Lupus
Lupus and Vitamin D Overview
items in LUPUS category
Some of the studies in VitaminDWiki
Every aspect of Lupus is associated with low viramin D – Oct 2021
It is time to routinely give vitamin D to Lupus patients – Dec 2016
93 pcnt of these Lupus patients supplemented with Vitamin D - Nov 2020
Lupus patients are 5 X more likely to have low vitamin D – many meta-analyses
Lupus fought by Vitamin D in all trials longer than 12 weeks - review Sept 2017
See also Autoimmune: items Rheumatoid Arthritis: items
Autoimmune disease, mainly in 50 per 100,000 women, no cure, many parts of body Wikipedia
People with Lupus benefit from supplementation if they have low levels of Vitamin D – Sept 2024
Vitamin D and systemic lupus erythematosus: Causality and association with disease activity and therapeutics
Biochemical Pharmacology Volume 227, September 2024 https://doi.org/10.1016/j.bcp.2024.116417
The major role of bioactive vitamin 1,25-dihydroxyvitamin D3 (1,25(OH)2D or calcitriol) is to maintain the levels of calcium and phosphorus to achieve bone and mineral homeostasis. Dietary intake and adequate natural light exposure are the main contributors to normal vitamin D status. In addition to regulating metabolism, vitamin D exerts various immunomodulatory effects that regulate innate and adaptive immunity through immune effector cells such as monocytes, macrophages, T and B lymphocytes, and natural killer cells and nonimmune cells that express vitamin D receptors. Systemic lupus erythematosus (SLE) is an autoimmune disease with an unknown etiology, and the association between vitamin D and SLE remains incompletely understood. Given that the current treatment for SLE relies heavily on corticosteroids and that SLE patients tend to have low vitamin D status, vitamin D supplementation may help to reduce the dosage of corticosteroids and/or attenuate disease severity. In this review, we address the associations between vitamin D and several clinical aspects of SLE. In addition, the underlying immunomodulatory mechanisms accounting for the potential vitamin D-mediated therapeutic effects are discussed. Finally, several confounding factors in data interpretation and the execution of clinical trials and perspectives targeting vitamin D supplementation in patients with SLE are also addressed.
Introduction
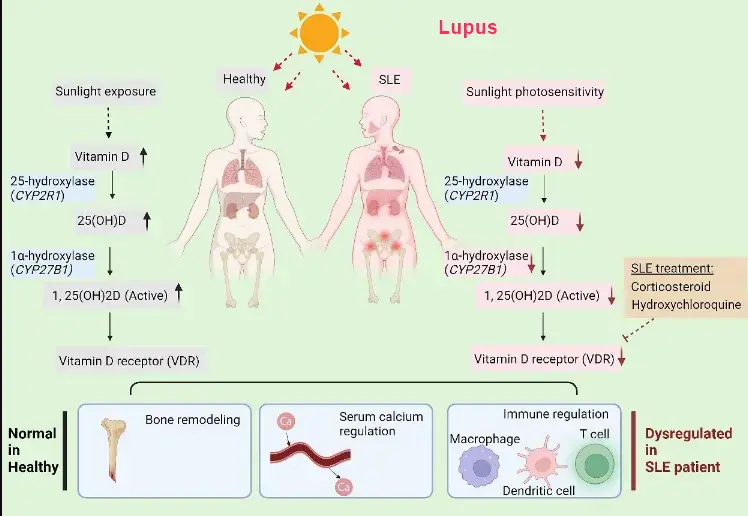
The term “vitamin D” is a group of fat-soluble secosteroids and has been collectively referred to two very similar molecules, namely vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). Both vitamin D2 and vitamin D3 can be obtained from food or provided as supplements and vitamin D3 can also be generated in the skin [1], [2]. In the skin exposed to sunlight, 7-dehydrocholesterol (7DHC) undergoes a photochemical reaction and transforms after absorbing ultraviolet B (UVB) energy to become pre-vitamin D3. Pre-vitamin D3 is thermodynamically unstable and following a thermal isomerization step, pre-vitamin D3 becomes vitamin D3 [3], [4]. Exposure of pre-vitamin D3 to higher doses of UVB may result in the formation of lumisterol and tachysterol. In general, only vitamin D3 is recognized as a prohormone, and lumisterol, tachysterol, and other derivatives are considered as biologically inert compounds or degradative products [3]. Vitamin D3 and vitamin D2 from oral intake or from the skin synthesis are biologically inactive and their activation requires the participation of cytochrome P450 (CYP) hydroxylation enzymes that are primarily, but not exclusively, in the liver and the kidney. In the liver, vitamin D3 or vitamin D2 is converted to calcifediol (25-hydroxycholecalciferol, 25(OH)D3) or ercalcidiol (25-hydroxyergocalciferol, 25(OH)D2), respectively, by mainly the hydroxylase CYP2R1 or CYP27A1 [3]. Many enzymes preserve 25-hydroxylase activity [2]; however, CYP2R1, as the major enzyme responsible for 25(OH)D (referred collectively for 25(OH)D2 and 25(OH)D3) production in physiological conditions, contributes to more than 50 % of blood circulating 25(OH)D, as the results demonstrated in Cyp2r1-knockout mice [5].
CYP2R1 25-hydroxylates both vitamin D2 and vitamin D3 with comparable kinetics; however, CYP27A1 does not 25-hydroxylate vitamin D2 [2]. Both 25(OH)D3 and 25(OH)D2 are the major substrates for production of calcitriol (1,25-dihydroxyvitamin D3, 1,25(OH)2D3) and 1,25-dihydroxyvitamin D2 (1,25(OH)2D2), respectively, by the enzyme 25(OH)D 1α-hydroxylase (CYP27B1) in the kidney and other tissues or cells like immune cells expressing CYP27B1 [6]. Vitamin D metabolites produced in local tissues are transported to target organs by binding to vitamin D binding protein (DBP), and about 85 % of the circulating 25(OH)D and 1,25(OH)2D (referred collectively for 25(OH)2D2 and 25(OH)2D3) are bound by DBP. In comparison, vitamin D2 and its metabolites have lower affinity for DBP and are cleared faster from the bloodstream than vitamin D3 and its metabolites [2]. Both 1,25(OH)2D3 and 1,25(OH)2D2 are the dominant and biologically active vitamin D metabolites in human bodies and have comparable affinities for the vitamin D receptor (VDR) [7], [8] (Fig. 1). Noticeably, vitamin D2 supplementation cannot reach as high blood 25(OH)D concentration as comparable amounts of vitamin D3 [9]. The levels of 25(OH)D circulating in the blood have been frequently used to reflect a person's nutritional vitamin D status although the measurement of active 1,25(OH)2D can be of use in some clinical conditions [10].
CYP27B1 can be activated by parathyroid hormone (PTH), whereas fibroblast growth factor 23 (FGF 23), high calcium and phosphate inhibits CYP27B1 in the kidney. In addition, 1,25(OH)2D can regulate its own production through inhibiting CYP27B1 expression and PTH production, stimulating FGF 23 production, and activating CYP24A1, a 24-hydroxylase found in most tissues, that is critical in mediating catabolism of 25(OH)D and 1,25(OH)2D [11], [12], [13]. Together, CYP24A1 and CYP27B1 tightly regulate levels of 1,25(OH)2D and calcium and phosphate concentrations [12]. The regulation of 1,25(OH)2D production in non-renal cells is different from that in renal cells suggesting the possibility that 1,25(OH)2D may function as an autocrine and/or paracrine in these cells [14]. Aside from that 1,25(OH)2D can regulate CYP27B1 expression in tissues like the skin or parathyroid glands with much less efficiency, in most non-renal tissues, CYP27B1 may not respond to PTH or 1,25(OH)2D, whereas cytokines such as tumor necrosis factor (TNF-α) and interferon-γ (IFN-γ) appear to play critical roles in regulating CYP27B1 expression [14]. In immune cells such as monocytes/macrophages and lymphocytes, the regulation of CYP27B1 is strikingly affected by inflammatory conditions, for example, responding to infection by pathogens [11].
VDR is a member of a multi-protein complex comprising, for example, co-receptors (RXR), pioneer factors such as PU.1, CEBPα, GABPα, ETS1, RUNX2, and BACH2, chromatin modifiers like KDM1A and KDM6B, chromatin remodelers like BRD7 and BRD9, co-activators (MED1) and co-repressors such as NCOR1 and COPS2 [15]. The chromatin-remodeling proteins in the complex function as modifiers to help and to facilitate VDR to bind to the enhancer region of target genes or to modulate the epigenome of its target tissues involving chromatin opening, histone marks and the recruitment of pioneer factors [15]. Specific activation of VDR by 1,25(OH)2D drives the activity of other members of the complex [16], [17]. The VDR-mediated signaling activates and regulates the expression of various genes such as those involved in maintaining the levels of calcium and phosphate in the blood and bone content in cooperation with PTH and calcitonin [18], [19]. In addition to regulating calcium and phosphorus homeostasis, expressed in immune cells like monocytes, activated TT and B cells, VDR can modulate many cellular processes such as cell proliferation and cell differentiation, programmed-cell death, photoprotection, and immune responses [20], [21]. Noticeably, through binding to more than 10,000 loci within the human genome, VDR can transcriptionally regulate the activation/inhibition of nearly 1000 target genes covering a large proportion of different cell types and human tissues [22]. Nevertheless, the epigenomic and transcriptomic analysis suggests that the vast majority of genomic VDR binding sites are likely contributing to epigenetic memory of a cell rather than directly to gene transcription [23]. Given that 1,25(OH)2D are potent activators for VDR, other forms of vitamin D metabolites bind to VDR with much lower affinity. Binding of 1,25(OH)2D to the ligand-binding pocket comprising 40 mostly non-polar amino acids of VDR at sub-nanomolar concentrations (KD 0.1 nM) suggests that VDR is the exclusive mediator for 1,25(OH)2D to exert its effects at physiological concentrations [24], [25].
Both canonical and noncanoical pathways are operating in regulating metabolism of vitamin D in human bodies. The noncanonical pathway of vitamin D activation is mediated by CYP11A1 and the generated products can be further modified by several enzymes such as CYP27A1, CYP27B1, CYP24A1, CYP2R1, and/or CYP3A4 [26], [27]. Both lumisterol and tachysterol and their precursors can be activated by CYP11A1 and CYP27A1 [28]. Unmodified vitamin D3 but not 25(OH)D3 is required for CYP11A1 to exert its catalytic activity [26]. Accordingly, the administration route of vitamin D3 is critical because oral intake of vitamin D3 will be mainly metabolized to 25(OH)D3 in the liver. To bypass the liver to initiate the noncanonical pathway of vitamin D3 activation, vitamin D3 must be transported to organs expressing CYP11A1 such as adrenal glands and other extra-adrenal tissues [3], [29]. Studies suggest that metabolites generated from the noncanonical pathway can mediate their effects through VDR and other alternative receptors such as retinoid-related orphan receptors α and γ, aryl hydrocarbon receptor, liver X receptor, and peroxisome proliferator–activated receptor γ [3], [21]. Importantly, these metabolites inhibit NF-κβ activity and the hedgehog and WNT/β-catenin pathways [30], and induce nuclear translocation of NRF2 and phosphorylated p53 from the cytoplasm to trigger signaling cascades [31].
By evaluating parameters such as bone mineral density (BMD), lower extremity function, dental health, and risk of falls, favorable serum concentrations of 25(OH)D are suggested to be greater than 75 nmol/l (30 ng/ml), which is relatively sufficient to meet health benefits in children and adults [32], [33]. Nevertheless, the optimal concentrations of 25(OH)D for these commonly used measurable outcomes are between 90 and 100 nmol/L (36–40 ng/ml) [33]. Accordingly, a collection of expert opinions defines a concentration of 25(OH)D lower than 20 ng/ml as vitamin D deficiency and a concentration of 25(OH)D ranging from 21-29 ng/ml as vitamin D insufficiency [32]. Both physiological and pathological conditions affect the levels of vitamin D or vitamin D metabolites, whereas pathological conditions like nephrotic syndrome tends to decrease serum levels of 25(OH)D, 1,25(OH)2D, and 24,25(OH)2D compared to those in healthy subjects, although certain arguments exist given different methods of measurements applied to determine concentrations of these vitamin D metabolites [34], [35]. Although ethnicity may be another factor affecting vitamin D status [36], a combination of other genetic and environmental factors such as the skin colour, sun exposure, conservative dress and diet significantly contribute to determine vitamin D status. Studies in the UK revealed that vitamin D deficiency and insufficiency are prevalent in the African-Caribbean population, and this situation is a public health concern [37]. By applying a new assay for determining actual vitamin D status, van Hoof et al. showed that the total level of 1,25(OH)2D, but not the serum level of free active 1,25(OH)2D, is greater in pregnant women than in healthy nonpregnant women [38]. On the other hand, patients with nephrotic syndrome present with lower levels of both total and serum free 1,25(OH)2D [38].
Epidemiological studies suggest an increased risk of autoimmune diseases in individuals with low vitamin D status, although the etiologies of autoimmune diseases remain unclear and are considered multifactorial. For example, in the immunological field, low 25(OH)D levels may be associated with the risk of several autoimmune diseases, such as rheumatoid arthritis (RA) [39], antiphospholipid syndrome [40], multiple sclerosis [41], and inflammatory bowel disease [42]. Moreover, vitamin D supplementation has the potential to decrease the incidence of autoimmune diseases such as RA and improve the prognosis [43].
In a nationwide, randomized, double-blind, placebo-controlled trial enrolling 25 871 participants with a follow-up period of 5.3 years, it was concluded that vitamin D3 supplementation (2000 IU/day) reduced the incidence of autoimmune diseases, including at least RA, polymyalgia rheumatica, autoimmune thyroid disease, psoriasis, and inflammatory bowel disease, by 22 %, although SLE was not specifically identified and analyzed [44]. In addition to vitamin D, synthetic VDRs have been suggested as potential therapeutics for treating various autoimmune diseases [45]. However, the results from some studies were not supportive of the benefit of vitamin D supplementation in treating autoimmune diseases (discussed below). Given these findings, the relationship between SLE and vitamin D remains incompletely understood. In the present report, aiming at several different aspects of patients with SLE, we discuss how vitamin D may have an impact on SLE-related issues.
Section snippets
Immunomodulatory effects and mechanisms of vitamin D in SLE
It is recognized that 1,25(OH)2D preserves immunomodulatory effects by regulating the functions of immune effector cells such as monocytes, macrophages, dendritic cells, natural killer cells, and lymphocytes and many other nonimmune cells [46], [47], [48] (Fig. 2). Noticeably, 1,25(OH)2D induces the differentiation of cells toward the monocyte–macrophage series but not the granulocyte series of cells [49]. However, when cocultured with 1,25(OH)2D, monocytes fail to mature and become effective…
Is low vitamin D status associated with a high risk of developing SLE?
Many factors are related to SLE susceptibility, such as hormonal, genetic, environmental, and immunological factors. As shown in Fig. 1, several factors in patients with SLE likely contribute to the development of low vitamin D status, such as the avoidance of sunlight exposure due to photosensitivity, the suppression of VDR by the disease itself [82] or through the use of corticosteroids [83] and disease modifying antirheumatic drugs (DMARDs) such as hydroxychloroquine [84], and the reduced…
The association between vitamin D status and SLE disease activity
The association between vitamin D status and disease activity in patients with autoimmune disorders is an interesting topic of study, suggesting the potential usefulness of evaluating vitamin D status as a disease activity parameter. In patients with multiple sclerosis who received interferon beta-1b treatment, higher 25(OH)D levels in the early phase of the disease were associated with reduced disease activity and a slower rate of disease progression [107]. Studies investigating the…
Potential therapeutic benefits of vitamin D supplementation in SLE patients
To investigate the impact of vitamin D treatment on SLE, a series of studies were carried out in lupus-prone mice. Early studies by Freitas et al. revealed a benefit of 1,25(OH)2D treatment with 2 μg/kg/day every 2 days for arthritis but not for proteinuria, glomerular mesangial expansion and inflammation or IgG and IgM deposition in the kidney of female BALB/c mice with pristane-induced lupus nephritis [124]. When lupus-mimic MRL/lpr mice maintained on low-calcium chow were intraperitoneally…
Confounding factors and other considerations
The studies summarized in this article provide up-to-date knowledge regarding the association between vitamin D status and SLE as well as a rationale for the potential benefits of maintaining vitamin D status in SLE patients. As discussed, there are inconsistent results from cellular, animal, and clinical observations about various aspects of vitamin D in SLE. Several adjustable confounders, including body mass index, renal function, drugs, especially hydroxychloroquine, prednisolone and…
Conclusion and perspectives
From a clinical viewpoint, it is highly recommended that all patients with SLE have their serum 25(OH)D concentrations checked, and for those with low vitamin D status, supplemental vitamin D3 should be introduced to maintain serum 25(OH)D concentrations at 30–40 ng/ml to better control disease activity and to reduce corticosteroid dosage and likely the dosage of DMARDs (Fig. 3). The benefit of vitamin D supplementation is amplified in SLE patients with very low serum 25(OH)D concentrations and …
VitaminDWiki - studies in both categories Lupus and Intervention
{category}
Attached to this page
- Vitamin D and Lupus: Are we doing enough? Aug 2021 📄 PDF
VitaminDWiki studies in both categories Lupus and Vitamin D Receptor
{category}
Vitamin D Receptor is associated in over 40 autoimmune studies
LUPUS pages
{category}
Lupus: need Vitamin D, 1 in 3 get another autoimmune, 4 types of Lupus - Mercola May 2025
📄 Download the PDF from VitaminDWiki
See also web
- Women have 22X higher risk of Lupus if have 3 X chromasomes (1 in 1000 of women have them) - Feb 2025
The global burden of SLE: prevalence, health disparities and socioeconomic impact - 2016 📄 PDF from SciHub via VitaminDWiki
- Latitude prevelences: little in Mexico, variable in US, high in Canada
Lupus Vitamin D Council major overview Feb 2014, 17 references
-
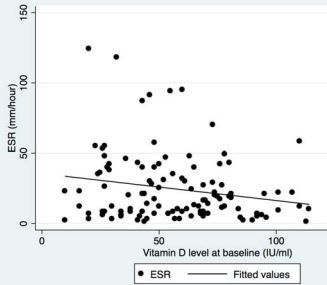
- April 2015, 3X more likely to get Lupus if < 40 nmol of Vitamin D, free PDF online
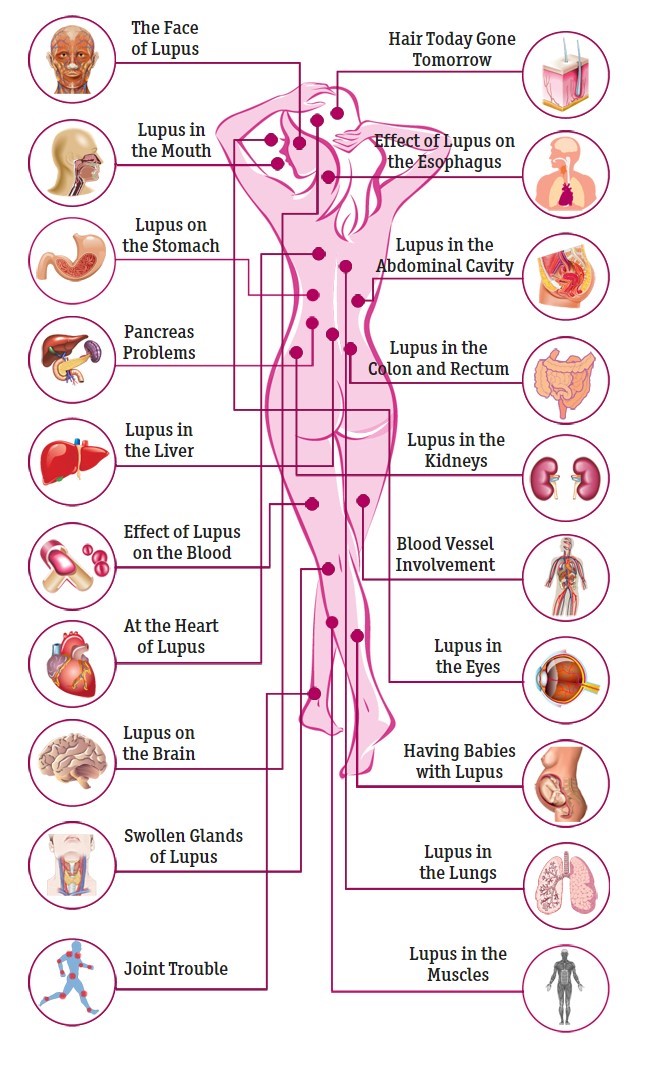
- Visualize the Effects of Lupus on the Body Healthline
- What Levels of Vitamin D should we Aim for in Patients with Systemic Lupus Erythematosus?
A Systematic Review of the Literature Poster June 2016 - A Stress Link to Lupus New York Times Sept 2017
- Those with PTSD were 3X more likely to get Lupus
- VitaminDWiki notes that Vit D and Omega-3 both treat PTSD (94 items as of Sept 2017) and Lupus
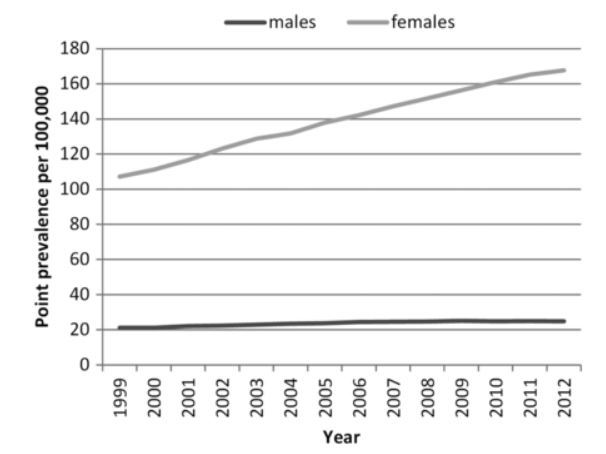
- The incidence and prevalence of systemic lupus erythematosus in the UK, 1999–2012 shows a 50% increase in Lupus
African-Americans (who typically have low levels of vitamin D) are 8X more likely to get Lupus (and many other heatlh problems)
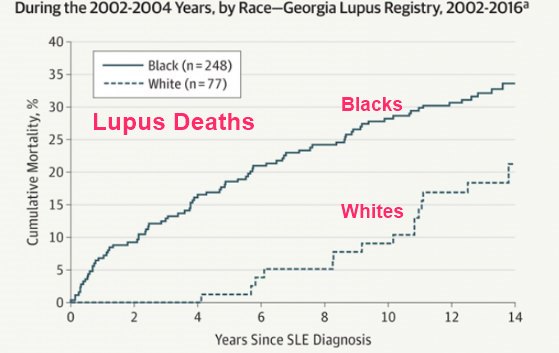
Disparities in Lupus Survival 2019
Note: Blacks have much lower vitamin D levels than whites
14 minute powerpoint presentation on SLE by Sarah McSpadden, April 2018