Restless legs syndrome 5X more likely if low vitamin D
Possible association between vitamin D deficiency and restless legs syndrome
Neuropsychiatric Disease and Treatment 21 May 2014
Mustafa Oran1 cuneyt Unsal2 Yakup Albayrak2 Feti Tulubas3 Keriman Oguz4 Okan Avci1 Nilda Turgut4 Recep Alp4 Ahmet Gurel3
'Department of internal Medicine, 2Department of Psychiatry, 3Department of Biochemistry, 4Department of Neurology, Namik Kemal University, Faculty of Medicine, Tekirdag, Turkey
Correspondence: Mustafa Oran Department of Internal Medicine, School of Medicine, Namik Kemal University, Tunca Caddesi, Tekirdag, 59100, Turkey Tel +90 542 388 4317 Fax +90 282 250 0001 Email oranmmd@yahoo.com
Background and aim: Restless legs syndrome (RLS) is a distressing sleep disorder that occurs worldwide. Although there have been recent developments in understanding the pathophysiology of RLS, the exact mechanism of the disease has not been well elucidated. An increased prevalence of neurologic and psychiatric diseases involving dopaminergic dysfunction in vitamin D-deficient patients led us to hypothesize that vitamin D deficiency might result in dopaminergic dysfunction and consequently, the development of RLS (in which dopaminergic dysfunction plays a pivotal role). Thus, the aim of this study was to evaluate the relationship between vitamin D deficiency and RLS.
Methods: One hundred and fifty-five consecutive patients, 18-65 years of age, who were admitted to the Department of Internal Medicine with musculoskeletal symptoms and who subsequently underwent neurological and electromyography (EMG) examination by the same senior neurologist, were included in this study. The patients were divided into two groups according to serum 25-hydroxyvitamin D (25(OH)D) (a vitamin D metabolite used as a measure of vitamin D status) level: 36 patients with serum 25(OH)D levels >20 ng/mL comprised the normal vitamin D group, and 119 patients with serum 25(OH)D levels <20 ng/mL comprised the vitamin D deficiency group. The two groups were compared for the presence of RLS and associated factors.
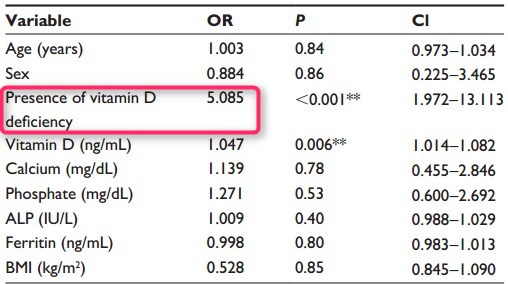
Results: The two groups were similar in terms of mean age, sex, mean body mass index (BMI), and serum levels of calcium, phosphate, alkaline phosphatase (ALP), and ferritin. The presence of RLS was significantly higher in the vitamin D deficiency group (up arrow) 2=12.87, P<0.001). Regression analysis showed vitamin D deficiency and serum 25(OH)D level to be significantly associated with the presence of RLS (odds ratio [OR] 5.085, P<0.001 and OR 1.047, P=0.006, respectively).
Conclusion: The present study demonstrated a possible association between vitamin D deficiency and RLS. Given the dopaminergic effects of vitamin D, 25(OH)D depletion may lead to dopaminergic dysfunction and may have a place in the etiology of RLS. Prospective vitamin D treatment studies are needed to confirm this relationship and to evaluate the efficacy of vitamin D as a treatment for RLS patients.
Introduction
Restless legs syndrome (RLS) is a distressing sleep disorder that affects many adults.1 The major characteristic of this disease is a compelling urge to move the legs, with an unpleasant feeling in the lower extremities. It typically starts or becomes worse during rest periods and deteriorates during the evening and at night; the symptoms partially or totally disappear with movement.2 RLS is an important clinical and public health problem in the Western industrialized world, affecting 4%-29% of adults.3
RLS is divided into primary and secondary forms. Primary (or idiopathic) RLS lacks physiological context or comorbidities that can be related to the patient's symptoms. Alternately, secondary RLS patients have comorbidities or other recognizable causes, such as chronic renal failure, medications, or iron deficiency.4 While the pathophysiology of RLS has not yet been fully elucidated, brain iron deficiency and dopaminergic dysfunction have long been regarded as the key culprits.5 The successful results of dopamine agonist use in RLS treatment supports the dop-aminergic dysfunction hypothesis in RLS.6,7 Furthermore, brain iron deficiency has been shown to be associated with dopaminergic dysfunction,8 and it has been suggested that dopaminergic dysfunction is a consequence of iron deficiency in RLS.9
Vitamin D is synthesized photochemically in human epithelial cells and acquired from dietary sources. The classical effects of vitamin D include the regulation of calcium and phosphorus homeostasis and bone metabolism. Recently, nonclassical effects of vitamin D have gained renewed attention. One of the important functions of this vitamin is the regulation of nervous system development and function. It has been suggested that vitamin D deficiency might lead to an increased risk of central nervous system disease, such as schizophrenia and multiple sclerosis.10 It has been shown that low doses of 1 alpha, 25 dihydroxyvitamin D3 (1,25-(OH)2D3) (the hormonally active form of vitamin D) are able to protect the mesencephalic dopaminergic neurons against toxins that cause a decrease in glutathione con-tent.11 Decreased glutathione content has been shown to cause selective dopaminergic neuron death.12
Although a large number of studies have investigated the role of vitamin D in neurological disorders, data regarding the relationship between vitamin D and RLS are scarce. In the present study, we aimed to investigate the relationship between vitamin D and RLS by evaluating RLS and symptom severity in patients with and without normal 25-hydroxy-vitamin D (25(OH)D) (a vitamin D metabolite used as a measure of vitamin D status) levels.
Patients and methods
Patients
The study was conducted from September 2013 to January 2014 at the Internal Medicine and Neurology Clinics of the Namik Kemal University Medical Faculty. Two hundred and forty-three consecutive patients 18-65 years of age who were admitted to the Department of Internal Medicine with musculoskeletal symptoms were included. Patients were excluded if they had abnormal complete blood biochemistry results; abnormal thyroid hormone, sedimentation, vitamin B12, or ferritin levels; a familial history of RLS or known causes of secondary RLS; osteoporosis or systemic inflammatory and connective tissue disease; consumed alcohol excessively (>40 g/day) or smoked; diabetes, renal failure, or chronic hepatic failure; or medical treatment with corticosteroids, calcitonin, estrogen, calcium, bisphosphonates, or vitamin D. According to the exclusion criteria, 68 patients were excluded from the study, and 175 patients were included and referred to the neurology clinics at the same center. All of the subjects underwent neurological examination. Nerve conduction studies were carried out by the same senior neurologist (who was blinded to the vitamin D status of the participants), using a Nihon Kohden Neuropack M1 electromyography (EMG) device (Nihon Kohden Corp, Tokyo, Japan), to evaluate for the presence of RLS and to exclude patients with any symptom or sign of neurological disorders or neuropathy. Twenty more patients with EMG findings of peripheral neuropathy were excluded following the neurological and EMG examination. Thus, 155 patients were finally included in the study. Demographic and clinical characteristics, including age, sex, and body mass index (BMI), were noted. The patients were divided into two groups according to serum 25(OH)D level: 36 patients (six male and 30 female) with serum 25(OH)D levels >20 ng/mL comprised the normal vitamin D group (VDN), and 119 patients (nine male and 110 female) with serum 25(OH)D levels <20 ng/mL comprised the vitamin D deficiency group (VDD). The study was conducted in accordance with the principles of the Helsinki Declaration and was approved by the local ethics committee; written informed consent was obtained from each participant.
Biochemical measurements
Venous blood samples (20 mL) were collected in plain glass tubes from all of the participants for the 25(OH)D assessment. Serum 25(OH)D levels were estimated using an electro chemiluminescence method (Cobas® e 411 immu-nochemistry analyzer; F. Hoffmann-La Roche Ltd, Basel, Switzerland) with interassay and intra-assay coefficients of variation of 2.6% and 5.8%, respectively. Vitamin D concentrations <20 ng/mL were defined as insufficiency, and concentrations >20 ng/mL were considered adequate.13 Calcium, phosphate, alkaline phosphatase (ALP), and ferritin levels were estimated with commercially available kits (Cobas C 501; F. Hoffmann-La Roche Ltd).
Evaluation of RLS
The International Restless Legs Syndrome (IRLS) Study Group essential criteria scores were used by the same neurologist to evaluate RLS in the patient and control groups.14
Statistics
The data were analyzed using the Statistical Package for the Social Sciences, Version 16.0 (SPSS, Inc., Chicago, IL, USA). A confidence interval (CI) of 95% and a two-tailed P-value <0.05 were considered to be statistically significant for all analyses. Variables were tested for homogeneity of variance with Levene's test and for normality of distribution with the Kolmogorov-Smirnov test. Statistical comparisons were carried out with chi-square (x2) and independent samples Student's 2-tests. The strengths of association between presence of RLS (dependent variable) and vitamin D deficiency, age, BMI, sex, and serum levels of vitamin D, calcium, phosphate, ferritin, and ALP were examined with a series of logistic regressions. The results are presented as odds ratios (ORs) for independent variables of interest, with associated 95% CIs.
Results
The mean age of the patients was 49.32+11.63 years old. Fifteen patients were male (9.7%), and 140 were female (90.3%). Vitamin D serum levels were in the normal range in 36 patients (23.2%) and lower than normal in 119 patients (76.8%). Sixty-six patients were diagnosed with RLS (42.5%).
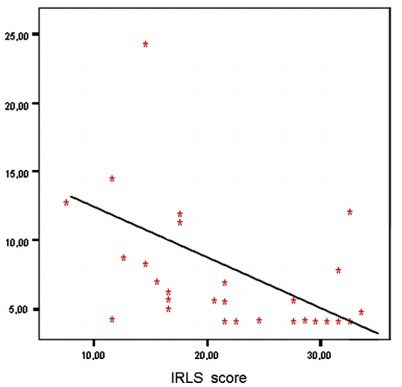
The mean age of the VDD group was 48.80+11.44 years, and the mean age of the VDN group was 51.03+12.31 years. The mean ages and sex ratios of the two groups were similar (P>0.05). Sixty patients (50.4%) in the VDD group and six patients (6.7%) in the VDN group were diagnosed with RLS. The percentage of presence of RLS was significantly higher in the VDD group (X=12.87, P<0.001). The mean vitamin D serum levels were 11.19+4.73 ng/mL in the VDD group and 37.19+9.97 ng/mL in the VDN group (2=21.59, P<0.001). The mean serum phosphate levels were 3.27+0.44 mg/dL in the VDD group and 3.61 +0.52 mg/dL in the VDN group (2=3.79, P<0.001). The calcium, ferritin, and ALP serum levels and the BMI scores of the two groups were found to be similar (P>0.05). The percentages of upper extremity involvement were 8.2% in the VDD group and 33% in the VDN group. The mean IRLS scores were 23.54+4.25 in the VDD group and 21.50+3.72 in the VDN group (Table 1).
Age, BMI, and serum levels of vitamin D, calcium, phosphate, ferritin, and ALP, as continuous predictors, and sex and vitamin D deficiency, as categorical predictors, were included in a logistic regression. The logistic regression equation was able to classify 62.3% of the cases correctly. Presence of vitamin D deficiency was significantly associated with presence of RLS (OR =5.085, 95% CI: 1.972-13.113), as was serum vitamin D level (OR =1.047, 95% CI: 1.014-1.082). The associations between the other
Table 1. Comparisons of demographical and clinical factors among the VDD and VDN groups
| age (years) | 48.80+11.44 | 51.03+12.31 | t=i.0i, P=0.31 |
| sex | |||
| Male | 9 (7.6%) | 6 (16.7%) | x2=2.62, P=0.I0 |
| Female | 110 (92.4%) | 30 (83.3%) | |
| RLs | |||
| Present | 60 (50.4%) | 6 (16.7%) | x2= 12.87, P<0.00I* |
| absent | 59 (49.6%) | 30 (83.3%) |
Notes: Values are expressed as mean + SD. A P-value <0.05 was considered statistically significant. Significant P-value.
Abbreviations: ALP, alkaline phosphatase; BMI, body mass index; IRLS, International Restless Legs Syndrome rating scale; RLS, restless legs syndrome; SD, standard deviation; VDD, vitamin D deficiency; VDN, normal vitamin D.
Table 2 The logistic regression analysis of variables associated with RLS
Discussion
Although RLS has long been a clinical problem, recent developments have contributed to understanding the pathophysiology and treatment of the disease. The exact pathophysiology of RLS has not yet been well elucidated; however, accumulating data have shown that the pathophysiology of RLS is centered on dopaminergic dysfunction and brain iron deficiency.15,16 One of the most persuasive arguments for dopaminergic dysfunction is the improvement of RLS symptoms with dopaminergic drugs.17 Imaging studies further support the role of dysfunction of the dopaminergic pathways.18 Single-photon emission computed tomography imaging of striatal pre-and postsynaptic dopaminergic status has shown a mild reduction in postsynaptic dopaminergic status.19 Similarly, fluorine-18-L-dihydroxyphenylalanine (18F-dopa) positron emission tomography studies have shown a decrease in RLS patients' striatal 18F-dopa uptake compared with that of healthy controls, supporting presynaptic dopaminergic dysfunction in the striatum.20,21 Moreover, in an autopsy study, a significant decrease in dopamine 2 (D2) receptors was observed in the putamina of eight patients with primary RLS compared with those of a neurologically normal control group; reduction in D2 receptors was correlated with the severity of RLS.22 Increased incidence of RLS in neurological diseases that involve dopaminergic systems, such as Huntington's disease, Tourette syndrome, and essential tremor, may also support dopaminergic dysfunction in RLS patients.23-25
Iron deficiency has been shown to be an important risk factor and to play a pivotal role in the pathophysiology of RLS.26 It has been demonstrated that RLS severity is negatively correlated with serum ferritin levels,27 even within normal laboratory ranges, and that symptoms improve with iron treatment.28 Recently, Rizzo et al used magnetic resonance imaging phase analysis to study brain iron content in idiopathic RLS in vivo. The researchers found that significantly higher phase values were present in RLS patients compared with healthy controls, at the level of the substantia nigra, thalamus, putamen, and pallidum, implying reduced iron content in several regions of the patients' brains.29 A clear link between iron deficiency and nigrostriatal dop-amine dysfunction has also been reported, which appears to have an important role in RLS.30 Nevertheless, the underlying mechanisms remain unclear. In a recent animal study conducted by Jellen et al iron deficiency was shown to alter the expression of dopamine-related genes in the ventral midbrain in mice.31
The important role of vitamin D in skeletal health has been well described; however, many recent studies have also linked vitamin D deficiency to various nonskeletal conditions, including cardiovascular disease, cancer, metabolic disorders, and neuropsychiatric diseases. Accumulating data have provided evidence that 1,25-(OH)2D3 is involved in brain function, and the nuclear receptor for 1,25-(OH)2D3 has been localized in neurons and glial cells.32 1,25-(OH)2D3 has been reported to increase glutathione levels, suggesting that the hormone has a role in brain detoxification path-ways.33 Nakamura et al showed that decreased glutathione content may cause the selective death of dopaminergic neurons.12 Furthermore, results of a study by Shinpo et al found that low doses of 1,25-(OH)2D3 were able to protect mesen-cephalic dopaminergic neurons against toxicity induced by L-buthionine sulfoximine and 1-methyl-4-phenylpyridium ions, which was reported to cause a depletion of glutathione content.11 Similarly, Orme et al observed a dose-responsive increase in numbers of rat primary dopaminergic neurons when 1,25-(OH)2D3 was added to culture media.34 They suggested that 1,25-(OH)2D3 increases dopaminergic neurons by upregulating the expression of glial-derived neurotrophic factor.
While various studies have investigated the relationship of vitamin D depletion and neuropsychiatric disorders, such as schizophrenia, dementia, Parkinson's disease, and multiple sclerosis, only one study has been conducted to evaluate the relationship of vitamin D and RLS. Balaban et al evaluated 25(OH)D levels in patients with and without RLS and found statistically significantly lower serum 25(OH) D levels in female RLS patients compared with control subjects matched for sex.35 They concluded that decreased 25(OH)D levels, which may affect dopamine function, might be an etiological factor for RLS. However, this conclusion should be questioned as both the patient and control groups had low average levels of 25(OH)D. In our study, subjects with and without normal levels of 25(OH)D were compared for the presence of RLS, and an association was found between 25(OH)D serum level and presence of RLS. In addition, the VDD group had an increased prevalence of RLS, and the presence of vitamin D deficiency was found to be strongly associated with RLS. Given the dopaminer-gic effects of vitamin D, decreased 25(OH)D may lead to dopaminergic dysfunction, and consequently, to increased RLS prevalence.
Some limitations of the present study should be mentioned. Evaluation of disorders that have been shown to be frequent in RLS patients, such as periodic limb movements during sleep, would be useful to support RLS diagnosis; however, we did not evaluate these because a variety of disorders have been argued to be frequent in RLS patients. Also, it would have been better to calculate a sample size estimation for each group; instead, we included all patients who had adequate inclusion and exclusion criteria. This might be considered as another limitation.
In conclusion, our findings may support an association between vitamin D deficiency and RLS. Vitamin D deficiency should be considered in RLS patients, particularly those who have been diagnosed with idiopathic RLS. However, prospective vitamin D treatment studies are needed to support this relationship and to evaluate the efficacy of vitamin D as a treatment in RLS patients.
References
Innes KE, Selfe TK, Agarwal P. Restless legs syndrome and conditions associated with metabolic dysregulation, sympathoadrenal dysfunction, and cardiovascular disease risk: a systematic review. Sleep Med Rev. 2012;16(4):309-339.
Yeh P, Walters AS, Tsuang JW. Restless legs syndrome: a comprehensive overview on its epidemiology, risk factors, and treatment. Sleep Breath. 2012;16(4):987-1007.
Innes KE, Selfe TK, Agarwal P. Prevalence of restless legs syndrome in North American and Western European populations: a systematic review. Sleep Med. 2011;12(7):623-634.
Kushida CA. Clinical presentation, diagnosis, and quality of life issues in restless legs syndrome. Am J Med. 2007;120(1 Suppl 1):S4-S12.
Nagandla K, De S. Restless legs syndrome: pathophysiology and modern management. Postgrad Med J. 2013;89(1053):402-410.
Zintzaras E, Kitsios GD, Papathanasiou AA, et al. Randomized trials of dopamine agonists in restless legs syndrome: a systematic review, quality assessment, and meta-analysis. Clin Ther. 2010;32(2):221-237.
Winkelman JW. Considering the causes of RLS. Eur J Neurol. 2006; 13 Suppl 3:S8-S14.
Snyder AM, Connor JR. Iron, the substantia nigra and related neurological disorders. Biochim Biophys Acta. 2009;1790(7):606-614.
Allen RP, Barker PB, Wehrl F, Song HK, Earley CJ. MRI measurement of brain iron in patients with restless legs syndrome. Neurology. 2001;56(2):263-265.
Wrzosek M, Lukaszkiewicz J, Wrzosek M, et al. Vitamin D and the central nervous system. Pharmacol Rep. 2013;65(2):271-278.
Shinpo K, Kikuchi S, Sasaki H, Moriwaka F, Tashiro K. Effect of 1,25-dihydroxyvitamin D(3) on cultured mesencephalic dopamin-ergic neurons to the combined toxicity caused by L-buthionine sul-foximine and 1-methyl-4-phenylpyridine. J Neurosci Res. 2000;62(3): 374-382.
Nakamura K, Wang W, Kang UJ. The role of glutathione in dopamin-ergic neuronal survival. J Neurochem. 1997;69(5):1850-1858.
Gomez Alonso C, Naves Diaz M, Rodriguez Garcia M, Fernandez Martin JL, Cannata Andia JB. [Review of the concept of vitamin D "sufficiency and insufficiency"]. Nefrologia. 2003 ;23 Suppl 2:S73-S77. Spanish.
Allen RP, Picchietti D, Hening WA, Trenkwalder C, Walters AS, Montplaisi J; Restless Legs Syndrome Diagnosis and Epidemiology workshop at the National Institutes of Health; International Restless Legs Syndrome Study Group. Restless legs syndrome: diagnostic criteria, special considerations, and epidemiology. A report from the restless legs syndrome diagnosis and epidemiology workshop at the National Institutes of Health. Sleep Med. 2003;4(2):101-119.
Sethi KD, Mehta SH. A clinical primer on restless legs syndrome: what we know, and what we don't know. Am J Manag Care. 2012; 18(5 Suppl):S83-S88.
Connor JR, Boyer PJ, Menzies SL, et al. Neuropathological examination suggests impaired brain iron acquisition in restless legs syndrome. Neurology. 2003;61(3):304-309.
Rios Romenets S, Postuma RB. Treatment of restless legs syndrome. Curr Treat Options Neurol. 2013;15(4):396-W9.
Rizzo G, Tonon C, Manners D, Testa C, Lodi R. Imaging brain functional and metabolic changes in restless legs syndrome. Curr Neurol Neurosci Rep. 2013;13(9):372.
Michaud M, Soucy JP, Chabli A, Lavigne G, Montplaisir J. SPECT imaging of striatal pre- and postsynaptic dopaminergic status in restless legs syndrome with periodic leg movements in sleep. J Neurol. 2002;249(2):164-170.
Turjanski N, Lees AJ, Brooks DJ. Striatal dopaminergic function in restless legs syndrome: 18F-dopa and 11C-raclopride PET studies. Neurology. 1999;52(5):932-937.
Ruottinen HM, Partinen M, Hublin C, et al. An FDOPA PET study in patients with periodic limb movement disorder and restless legs syndrome. Neurology. 2000;54(2):502-504.
Connor JR, Wang XS, Allen RP, et al. Altered dopaminergic profile in the putamen and substantia nigra in restless leg syndrome. Brain. 2009;132(Pt 9):2403-2412.
Evers S, Stogbauer F. Genetic association of Huntington's disease and restless legs syndrome? A family report. Mov Disord. 2003;18(2):225-227.
Ondo WG, Lai D. Association between restless legs syndrome and essential tremor. Mov Disord. 2006;21(4):515-518.
Lesperance P, Djerroud N, Diaz Anzaldua A, Rouleau GA, Chouinard S, Richer F; Montreal Tourette Study Group. Restless legs in Tourette syndrome. Mov Disord. 2004;19(9):1084-1087.
Allen RP, Earley CJ. The role of iron in restless legs syndrome. Mov Disord. 2007;22 Suppl 18:S440-S448.
Sun ER, Chen CA, Ho G, Earley CJ, Allen RP. Iron and the restless legs syndrome. Sleep. 1998;21(4):371-377.
O'Keeffe ST, Noel J, Lavan JN. Restless legs syndrome in the elderly. Postgrad Med J. 1993;69(815):701-703.
Rizzo G, Manners D, Testa C, et al. Low brain iron content in idiopathic restless legs syndrome patients detected by phase imaging. Mov Disord. 2013;28(13):1886-1890.
Lozoff B. Early iron deficiency has brain and behavior effects consistent with dopaminergic dysfunction. JNutr. 2011;141(4):740-746.
Jellen LC, Lu L, Wang X, et al. Iron deficiency alters expression of dopamine-related genes in the ventral midbrain in mice. Neuroscience. 2013;252:13-23.
Garcion E, Wion-Barbot N, Montero-Menei CN, Berger F, Wion D. New clues about vitamin D functions in the nervous system. Trends EndocrinolMetab. 2002;13(3):100-105.
Garcion E, Sindji L, Leblondel G, Brachet P, Darcy F. 1,25-dihydroxyvitamin D3 regulates the synthesis of gamma-glutamyl trans-peptidase and glutathione levels in rat primary astrocytes. J Neurochem. 1999;73(2):859-866.
Orme RP, Bhangal MS, Fricker RA. Calcitriol imparts neuroprotec-tion in vitro to midbrain dopaminergic neurons by upregulating GDNF expression. PLoS One. 2013;8(4):e62040.
Balaban H, Yildiz OK, Cil G, et al. Serum 25-hydroxyvitamin D levels in restless legs syndrome patients. Sleep Med. 2012;13(7):953-957.
See also VitaminDWiki
Restless Legs Syndrome - Vitamin D, Iron, etc. - many studies
Restless legs and growing pains may be both due to low vitamin D – Sept 2013
Search VitaminDWiki for "RESTLESS LEG" 1850 items as of June 2014
Those with restless leg syndrome had 40 percent less vitamin D – June 2012 which has the following chart