Drug–Vitamin D Interactions, A Systematic Review
Nutr Clin Pract January 10, 2013 0884533612467824
Kim Robien, PhD, RD, CSO 1
Sarah J. Oppeneer, MS 2
Julia A. Kelly, MS, MLIS 3
Jill M. Hamilton-Reeves, PhD, RD 4
1 Department of Epidemiology and Biostatistics, George Washington University School of Public Health and Health Services, Washington, DC
2 Division of Epidemiology and Community Health, University of Minnesota, Minneapolis
3 University Libraries, University of Minnesota, Minneapolis
4 University of Kansas Medical Center, School of Health Professions, Department of Dietetics & Nutrition, Kansas City, Kansas
Kim Robien, PhD, RD, CSO, Associate Professor, Department of Epidemiology and Biostatistics, George Washington University School of Public Health and Health Services, 2100-W Pennsylvania Ave NW, 8th Floor, Washington, DC, 20037, USA. Email: krobien@gwu.edu.
Extensive media coverage of the potential health benefits of vitamin D supplementation has translated into substantial increases in supplement sales over recent years. Yet, the potential for drug–vitamin D interactions is rarely considered. This systematic review of the literature was conducted to evaluate the extent to which drugs affect vitamin D status or supplementation alters drug effectiveness or toxicity in humans. Electronic databases were used to identify eligible peer-reviewed studies published through September 1, 2010 . Study characteristics and findings were abstracted, and quality was assessed for each study.
A total of 109 unique reports met the inclusion criteria.
The majority of eligible studies were classified as class
C (nonrandomized trials, case-control studies, or time series) or
D (cross-sectional, trend, case report/series, or before-and-after studies).
- Only 2 class C and 3 class D studies were of positive quality.
Insufficient evidence was available to determine whether
lipase inhibitors,
antimicrobial agents,
antiepileptic drugs,
highly active antiretroviral agents, or
H2 receptor antagonists
- alter serum 25(OH)D concentrations.
{However,}
Atorvastatin appears to increase 25(OH)D concentrations, whereas
concurrent vitamin D supplementation decreases concentrations of atorvastatin.
Use of thiazide diuretics in combination with calcium and vitamin D supplements
- may cause hypercalcemia in the elderly or those with compromised renal function or hyperparathyroidism.
Larger studies with stronger study designs are needed to clarify potential drug–vitamin D interactions,
especially for drugs metabolized by cytochrome P450 3A4 (CYP3A4) .
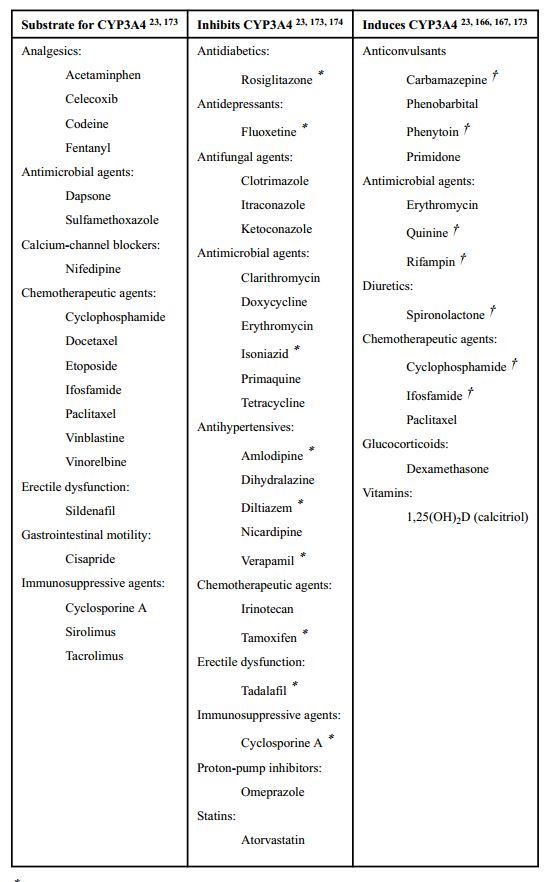
Healthcare providers should be aware of the potential for drug–vitamin D interactions.
Partial PDF Table of Contents

📄 Download the PDF from VitaminDWiki
VitaminDWiki - Interactions with Vitamin D contains
{include}
