19X fewer COVID-19 infections if innate immune system activated – trial
Innate Immune Response Modulation and Resistance to SARS-CoV-2 infection: A Prospective Comparative Cohort Study in High Risk Healthcare Workers.
Preprint https://doi.org/10.1101/2020.10.20.20214965
📄 Download the PDF from VitaminDWiki
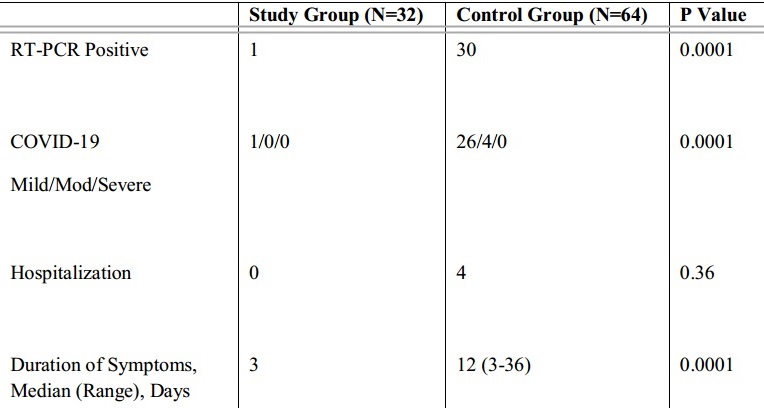
(subset of) Table 1: Characteristics and outcome of study and control subjects in Mw Prophylaxis study
Sarita Rani Jaiswal, MD 1,2, Anupama Mehta, BSc2, Gitali Bhagwati, MD 3, Rohit Lakhchaura, BSc2, Hemamalini Aiyer, MD3, Bakulesh Khamar, MS4, Supamo Chakrabarti 1,2 Cellular Therapy and Immunology, Manashi Chakrabarti Foundation, New Delhi1, Department of Blood and Marrow Transplantation2 and Department of Pathology and Microbiology, Dharamshila Narayana Super-speciality Hospital , New Delhi3, Research & Development, Cadila Pharmaceuticals Ltd, Ahmedabad4, India.
To evaluate ability of modulated innate immune response to provide resistance to development of symptomatic RT-PCR confirmed COVID-19, 96 inpatient front line health care workers (HCW) were cohorted in 1:2 ratio to receive TLR2 agonist (heat killed Mycobacterium w, Mw; n=32) as innate immune response modulator or observation (n=64). All were followed up for 100 days. The incidence of COVID-19 was 31 (32.3%) for the entire cohort, with only one developing COVID-19 in Mw group (3.1% vs 46.8%. protective efficacy - 93.33%, p=0.0⑻1; 95% CI 53.3-99.1). Self-limiting local injection site reaction was the only side effect and was seen in 14 HCW. Findings from the study suggest the potential for providing resistance against novel pathogen like SARS-CoV-2 by modulating innate immune response.
Introduction
Front-line healthcare workers (HCW) are at the highest risk of acquiring COVID-19 with a 30-day hazard ratio (HR) of 24.3, compared to the general community1. Innate immune response is a conserved, prompt mechanism, which resists infections by recognizing the conserved pathogen-associated molecular patterns (PAMP) of an infectious agent by pathogen recognizing receptors (PRR) like toll like receptors (TLR). It is relevant in infections by novel pathogens like SARS-COV-22. A TLR2 agonist, heat killed Mycobacterium w (Mw), also known as Mycobacterium Indicus Pranii, is, an approved immunomodulator in India3 and is used in the management of leprosy and other conditions. In a prospective open label cohort control study, we explored the efficacy of Mw in inducing resistance to the development of COVID-19 infection in HCW at a high risk of exposure, in a tertiary-healthcare set-up.
Methods
Thirty-two HCWs from the Department of Blood and Marrow Transplantation and Hematology were administered 0.1 ml Mw (Sepsivac, Cadila Pharmaceuticals, India) intradermally in each arm on day 1 of the study (Mw group) and followed up for 100 days. 64 age matched HCWs from the rest of the hospital were enrolled in a control group. All HCW included in the study had a nasopharyngeal swab evaluated for SARS-CoV-2 by reverse transcriptase-polymerase chain reaction (RT-PCR), on development of symptoms suggestive of COVID-19 or following exposure to an infected person. Body temperature, pulse rate, oxygen saturation and self-reporting of symptoms was evaluated before and after each working day in the Mw group. ‘Exposure5 was defined as close contact with SARS-CoV-2 infected individuals without full protective gear. COVID-19 was graded as mild, moderate or severe as per WHO criteria. Subjects in the Mw group underwent two additional random SARS-CoV-2 specific RT-PCR evaluation 4 weeks apart. The study was approved by the institutional ethics committee.
Results
The characteristics and outcomes of the Mw and control groups are detailed in Table 1. Overall, 31 out of 96 HCW enrolled had RT-PCR-confirmed COVID-19 infection of which 30 (96.77%) were in control group. Of the 31, who developed COVID-19 infection, four required hospitalization. All belonged to the control group. Despite a greater number of exposures in the Mw group, only one out of 32 (3.13%) subjects had an RT-PCR confirmed mild COVID-19 infection. HR for developing COVID-19 in the control group compared to the Mw group was 19.025 (p=0.0038). Based on this study, the resistance to infection (protective efficacy) provided by Mw was 93.33% (p=0.0001; 95% CI 53.3-99.1).
The only side effect noted with Mw was injection site reactions (moderate to severe - 4 ; mild - 10 ), which were self-limiting and did not require any specific management.
Discussion
The study period coincided with the peak of the pandemic in New Delhi and the high rate of COVID-19 seen in control group is in line with that reported in similar HCW1. The resistance to COVID-19 seen in the Mw group suggests that a TLR2 agonist Mw might be useful in providing protection to subjects at a high risk of exposure. It will be interesting to study its long-term protective efficacy. BCG, another approved immunomodulator is also being evaluated for the prevention of COVID-19, and it will be interesting to study its outcome as there are differences in the innate immune response generated by the two in terms of being a TLR agonist and in ligand presentation4. Specific immune changes like upregulation of adaptive natural killer cells are being investigated by our group5 to understand the immune mechanism responsible for resistance to COVID-19. This study provides an initial proof to the concept of modulating the innate immune response for providing resistance to novel pathogens like SARS-COV2 until the availability of vaccines.
References:
Nguyen LH, Drew DA, Graham MS, et al. Risk of COVID-19 among front-line health-care workers and the general community: a prospective cohort study. Lancet Public Health. 2020;5(9):e475-e483.
Mantovani A, Netea MG. Trained Innate Immunity, Epigenetics, and Covid-19. N Engl JMed. 2020;383(11):1078-1080.
Pandey RK, Sodhi A, Biswas SK, Dahiya Y, Dhillon MK. Mycobacterium indicus pranii mediates macrophage activation through TLR2 and NOD2 in a MyD88 dependent manner. Vaccine. 2012;30(39):5748-5754.
Kumar P, Tyagi R, Das G, Bhaskar S. Mycobacterium indicus pranii and Mycobacterium bovis BCG lead to differential macrophage activation in Toll-like receptor-dependent manner. Immunology. 2014;143(2):258-268.
Jaiswal SR, Malhotra P, Mitra DK, Chakrabarti S. Focusing On A Unique Innate Memory Cell Population Of Natural Killer Cells In The Fight Against COVID-19: Harnessing The Ubiquity Of Cytomegalovirus Exposure. Mediterr J Hematol Infect Dis. 2020;12(1):e2020047.
Title change made Nov 2020 caused the visitor count to reset.
There have actually been visitors to this page since it was originally made
