Topical Vitamin D
Topical is one of the many ways of increasing the vitamin D in your body
Topical is 2X to 50X more expensive than oral form
Topical is great for those who not want to swallow pillls nor fortifiy their food/drink with Vitamin D
Topical is one of the many gut-friendly forms of vitamin D (good bioavailability for those with poor guts
Topical is especially good form to apply directly to skin problems:
- i.e. Warts Burns Eczema Wounds (lasts about a day)
The skin, like most tissues of the body, can fully-activate Vitamin D locally - no liver nor kidney needed.
Provides perhaps 100X higher concentration of Vitamin D to that portion than if same dose were taken orally
There were 98 topical Vitamin D products on Amazon Sept 2024 Creams, oils, patches, nanoemulsions, etc.
I prefer Nutrasal nanoemulsion form
Suspect that topical Vitamin D is best absorbed where the skin gets Vitamin D naturally
- - - face and hands vs the back or the inside of arms
Nanoemulsions can also be applied topically under the tongue and inhaled
Topical in VitaminDWiki
Topical Vitamin D provides more benefits than oral sometimes - many studies
Psoriasis paradigm shift – use topicals, like Vitamin D – Aug 2021
Topical processed by CYP11A1 is not detected by Vitamin D blood test
Skin category listing has items along with related searches
Psoriasis category listing has items along with related searches
Topical vitamin D raised blood level to 38 ng (used Aloe Vera gel) – RCT March 2014
Topical vitamin D might be more bio-available than oral – Oct 2015
Transcutaneous enhancers (DMSO, etc) for Vitamin D, Vitamin C, Resveratrol, Magnesium, etc.
Getting Vitamin D into your blood and cells topical is one of many ways
28+ VitaminDWiki pages having TOPICAL in the title
This list is automatically updated
{LIST()}
Off-label uses of topical vitamin d in dermatology: a systematic review - 2014
J Cutan Med Surg. 2014 Mar 1;18(2):91-108.
Wat H, Dytoc M.
Background:Topical vitamin D is approved by the US Food and Drug Administration for the treatment of psoriasis but is also used off-label in the treatment of a variety of cutaneous diseases despite a lack of evidence-based guidelines.
Objective:The objective of this study was to provide evidence-based clinical guidelines for the off-label use of topical vitamin D in the treatment of dermatologic disease.Methods:A systematic literature review was conducted via the MEDLINE, Embase, and CENTRAL databases for off-label uses of topical vitamin D analogues in the treatment of dermatologic disease other than psoriasis. The data were synthesized, and evidence-based recommendations were rendered according to the highest level of evidence available.
Results:A total of 165 articles met the inclusion criteria.
A moderate to strong recommendation was given for the use of
topical vitamin D in combination with corticosteroids and phototherapy in vitiligo and as
monotherapy for various
- ichthyoses, [dry, scaling skin that may be thickened or very thin]
- morphea, [ isolated patches of hardened skin]
- pityriasis alba, [dry, fine-scaled, pale patches on the face]
- prurigo nodularis, [(itchy) nodules which usually appear on the arms or legs] and
- polymorphous light eruption. [itchy rash caused by sun exposure in people who have developed a sensitivity to sunlight]
There is evidence showing that topical vitamin D is ineffective in the treatment of
actinic keratosis,
seborrheic keratosis, [premalignant condition of thick, scaly, or crusty patches of skin.]
lichen planus,
seborrheic dermatitis,
alopecia areata, c
hemotherapy-induced alopecia, and
hypertrophic scars.
Conclusion:Topical vitamin D analogues have an important role in the off-label treatment of dermatologic disease, but higher quality studies are still required.
Topical 5,000 IU raised blood levels to 37 ng - RCT Oct 2018
Topical vitamin D3: A randomized controlled trial (RCT)
Clin Nutr ESPEN. 2018 Oct;27:16-19. doi: 10.1016/j.clnesp.2018.05.009. Epub 2018 Jun 28. PMID: 24636434
Topical vitamin D3: A randomized controlled trial (RCT).
Bubshait DA1, Al-Dakheel DA2, Alanii FM2.
1 Department of Orthopaedic Surgery, College of Medicine, Imam AbdulRahman Bin Faisal University, Dammam; King Fahd Hospital of the University, Al Khobar, Saudi Arabia. Electronic address: dbubshait@iau.edu.sa.
2 Department of Orthopaedic Surgery, College of Medicine, Imam AbdulRahman Bin Faisal University, Dammam; King Fahd Hospital of the University, Al Khobar, Saudi Arabia.
Note: For some people the vitamin D levels were DECREASED
Note: Vitamin D levels can be decreased for oral Vitamin D supplementation as well for some people
📄 Download the PDF from VitaminDWiki
OBJECTIVE: The intent of this study was to test the effect of Top-D, a topical Vitamin D preparation, in delivering vitamin D.
METHODS:
Five hundred and fifty healthy patients, with vitamin D insufficiency and deficiency were recruited after written informed consent. Demographic data was recorded, adequate history and clinical examination was done to rule out any metabolic diseases. Complete blood picture, serum calcium, phosphorous, Parathormone and 25 Hydroxy-vitamin D3 (25OHD) was carried out before enrollment of the patients. Patients were divided randomly into two groups 350 in study group and 200 in the control group. Patients in the study group were given Top-D (Vitamin D3 gel made from proniosomal technology) to apply daily on the skin. Top-D 1 g contained 5000 IU of vitamin D3. The control group was given 1 g of Aloe vera gel to be applied every day. The two groups had no knowledge to which group they belong. After 4 months serum 25OHD was tested again.
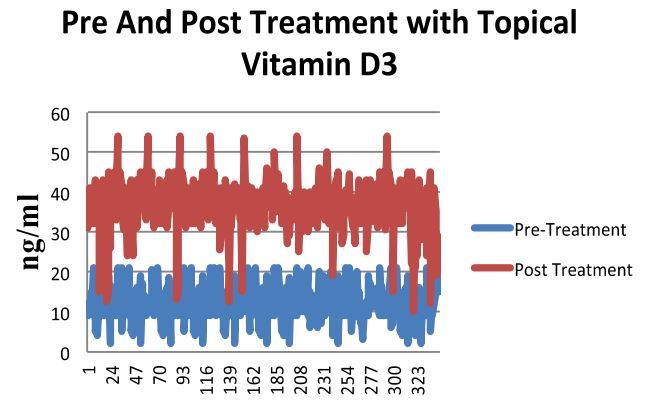
RESULTS: Three hundred and forty five patients in study group and 192 in control group completed the study. The mean age of the patients in the both the groups was 42 years (18-80 years). The pretreatment 25OHD level in the study group was 11.03 ± 4.57 (2-12) ng/l compared to the control group 10.36 ± 4.09 (2-21) and post treatment the levels were 37.17 ± 6.04 (12-54) ng/ml and 10.51 ± 3.5 (2-19) ng/ml (p < 0.001).
CONCLUSION: The results of this study indicate that transdermal route of vitamin D is potentially, safe and can give desired results to raise the vitamin D levels. This route is an alternate route for supplementation of vitamin D which should be utilized.
Seborrheic Keratoses treated by topical Vitamin D, etc. - Nov 2017
Managing Seborrheic Keratoses: Evolving Strategies for Optimizing Patient Outcomes.
J Drugs Dermatol. 2017 Nov 1;16(11):1064-1068. PMID: 29141054
Ranasinghe GC, Friedman AJ.
The seborrheic keratosis is the most common benign skin tumor of middle-aged and elderly adults, affecting nearly 83 million individuals in the US alone. Although these are benign lesions, many patients still undergo some form of treatment. Clinicians are frequently presented with a challenge when determining whether to remove a seborrheic keratosis, and which treatment modality to use when doing so. The most commonly used method of removal is cryotherapy, however there are numerous other options that can be employed with varying degrees of efficacy. In this article, we highlight the use of topical keratolytics, vitamin D analogues, and lasers, to name a few. We also address potential side effects associated with these treatment options, as well as discuss patients' preferences and concerns. We conclude with the most recent advances in topical treatments currently under clinical investigation, and offer treatment strategies aimed at maximizing patient satisfaction.
Transdermal vitamin D supplementation—A potential vitamin D deficiency treatment - July 2019
Journal of Cosmetic Dermatology DOI: 10.1111/jocd.13085
Sujata Sawarkar MPharm, PhD© | Ashwini Ashtekar MPharm ©
Department of Pharmaceutics, SVKM's Dr. Bhanuben Nanavati College of Pharmacy, University of Mumbai, Mumbai, India
Correspondence
Sujata Sawarkar, Department of Pharmaceutics, SVKM's Dr. Bhanuben Nanavati College of Pharmacy, University of Mumbai, Gate No.1, Mithibai College Campus, V.M. Road, Navpada, Suvarna Nagar, Vile Parle West, Mumbai 400056, Maharashtra, India.
Emails: sujata.sawarkar@bncp.ac.in; sujatasawarkar19@gmail.com
Vitamin D deficiency has high prevalence worldwide. Vitamin D3, the active form of vitamin D, exhibits array of roles in body, from calcium homeostasis and bone mineralization to cancer, neurological disorders, immunomodulatory action, and cardiac health. Current approaches for supplementing vitamin D3 are restricted to oral and parenteral routes. This review highlights recent research in the field of transdermal delivery of vitamin D, its active form and analogues with the aid of penetration enhancers and novel carrier system as nutritional supplement in case of vitamin D deficiency. The penetration of vitamin D3 is challenging; however, by means of reducing hydrophobicity of the active and encapsulating vitamin D3 in a suitable carrier system, penetration is achieved . The results show that penetration of vitamin D3 through skin is feasible. Further clinical trials could strengthen these results. However, the present research till date shows transdermal vitamin D3 a promising way of supplementation.
📄 Download the PDF from Sci-Hub via VitaminDWiki
See also" inhaled Vitamin D
Perhaps drugs such as inhaled vitamin D can get directly to the brain – March 2018
Vitamin D prevents smoke lung damage in mice (If you must smoke, take vitamin D) – Nov 2019
Liposomal Vitamin D cocktail targets some lung cancers – June 2018
Lung cells activate Vitamin D, without liver or kidney – Nov 2016
- Perhaps the tissues of the nose and the brain can also activate Vitamin D
Title of this page was revised July 2019 which caused the visitor count to reset.
There have actually been visitors to this page since it was originally made- Perhaps the tissues of the nose and the brain can also activate Vitamin D

