Vitamin D Receptor Activator might help treat or prevent various diseases
Your vitamin D blood test can be good, but poor Vitamin D receptors will reduce the cell benefit
- Vitamin D Receptor category has the following
{include}
Apparently you might also try to get a prescription for a Vitamin D Receptor Activator
Activators appear to include paricalcitol, Maxacalcitol, : some are oral, some are injection
unknown amount of benefit from VDRA
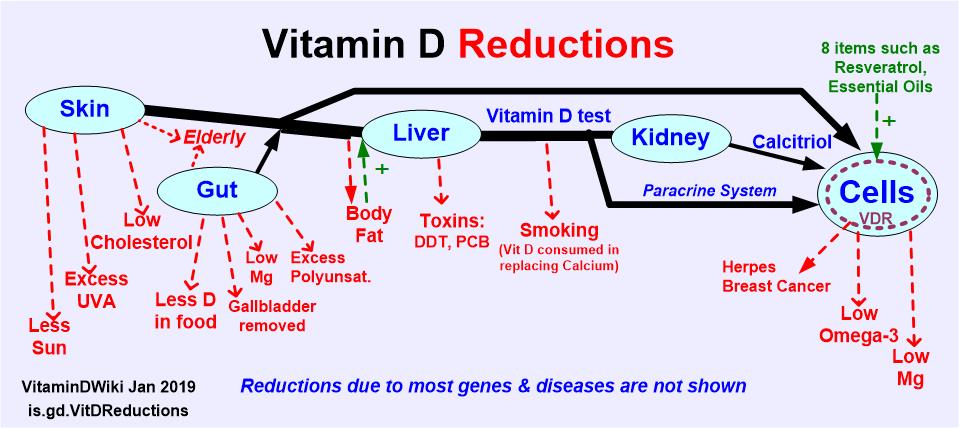
click on chart for details
See also VitaminDWiki
Vitamin D Receptor category listing has items along with related searches
Health problems that run in families are often associated with low vitamin D
- Perhaps a VDR gene problem runs in the family
Many autoimmune diseases associated with latitude and vitamin D receptor – March 2016
Vitamin D level can be high, but little benefit: due to kidney, genes, low Magnesium etc.
Vitamin D dose size needed – VitD testing tells only a portion of the story – Jan 2016
Vitamin D Receptor activators (such as paricalcitol) treat dialysis patients similarly – Aug 2017
PubMed: "Vitamin D Receptor Activator " 89 items as of Oct 2018
Predialysis Vitamin D Receptor Activator Treatment and Cardiovascular Events after Dialysis Initiation: A Multicenter Observational Study – April 2016
Nephron, (DOI:10.1159/000445507)
Inaguma D. · Tanaka A. · Shinjo H. · Kato A. · Murata M. · Aichi cohort study of prognosis in patients newly initiated into dialysis (AICOPP) group
Department of Nephrology, Japanese Red Cross Nagoya Daini Hospital, Nagoya, Japan
Background: Vitamin D receptor activator (VDRA) administration has been linked with a reduced incidence of cardiovascular disease (CVD). However, it is unclear whether VDRA administration during the predialysis stage is associated with CVD incidence after dialysis initiation in patients with chronic kidney disease. Therefore, we examined the association between VDRA use and CVD events.
Methods: This multicenter observational study included 1,516 patients; they were divided into 2 groups: those who did and did not receive oral VDRA for at least 3 months before dialysis initiation. The CVD incidence was compared between these groups. Factors that impacted CVD incidence were extracted through a multivariate analysis. Subgroups were created based on prior CVD history and serum CRP levels.
Results: The incidence of CVD was significantly lower in the VDRA group (log-rank test, p = 0.014). Stepwise multivariate analyses identified age, gender, diabetes, CVD history, calcium-channel blockers, beta-blockers, loop diuretics, anti-platelet agents, phosphate binders, VDRA, erythropoiesis stimulating agents, and cardiothoracic ratio as factors affecting CVD incidence. In the group with no CVD history, VDRA use was associated with a low incidence of CVD (HR 0.35). In the group with serum CRP levels <1.0 mg/dl, VDRA use was associated with a low incidence of CVD (HR 0.47).
Conclusion: Administration of VDRA during predialysis was associated with a low incidence of CVD onset after dialysis initiation.
The pleiotropic effects of paricalcitol: Beyond bone-mineral metabolism – Jan 2016
Nefrologia. 2016 Jan-Feb;36(1):10-18. doi: 10.1016/j.nefro.2015.11.003. Epub 2015 Dec 23.
[Article in English, Spanish]
Egido J1, Martínez-Castelao A2, Bover J3, Praga M4, Torregrosa JV5, Fernández-Giráldez E6, Solozábal C7.
Secondary hyperparathyroidism (SHPT) is a common complication in patients with chronic kidney disease (CKD) that is characterised by elevated parathyroid hormone (PTH) levels and a series of bone-mineral metabolism anomalies. In patients with SHPT, treatment with paricalcitol, a selective vitamin D receptor activator, has been shown to reduce PTH levels with minimal serum calcium and phosphorus variations. The classic effect of paricalcitol is that of a mediator in mineral and bone homeostasis. However, recent studies have suggested that the benefits of treatment with paricalcitol go beyond PTH reduction and, for instance, it has a positive effect on cardiovascular disease and survival. The objective of this study is to review the most significant studies on the so-called pleiotropic effects of paricalcitol treatment in patients with CKD.
Copyright © 2015 Sociedad Española de Nefrología. Published by Elsevier España, S.L.U. All rights reserved.
📄 Download the PDF from VitaminDWiki
The Vitamin D Receptor Activator Maxacalcitol Provides Cardioprotective Effects in Diabetes Mellitus. – Dec 2015
Cardiovasc Drugs Ther. 2015 Dec;29(6):499-507.
Fujii H1, Nakai K2, Yonekura Y2, Kono K2, Goto S2, Hirata M3, Shinohara M4, Nishi S2, Fukagawa M2,5.
PURPOSE:
Recent reports showed a significant association between vitamin D levels and cardiovascular disease events and mortality. In the current study, we investigated the effect of the vitamin D receptor activator maxacalcitol (OCT) on cardiac damage in a rat model of type 2 diabetes.
METHODS:
At 20 weeks of age, the rats were divided into three groups: vehicle-treated (DM), insulin-treated (INS) and OCT-treated (OCT). At 30 weeks, the rats were sacrificed and urinary and blood biochemical analyses and cardiac histological and immunohistochemical analyses were performed. To evaluate the effect of OCT on the renin-angiotensin system, we performed a further study using aliskiren (ALS). At 20 weeks, the diabetic rats were divided into two groups: the ALS-treated group (ALS) and the ALS plus OCT-treated group (ALS + OCT), and we evaluated the renin-angiotensin system (RAS) and cardiac lesions at 30 weeks.
RESULTS:
At 30 weeks, despite comparable blood pressure and renal function, heart volume, intracardiac oxidative stress by immunohistological analysis, cardiac and perivascular fibrosis and urinary excretion of 8-hydroxydeoxyguanosine and serum N-terminal pro-brain natriuretic peptide levels were significantly decreased in the OCT group compared to the DM group. mRNA expressions of dihydronicotinamide adenine dinucleotide phosphate (NADPH) p47 subunit and cardiac injury-related markers in the heart were also significantly decreased in the OCT group compared to the DM group. The cardioprotective effect of OCT was preserved even in the context of RAS inhibition.
CONCLUSION: Our results suggest that OCT prevents the development of cardiac damage in DM, independent of RAS inhibition.
Use of vitamin D receptor activator, incident cardiovascular disease and death in a cohort of hemodialysis patients – June 2015
Ther Apher Dial. 2015 Jun;19(3):235-44. doi: 10.1111/1744-9987.12274. Epub 2014 Dec 19.
Shoji T1,2, Marubayashi S2, Shigematsu T2, Iseki K2, Tsubakihara Y2; Committee of Renal Data Registry; Japanese Society for Dialysis Therapy.
The use of vitamin D receptor activators (VDRAs) is an independent predictor of a lower risk of death from cardiovascular disease (CVD) in patients with chronic kidney disease (CKD). We examined whether the use of VDRAs and other CKD-mineral bone disorder (MBD)-related factors are associated with incident CVD or death after CVD in hemodialysis patients. This is a historical cohort study of 37 690 prevalent hemodialysis patients without previous history of CVD at the end of 2004 extracted from a nationwide registry in Japan. The key exposure was the use of VDRAs, and the outcomes were incident CVD (myocardial infarction, cerebral infarction, cerebral hemorrhage, and sudden death) and death after CVD during the 1-year follow-up. VDRAs were used in 57% of the subjects at baseline. We identified 2433 patients with incident CVD and 397 deaths after the events. In multivariate logistic regression models, independent predictors of incident CVD were non-use of VDRA, higher intact PTH, non-use of calcium-based phosphate-binder, and non-use of non-calcium-based phosphate binder. Risk of death after CVD was not significantly associated with VDRA, whereas it was lower in those with lower corrected calcium, and the risk was higher in those with higher phosphate and in non-users of calcium-based phosphate binders. The use of VDRAs was associated with a lower risk of incident CVD but not with death after CVD in this large cohort of hemodialysis patients. The CKD-MBD-related predictors of poor outcomes are associated with the risk of incident CVD, the risk of death after CVD, or both.
Can read at DeepDyve
Ability of vitamin D receptor activator to prevent pulmonary congestion in advanced chronic kidney disease. – June 2015
Clin Exp Nephrol. 2015 Jun;19(3):371-8. doi: 10.1007/s10157-014-0994-x. Epub 2014 Jun 11.
Sueta S1, Morozumi K2, Takeda A2, Horike K2, Otsuka Y2, Shinjo H2, Murata M2, Kato Y2, Goto K2, Inaguma D2; Aichi cohort study of prognosis in patients newly initiated into dialysis (AICOPP) study group.
Collaborators (19)
BACKGROUND:
Vitamin D deficiency is common among patients with chronic kidney disease (CKD). However, the benefits of vitamin D supplementation versus vitamin D receptor activator (VDRA) administration have yet to be established. Recently, an association between activated vitamin D and cardiovascular factors was reported. To evaluate the benefits of VDRA in advanced CKD, we analyzed the association between VDRA administration and the prevalence of pulmonary congestion.
METHODS:
This retrospective, cross-sectional analysis included patients initiated on dialysis between October 2011 and September 2013 at 17 Japanese institutions. Data from 952 participants were analyzed using a multivariate logistic regression model and a linear regression model. We also analyzed subgroup data for groups classified by selection of peritoneal dialysis or hemodialysis.
RESULTS:
Of the 952 participants, 303 patients received VDRA. VDRA administration was associated with a low prevalence of pulmonary congestion in the multivariate logistic regression model (odds ratio [OR], 0.64; 95 % confidence interval [CI], 0.44-0.94; P = 0.02). There was no significant association between VDRA administration and systolic blood pressure, diastolic blood pressure, or pulse pressure. Subgroup analysis revealed a tendency that VDRA administration was associated with low prevalence of pulmonary congestion in both groups.
CONCLUSIONS:
In this study, VDRA administration was associated with a low prevalence of pulmonary congestion in patients initiated on dialysis. Appropriate VDRA administration may prevent pulmonary congestion.
The influence of selective vitamin D receptor activator paricalcitol on cardiovascular system and cardiorenal protection.= 2013
Clin Interv Aging. 2013;8:149-56. doi: 10.2147/CIA.S38349. Epub 2013 Feb 11.
Duplancic D1, Cesarik M, Poljak NK, Radman M, Kovacic V, Radic J, Rogosic V.
The ubiquitous distribution of vitamin D receptors in the human body is responsible for the pleiotropic effects of vitamin D-receptor activation. We discuss the possible beneficial effects of a selective activator of vitamin D receptor, paricalcitol, on the cardiovascular system in chronic heart failure patients and chronic kidney patients, in light of new trials. Paricalcitol should provide additional clinical benefits over the standard treatment for chronic kidney and heart failure, especially in cases of cardiorenal syndrome.
📄 Download the PDF from VitaminDWiki
Comparison of oral versus intravenous vitamin D receptor activator in reducing infection-related mortality in hemodialysis patients: the Q-Cohort Study - June 2016
6 times decrease in mortality with Intravenous VDRA
Nephrol. Dial. Transplant. (2016) 31 (7): 1152-1160. doi: 10.1093/ndt/gfw205
Shigeru Tanaka1,2, Toshiharu Ninomiya2,3, Masatomo Taniguchi2, Kiichiro Fujisaki2, Masanori Tokumoto1, Hideki Hirakata4, Hiroaki Ooboshi1, Takanari Kitazono2,3 and Kazuhiko Tsuruya2,5
Background Hemodialysis patients who receive vitamin D receptor activator (VDRA) reportedly have better survival after infection than those who do not. However, the optimal route of its administration for minimizing death from infection remains unclear.
Methods This prospective cohort study aimed to compare the effectiveness of oral versus intravenous VDRA regarding infection-related mortality in 3372 hemodialysis patients. Eligible subjects were divided into the following three groups by route of administration of VDRA: oral (n = 1868), intravenous (n = 492) and not administered (n = 1012). The effect of VDRA on infection-related mortality was examined using a Cox regression model with propensity score-based adjustments.
Results During follow-up (median, 4.0 years), 118 study patients died of infection. There was a significantly lower incidence of death from infection in subjects who received intravenous VDRA than in those who did not receive VDRA; however, oral VDRA did not significantly reduce the risk of mortality from infection compared with those who did not receive VDRA [hazard ratio (HR) for intravenous VDRA, 0.16 ; 95% confidence interval (CI), 0.10–0.25, and HR for oral VDRA, 0.78; 95% CI, 0.60–1.01]. Direct comparison between the oral and intravenous VDRA groups showed that the intravenous group had significantly better survival than the oral group (HR, 0.39; 95% CI, 0.27–0.62).
Conclusions Treatment with intravenous VDRA more effectively reduces the incidence of mortality from infection than oral VDRA in hemodialysis patients.
