Dementia less likely with increased levels of Magnesium etc.
See VitaminDWiki
Overview Magnesium and vitamin D notice that Alzheimer's is decreased by BOTH Magnesium and vitamin D
A balanced diet is no longer enough – supplements needed Vitamin D, Magnesium, etc.
Vitamin D pages containing "Dementia" in title (35 as of June 2021)
Dementia risk increases 60 percent if have low vitamin D – Nov 2012
Reversal of cognitive decline with multitherapy (not monotherapy) – Sept 2014
Items in both categories Cognition and Magnesium:
{category}
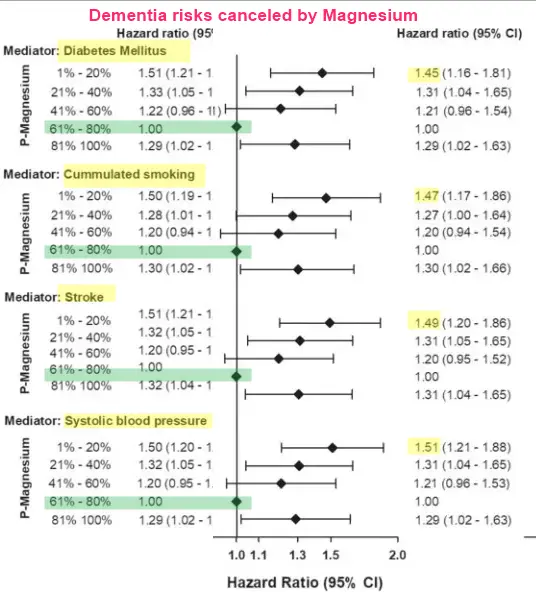
4 Dementia risk factors cancelled if have enough Magnesium - June 2021
Plasma Concentrations of Magnesium and Risk of Dementia: A General Population Study of 102 648 Individuals
Clinical Chemistry, Volume 67, Issue 6, June 2021, Pages 899–911, https://doi.org/10.1093/clinchem/hvab041
Jesper Qvist Thomassen, Janne S Tolstrup, Børge G Nordestgaard, Anne Tybjærg-Hansen, Ruth Frikke-Schmidt
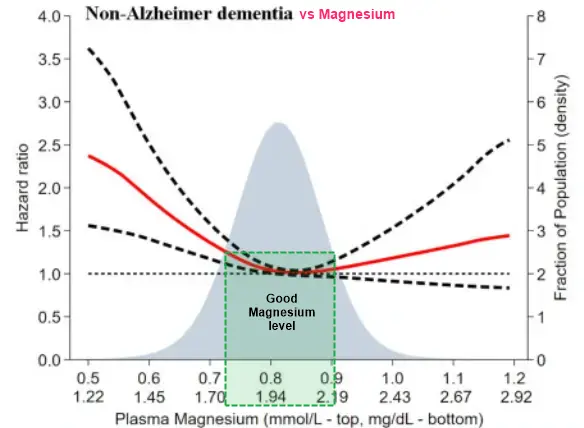
Background
Low and high concentrations of plasma magnesium are associated with increased risk of future all-cause dementia; however, the underlying reasons remain elusive. The magnesium ion is an important electrolyte serving as a cofactor in many enzymatic processes in the human organism. Magnesium affects both neuronal and vascular functions. We investigated the associations of plasma concentrations of magnesium associate with common subtypes of dementia as Alzheimer dementia and non-Alzheimer dementia, and potential pathways by which magnesium may affect risk of dementia.
Methods
Plasma concentrations of magnesium were measured in 102 648 individuals from the Copenhagen General Population Study. Cox regression and natural effects mediation analyses evaluated associations with either Alzheimer dementia or non-Alzheimer dementia.
Results
Multifactorially adjusted hazard ratios for non-Alzheimer dementia were 1.50 (95% confidence interval (CI):1.21–1.87) for the lowest and 1.34(1.07–1.69) for the highest vs the fourth quintile (reference) of plasma magnesium concentrations. Diabetes, cumulated smoking, stroke, and systolic blood pressure mediated 10.4%(3.1–22.8%), 6.8%(1.2–14.0%), 1.3%(0.1–3.6%), and 1.0%(0.2–2.6%), respectively, in the lowest quintile, whereas stroke mediated 3.2%(0.4–11.9%) in the highest quintile. No associations were observed for Alzheimer dementia.
Conclusions
Low and high plasma magnesium concentrations were associated with high risk of vascular-related non-Alzheimer dementia, with the lowest risk observed at a concentration of 2.07 mg/dL (0.85 mmol/L). No association was observed for Alzheimer dementia. Mediation analysis suggested that diabetes may be in the causal pathway between low plasma magnesium concentrations and high risk of non-Alzheimer dementia, while cumulated smoking, stroke, and systolic blood pressure played minor mediating roles.
📄 Download the PDF from VitaminDWiki
Dementia risk cut in half in those taking Magnesium (as a laxative) – Jan 2018
Magnesium oxide use and reduced risk of dementia: a retrospective, nationwide cohort study in Taiwan.
Curr Med Res Opin. 2018 Jan;34(1):163-169. doi: 10.1080/03007995.2017.1385449. Epub 2017 Oct 30.
Tzeng NS1,2, Chung CH3,4, Lin FH4, Huang CF5,6,7, Yeh CB1,8, Huang SY1,8, Lu RB1,8,9,10,11,12,13, Chang HA1,2, Kao YC1,14, Yeh HW1,15,16, Chiang WS1,17, Chou YC4, Tsao CH18,19, Wu YF18, Chien WC4,18.
OBJECTIVE:
Dietary magnesium may be associated with a lower risk of dementia; however, the impact of magnesium oxide (MgO), a common laxative, on dementia has yet to be elucidated. This study aimed to investigate the association between the usage of MgO and the risk of developing dementia.
METHODS:
We used a dataset from the National Health Research Institute Database (NHRID) of Taiwan containing one million randomly sampled subjects to identify patients aged ≥50 years with no history of MgO usage. A total of 1547 patients who had used MgO were enrolled, along with 4641 controls who had not used the MgO propensity score matched by age, gender and comorbidity, at a ratio of 1:3. After adjusting for confounding risk factors, a Cox proportional hazards model was used to compare the risk of developing dementia during a 10 year follow-up period.
RESULTS:
Of the enrolled patients, 44 (2.84%) developed dementia, when compared to 199 (4.28%) in the control group. The Cox proportional hazards regression analysis revealed that the patients who had used MgO were less likely to develop dementia with a crude hazard ratio of 0.617 (95% CI, 0.445-0.856, p = .004). After adjusting for age, gender, comorbidity, geographical area and urbanization level of residence, and monthly income, the adjusted hazard ratio was 0.517 (95% CI, 0.412-0.793, p = .001).
CONCLUSIONS:
The patients who used MgO had a decreased risk of developing dementia. Further studies on the effects of MgO in reducing the risk of dementia are therefore warranted.
