reduced oesophageal VDR expression and a reduced incidence of gastroesophageal reflux disease (GERD), Barrett’s oesophagus and EAC (12). In another study, 15 haplotype SNPs of the VDR gene were analyzed in patients with GERD, Barrett’s oesophagus or EAC paired with normal histological controls (12). The results demonstrated that there is higher VDR gene expression in tissue from Barrett’s oesophagus compared to healthy control samples. A specific SNP was identified in the VDR gene and patients with this haplotype had a two-fold reduced risk of GERD, Barrett’s oesophagus and EAC (12) (Table III). The mutated allele was found to cause the appearance of a GATA- 1 binding site. GATA-1 turns off transcription, which could explain how the presence of a mutated VDR gene would suppress inflammation and rapid cell proliferation in that region (47). The relationship between vitamin D and oesophageal cancer is essentially the opposite to that seen in other cancerous conditions. Whereas abundant and highly functioning vitamin D/VDR pathways are seen to reduce cancer risk, the likelihood of oesophageal cancer and its affiliated precursor diseases is heightened with robust vitamin D pathway activity. Another meta-analysis also found a significant association of reduced risk of oesophageal cancer with VDR SNPs, suggesting that a dysfunctional vitamin D network is advantageous in oesophageal cancer (48). Understanding the mechanisms of action of the VDR haplotype in oesophageal cancer could be of clinical utility by identifying patients in whom vitamin D chemoprevention would be efficacious. One of the potential explanations for the inverse relationship seen between vitamin D and oesophageal cancer is that VDR has been recently characterized as a bile acid receptor in addition to its well- characterized role as a vitamin receptor. Reflux of bile acid into the most distal parts of the oesophagus is one of the pervasive causes of dysplastic progression into EAC (48, 49).
Vitamin D plays a crucial role in the pathogenesis of colorectal cancer and epidemiological research has put forth evidence that vitamin D plays a protective role against colorectal cancer (50). The role of polymorphisms of VDR and CYP27B2 and CYP24A1, which are involved in the production and suppression of vitamin D, have all been linked to colorectal cancer risk (51). Similarly, a randomized clinical trial conducted with 2259 patients for 41 SNPs of seven genes [VDR, gene encoding vitamin D-binding protein previously known as gc-globulin (GC), 7-dehydrocholesterol reductase (DHCR7), CYP2R1, CYP27B1, CYP24A1 and calcium-sensing receptor (CASR)] concluded that VDR genotype may influence the prevention of advanced colorectal adenomas with vitamin D3 supplementation, suggesting the role of VDR in colorectal carcinoma (52). Another study in Japan found an association between common genetic variations in VDR and risk of colorectal cancer. The researchers selected 29 VDR SNPs and concluded a limited association existed between VDR polymorphism and risk of colorectal cancer (53). Furthermore, another cohort study suggested little effect of the variants in the VDR gene on the risk of colorectal cancer (54). Adenomatous polyps are the precursor for colorectal cancer and a study found FokI to be associated with modified risk for adenomatous polyps in males and association of BsmI/Apal/TaqI haplotype in females (55). The inconsistent results for the association between VDR polymorphism and risk of colorectal cancer indicate the need for further research as well as epidemiological studies in larger cohorts (Table III).
Skin Cancer
The protective effect of vitamin D against ultraviolet radiation-induced DNA damage and skin cancer development and the protective role of vitamin D in reducing skin cancer have been discussed. However, the association between vitamin D and the risk of skin cancer, including melanoma and keratinocyte carcinoma, is still unclear. Vitamin D intake has been positively associated with risk of basal cell carcinoma and melanoma, while non-statistically significantly reducing the incidence of squamous cell carcinoma and melanoma (61, 62). The main risk factors associated with skin cancers are sun exposure and skin phototype, anticarcinogenic and procarcinogenic effects of vitamin D. Along with these factors, VDR polymorphism is also involved in the pathogenesis of skin cancer and may increase the susceptibility for malignancy (14). The association of Fok1, Taq1, Apa1, and Bsm1 polymorphisms with skin cancer including the studies up to 2014 has been discussed by Ombra et al. (63). Recent studies have suggested VDR polymorphism as a risk factor for skin malignancies (14, 15) (Table IV). A meta-analysis showed that VDR polymorphism is associated with the risk of keratinocyte cancer, while rs2228570, rs927650 and rs1544410 recessive genotypes are associated with a reduced risk of squamous cell carcinoma. VDR rs7975232 and rs739837 recessive genotypes were also associated with a reduced risk of basal cell carcinoma (14). Another study found an association of Bsml polymorphism with melanoma in Europeans (15). Furthermore, a meta-analysis by Orlow et al. found a significant association between VDR polymorphism and melanoma-specific survival, however, no significant association was found between Breslow thickness, ulceration or mitosis, and VDR polymorphism (64). These results suggest the need for more research.
Table III. Vitamin D receptor (VDR) polymorphisms and cancer of the gastrointestinal tract.
Urinary Tract Cancer
Vitamin D and VDR are involved in cell division, cell adhesion, and cell function. A normal renal function is necessary for vitamin D metabolism. The role of vitamin D, VDR, and retinoid-X-receptor in inhibition of cell proliferation and angiogenesis and induction of differentiation and apoptosis, and in the pathogenesis of renal cell carcinoma (RCC) has been discussed. Both low and high levels of circulating vitamin D have been associated with higher risk of death from any cause among RCC cases (66-68). This indicates that vitamin D levels may affect the pathogenesis of RCC and VDR might be involved in RCC carcinogenesis. Recent case-control studies by Yang et al. (66) and Pospiech et al. (69) suggest the association of increased risk of RCC with VDR polymorphism. There is experimental and epidemiological evidence that vitamin D has a protective role against bladder cancer, however, the causal relationship is unclear (2, 70, 71). Studying VDR polymorphisms may elucidate the causal relationship of bladder cancer. Recently, Ben Fradj et al. suggest that ff genotype and f allele of FokI polymorphism are associated with increased risk of urothelial bladder cancer (70). Furthermore, the associations of ApaI, BsmI, FokI, and Taq1 polymorphisms with RCC and prostate cancer, Fok1 with bladder cancer, VDR in relation to testicular cancer and testicular germ cell tumors have been reviewed (2). The results of the studies reported in Table V suggest that VDR polymorphisms may be risk factors for urological cancer. However, due to the limited number of studies, the evidence for a clear association between urological cancer and VDR polymorphism is not concrete and more epidemiological studies needed.
Table IV. Vitamin D receptor (VDR) polymorphisms and skin cancer.
Paediatric Tumors
Vitamin D plays a crucial role in regulating cellular homeostasis and proliferation. The role of vitamin D in the pathogenesis of various cancer types has been discussed in the literature as described above in relation to breast, prostate, gastric, and colorectal, etc. in adults. Vitamin D deficiency has been associated with increased prevalence of childhood cancer (72, 73). Vitamin D also plays a role in bone homeostasis and metabolism and its deficiency play a role in inflammation and the pathogenesis of osteoporosis and osteoarthritis (74). Furthermore, the association of osteosarcoma and Ewing sarcoma with a significantly higher frequency of the Ff genotype for the FokI polymorphism suggests the role of VDR polymorphism in bone tumors (75). In a pediatric population, no association of VDR polymorphism and Hodgkin’s lymphoma was found (76). However, in the case of pediatric solid tumors, a weak association between CT and CC genotype of FokI with reduced risk of pediatric solid cancer occurrence was reported (24). Moreover, the association of VDR polymorphic variant rs1544410 with minimal residual disease in pediatric B-cell precursor acute lymphoblastic leukaemia at day 15 suggests a prognostic value as well as the pathogenic role of this VDR polymorphism (77). Furthermore, the case-control studies in Table VI suggest that VDR polymorphism has an association with reduced pediatric solid tumors (FokI), no association with Hodgkin’s lymphoma, and may affect bone mineral density, patient height and overall survival. Vitamin D level and VDR expression may also serve as predictors for the occurrence and overall survival of solid tumors in the pediatric population (24, 75, 76, 78). The effects of VDR polymorphisms on tumourigenesis and other parameters indicate the need for deeper research into the role of VDR polymorphisms and their effects on vitamin D metabolism pertains to the human physiology.
Haematological Cancer
Table V. Urinary tract cancer and vitamin D receptor (VDR) polymorphisms.
A study on the association of plasmablastic B-cell malignancies including plasmablastic lymphoma, subsets of multiple myeloma, and diffuse large B-cell lymphoma and VDR polymorphism found that the Fok1 polymorphism was associated with greater vitamin D3-dependent tumor growth inhibition. The findings of the study suggest the possible therapeutic benefit of the disruption of the VDR pathway activity in order to enhance susceptibility to vitamin D treatment in these malignancies (79). Another study in a Chinese Han population on the association of VDR polymorphisms rs2228570 (FokI) and rs731236 (TaqI) and multiple myeloma susceptibility including 113 patients and 117 healthy controls reported that rs2228570 TT genotype and mutant C allele of rs731236 are significantly associated with increased risk of multiple myeloma. The study concluded that the polymorphisms rs2228570 and rs731236 might be important genetic factors in the susceptibility to multiple myeloma (80). Esfahani et al. studied the association of VDR polymorphisms (FokI, BsmI, TaqI and ApaI) with acute myeloid leukaemia involving 133 patients and 300 controls and found a significant association between VDR polymorphism and acute myeloid leukaemia, and association of Taql with complete remission (81).
Additional Cancer-related Interactions of VDR polymorphisms
Along with cancer of female genital tract, prostate, gastrointestinal tract, urological tumors, skin cancer, and pediatric tumors, VDR polymorphisms may also play a role in the carcinogenesis of lung and thyroid cancer. A case- control study including 426 patients with non-small cell lung cancer (NSCLC) and 445 controls from China studying VDR polymorphisms rs1544410 and rs731236 found a statistically significant relation between VDR polymorphism and reduction in NSCLC risk (82). Furthermore, the association of increased VDR mRNA expression with worse prognostic factors in papillary thyroid carcinoma suggests the role of VDR in thyroid cancer (83). Since vitamin D deficiency is associated with thyroid diseases and its metabolism is regulated by thyroid hormone, VDR polymorphism may play a role in pathogenesis and prognosis of thyroid cancer (84). A recent meta-analysis found a clear and strong association between low levels of baseline vitamin D and poorer survival and an association between VDR genetic variants and survival [rs7975232 (ApaI) with progression-free survival; rs1544410 (BsmI) with overall survival; and rs2228570 (Fokl) with overall survival] in lung cancer. However, from all the observational studies included in this meta-analysis, a causal relationship was not conclusively established (85).
Conclusion
The results of various studies suggest the crucial role of VDR polymorphism (mainly FokI, ApaI, BsmI, and Taql) in tumorigenesis of various cancer types by affecting vitamin D metabolism and the cellular response to vitamin D. From the above-discussed studies, it is also obvious that the association between VDR polymorphism and tumorigenesis varies with
age,
sex,
race and
ethnicity.
The results of these studies also suggest that studying VDR polymorphisms in order to strengthen our understanding of the vitamin D pathway could provide additional evidence for its protective therapeutic role against cancer development (33, 79). VDR polymorphisms might also serve as indicators for diagnosis, occurrence, and prognosis as well as survival in cancer (24, 64, 75-78, 83-85). There have been many epidemiological studies of breast, prostate and colorectal cancer, however, there are limited reports on the association of VDR polymorphism with lung, thyroid, oesophageal, ovarian, renal and hepatocellular carcinoma. These studies have shown the association of VDR polymorphism with increased risk of cancer, reduced risk of cancer, as well as no association. Thus, there is a need for more studies with larger cohorts in order to establish the significant associations and causal relationships between VDR polymorphism and cancer. Additionally, genetic variation in the vitamin D pathway should be considered when designing potential intervention strategies with vitamin D supplementation.
