HIV interactions with low Vitamin D
Vitamin D deficiency in HIV infection: an underestimated and undertreated epidemic
European Review for Medical and Pharmacological Sciences
M.R. P\NZONE, M. D\ ROSA, M. MAIAGUARNERA, G. MADEDDU, E. FOCA, G. CECCARELL\, G. D'ETTORRE, V. VULLO, R. F\S\CHELLA, B. CACOPARDO, G. NUNNAR\1,7
Corresponding Author: Giuseppe Nunnari, MD; e-mail: gnunnari@hotmail.com
Hypovitaminosis D is a very common disorder, regarding both Western and developing countries. A growing amount of data over the last years have shown vitamin D deficiency to be high prevalent among HIV-positive subjects. In addition to "classic" risk factors, such as female sex, low dietary intake, dark skin pigmentation and low sun exposure, HlV-related factors, including immune activation and anti-retroviral adverse effects, may affect vitamin D status. Even if both protease inhibitors and non-nucleoside reverse transcriptase inhibitors have been associated with low vitamin D levels, available evidences have failed to univocally associate hypovitaminosis D with specific antiretroviral class effects.
Low vitamin D is known to have a negative impact not only on bone health, but also on neurocognitive, metabolic, cardiovascular and immune functions. Similarly to the general population, several studies conducted on HIV-infected subjects have associated hypovitaminosis D with a greater risk of developing osteopenia/os-teoporosis and fragility fractures. Analogously, vitamin D deficiency has been described as an independent risk factor for cardiovascular disease and metabolic disorders, such as insulin resistance and type 2 diabetes mellitus.
Last EACS guidelines suggest to screen for hypovitaminosis D every HIV-positive subject having a history of bone disease, chronic kidney disease or other known risk factors for vitamin D deficiency. Vitamin D repletion is recommended when 25-hydroxyvitamin D levels are below 10 ng/ml. Furthermore, it may be indicated in presence of 25OHD values between 10 and 30 ng/ml, if associated with osteoporosis, osteomalacia or increased parathyroid hormone levels. The optimal repletion and maintenance dosing regimens remain to be established, as well as the impact of vitamin D supplementation in preventing comorbidities.
PDF is attached at the bottom of this page
See also VitaminDWiki
Many infant infections avoided with supplementation with 400 IU of vitamin D – Oct 2012
Vitamin D probably can prevent and treat TB and infectious diseases – 2012
Introduction
The natural history of Human Immunodeficiency Virus (HIV) infection has been profoundly changed by the introduction of highly active antiretroviral therapy (HAART). Current treatment has dramatically reduced AIDS-related morbidity and mortality, since in most patients it guarantees undetectable levels of plasma HIV RNA and it leads to immune restoration; however, HAART cannot eradicate HIV2-14. Furthermore, increased life expectancy and drugs-associated adverse events expose HIV-infected subjects to age-related morbidities, including metabolic and cardiovascular disease (CVD), cancer, neurocognitive disorders, renal and bone dis-ease15-32. Many of these conditions appear to occur earlier in patients with HIV if compared with the general population. There is a large body of evidences that identifies chronic inflammation and immune activation as key-factors to explain premature age-associated non-AIDS-related events in patients receiving HAART33; indeed, it has been shown that plasma levels of several inflammatory and coagulopathic biomarkers, such as interleukin-6 (IL-6), D-Dimer and highly sensitive C-reactive protein (hs-CRP), are higher in the setting of HIV and correlate with the outcome of HIV infection34.
Bone disease represents one of the most common long-term complications of HIV infection35-37. The prevalence of reduced bone mineral density (BMD) and the risk of fragility fractures are higher among HIV-infected people38-41. Vitamin D deficiency is a well-established risk factor for bone disease in the general population; considering the high prevalence of hypovi-taminosis D among HIV-infected subjects, it is not surprising to find an association between vitamin D deficiency and osteopenia/osteoporosis in several HIV-positive cohorts42-48. In addition, recent data suggest that vitamin D is not only involved in calcium homeostasis, but it also has non-skeletal functions, including cardiovascular and immune regulation, cancer prevention and brain health49-53.
In this review, we first briefly describe vitamin D metabolism and biological functions; then, we focus on the most recent experimental and epi-demiological data dealing with the relationship between vitamin D deficiency and HIV infection, evaluating the extent of the problem, the pathogenic mechanisms, the clinical implications and potential benefits of vitamin D supplementation among HIV-infected subjects.
Vitamin D Metabolism and Optimal Circulating Levels: an Overview
Vitamin D was first identified as a key regulator of calcium homeostasis, since its deficiency was associated with rickets and osteomalacia54. Humans can introduce vitamin D with the diet, even if few foods, like oily fishes, cod liver oil and dairy products, naturally contain it55; the major production of vitamin D occurs in the skin, where the photochemical action of UVB light is able to transform 7-dehydrocholesterol to previtamin D, which is converted in turn to vitamin D through a non-enzymatic thermal isomerization. Vitamin D is then metabolized in the liver to 25-hydroxyvitamin D (25OHD) by 25a-hydroxylase; 25OHD is subsequently converted to the biologically active compound 1,25-dihydroxyvita-min D (1,25(OH)2D) by 1a-hydroxylase (or CYP27B 1)55,56. While 1a-hydroxylase is predominantly expressed in the kidney, it is now well known that several extrarenal tissues and cells, like monocyte-macrophages, are also able to convert 25OHD to 1,25(OH)2D50,57. A catabolic pathway involving 24a-hydroxylase (CYP24A1) is responsible for 25OHD and 1,25(OH)2D hydroxylation to inactive metabolites, named 24,25(OH)2D and 1,24,25(OH)3D, respectively58 (Figure 1).
In the kidney, 1a-hydroxylase activity is strictly controlled by calcium homeostatic signals, especially by parathyroid hormone (PTH), whose release by parathyroid glands is elicited by hypocalcemia. 1,25(OH)2D response to low serum calcium levels drives osteoclasts to release calcium from the bone, stimulates intestinal calcium absorption and reduces renal calcium ex-cretion55. To exert its functions, vitamin D interacts with vitamin D receptor (VDR), which is expressed by a large number of organs, including brain, muscles, adipose tissue, pancreas, colon, breast and immune cells. Activation of these VDRs is responsible for so-called nonclassic effects of vitamin D49-51. Several studies have linked vitamin D status with autoimmune diseases, like type 1 diabetes mellitus59, cardiovascular disease60, cancer61,62 and infections53,63-66 (Figure 2). In this context, 1,25(OH)2D has been recognized as an important mediator of both innate and adaptive immune responses. As previously described, antigen presenting cells (APCs), like macrophages and dendritic cells (DCs), are able to locally produce 1,25(OH)2D67; VDR is expressed not only on APCs, but also on T and B cells68. Vitamin D shows strong antimicrobial functions: activation of Toll-like receptor (TLR) pathways by pathogen-associated membrane patterns (PAMPs), shed by microbial agents, like Mycobacterium tuberculosis, is able to stimulate 1,25(OH)2D synthesis in monocyte-macrophages, leading to the production of antimicrobial peptides, like defensins and cathelicidin63,64,67. Furthermore, 1,25(OH)2D3 was shown to trigger autophagy in human macrophages, thus resulting in inhibition of HIV replication69.
Vitamin D status is usually assessed by measuring 25OHD circulating levels; optimal 25OHD plasma levels are still matter of debate: currently, 25OHD values > 30 ng/ml (75 nmol/l) are believed to be an adequate cut off for vitamin D sufficiency; 25OHD levels < 20 ng/ml (50 nmol/l) define vitamin D deficiency, whereas 25OHD levels ranging between 20 and 30 ng/ml indicate vitamin D insufficiency55,70.

Figure 1.
Vitamin D metabolism. 25OHD: 25-hydroxyvitamin D; 1,25(OH)2D: 1,25-dihydroxyvitamin D; 24,25(OH)2D: 24,25-dihydroxyvitamin D; Ca2+: calcium; P: phosphate.
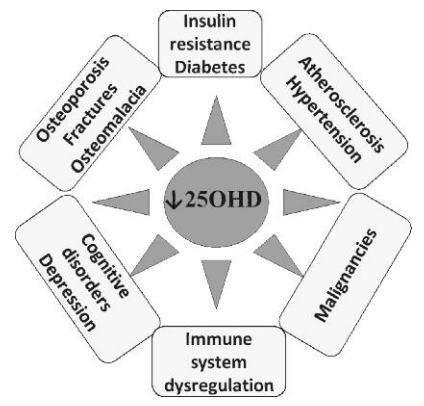
Figure 2 . Association between hypovitaminosis D and some morbidities.
Prevalence of Hypovitaminosis D in HIV-infected Subjects
Hypovitaminosis D is a worldwide disorder, with a high prevalence in the general population of both developing and Western countries71. According to the results coming from the National Health and Nutrition Examination Survey (NHANES), it is estimated that only 20-25% of American population has a serum 25OHD level of at least 30 ng/ml, whereas 25-30% is thought to be vitamin D deficient72,73. From this perspective, it is not surprising to find high rates of hypovitaminosis D even among HIV-infected subjects. A large US prospective cohort study (SUN study) assessed the prevalence of hypovita-minosis D in 672 HIV-positive subjects, demonstrating that 70.3% of them had 25OHD levels below 30 ng/ml, compared with 79.1% of HIV-negative US adults74. In a cross-sectional study evaluating vitamin D status in HIV-infected post-menopausal women living in New York, Stein et al44 found that 74% out of 89 HIV-positive women had 25OHD levels < 30 ng/ml; the prevalence rate was similar, however, in HIV-negative controls. They also found no differences in 1,25(OH)2D levels, which were normal in both groups. As already described in other reports, 25OHD levels were significant lower among African-American women, in comparison with Hispanic women. Hypovitaminosis D was recently reported to be highly prevalent also among HIV-positive premenopausal women. In a study enrolling 100 HIV-positive and 68 HIV-negative women, Yin et al45 observed that only 9% in both groups had 25OHD levels > 32 ng/ml; again, no difference by HIV status was found.
Data coming from EuroSIDA study, a prospective, observational work on a large cohort of HIV-positive subjects across 31 European countries, Israel and Argentina, confirmed hypovitaminosis D to be very common among HIV-positive indi-viduals75: 23.7% out of 1985 patients had indeed 25OHD below 10 ng/ml, 65.3% between 10 and 30 ng/ml and only 11% above 30 ng/ml. As expected, seasonality affected 25OHD levels, since the number of patients with hypovitaminosis D was higher among winter sampled individuals, in comparison with summer sampled ones. Similarly to the general population, older people were at higher risk of hypovitaminosis D; analogously, black ethnic origin was associated with a four-time odds to have low 25OHD. Furthermore, hy-povitaminosis D was independently associated with a higher risk of AIDS events and all-cause mortality. Kaplan-Meier curves of progression to these endpoints over a median 5-year follow up showed that the incidence of AIDS-defining events and death was significantly higher among vitamin D-deficient subjects, thus suggesting the possibility to use vitamin D as a new, independent, prognostic marker in HIV infection. In a French cohort of 2994 HIV-positive patients, Allavena et al77 have recently observed low 25OHD levels in 86.7% of subjects, including 55.6% with vitamin D insufficiency and 31.1% with vitamin D deficiency. In UK, Welz et al76 have demonstrated that 91% out of 1077 HIV-positive individuals had 25OHD below 75 nmol/l, with more than one-third showing severe vitamin D deficiency (25OHD < 25 nmol/l). In a recent Italian cross-sectional study, carried out by Vesci-ni et al78 with a large cohort of HIV-infected subjects (n=810), 47% and 6% of subjects were found to be vitamin D insufficient (30-75 nmol/l) and vitamin D deficient (< 30 nmol/l), respectively. Of interest, the Authors found a correlation between vitamin D insufficiency and the risk of cardiovascular events, diabetes mellitus and renal disease over a median 6.5-year follow up (relative hazard (RH) = 1.60, p = 0.05); furthermore, 25OHD levels below 30 nmol/l seemed to predict faster HIV progression (RH = 2.11, p = 0.08).
These data are in keeping with a number of previous investigations46,47,79,80 conducted on small HIV cohorts over the past few years, which had already described high prevalence rates of hypovitaminosis D in HIV-positive cohorts. In a group of 57 ambulatory HIV-positive patients, 25OHD levels below 32 ng/ml were reported in 74.4% of individuals by Rodriguez et al81. In Denmark, in a report that included 115 HIV-infected males, Bang et al82 observed that only 13% of subjects had 25OHD levels > 75 nmol/l. In 2010, another report revealed that 64% out of 200 HIV-subjects living in the South-Central United States had 25OHD < 20 ng/ml and 20.5% had 25OHD < 10 ng/ml. In multivariate analysis a significant correlation between low 25OHD levels and African-American race was found83. A similar prevalence rate for 25OHD deficiency was described by Mueller et al84 in the setting of the Swiss HIV cohort study. In a cohort of 211 HIV-positive individuals, 42% of patients in spring and 14% in fall had 25OHD levels below 30 nmol/l. It is important to note that 1-hydroxylation rate was significantly higher in patients with lower 25OHD, thus, suggesting a compensatory PTH-mediated response. Moreover, considering that normal 1,25(OH)2D levels may hide vitamin D deficiency and/or hyperparatyroidism, 1,25(OH)2D measurement does not represent a reliable option to evaluate vitamin D status. In addition, the Authors measured 1,25(OH)2D levels in a subgroup of 74 individuals and observed a significant association between a history ofAIDS-defining events and lower 1,25(OH)2D, but not lower 25OHD values. A number of investigations had previously reported 1,25(OH)2D to be lower in patients with advanced HIV infection48,85. It has been suggested that immunological hyperactivity and in particular tumor necrosis factor (TNF)-a overproduction may be responsible for the impairment of renal 1a-hy-droxylase, with the subsequent decrease in 1,25(OH)2D levels85.
Risk Factors for Vitamin D Deficiency in HIV Infection
The evaluation of risk factors for hypovitaminosis D in the setting of HIV infection includes the assessment of HIV-specific and HIV-independent risk factors (Figure 3); it is often challenging, however, to differentiate the direct impact of HIV infection from the effect of traditional risk factors which may be over-expressed in HIV-positive cohorts.
Some risk factors, such as female sex79, winter season75,76,78, increasing age45,75,78, low vitamin D dietary intake80,81,83 and dark skin pigmentation, are similar to those reported in HIV-negative cohorts71.
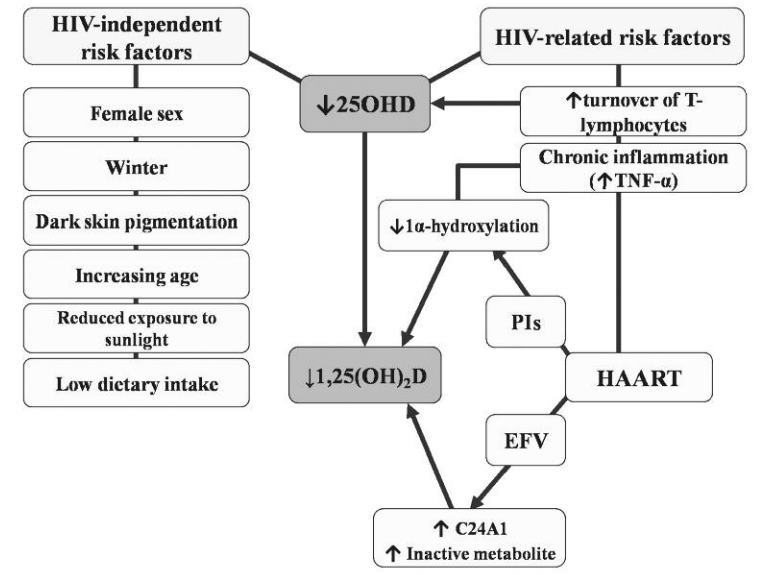
Figure 3. Risk factors for hypovitaminosis D in HIV infection. HIV: human immunodeficiency virus; 25OHD: 25-hydroxyvi-tamin D; 1,25(OH)2D: 1,25-dihydroxyvitamin D; HAART: highly active antiretroviral therapy; PIs: protease inhibitors; EFV: efavirenz; TNF-a: tumor necrosis factor-a.
Most reports have described a positive correlation between African American ethnicity and low 25OHD44,45,74-76,83,84. This observation is not unexpected, considering that in black people vitamin D cutaneous synthesis is markedly reduced86 and a greater exposure to UVB is necessary to produce the same amount of 25OHD as white individuals87. Conflicting data are available about the impact of body mass index (BMI) on 25OHD levels. As in the general population, some Authors74,83,84 have found a negative correlation between 25OHD serum levels and BMI, possibly because of vitamin D storage in adipose tissue88. By contrast, others have associated hypovitaminosis D with low BMI89 or have related vitamin D insufficiency with a higher risk of wasting, defined as BMI90 less than 18 kg/m2.
The relationship between 25OHD levels and CD4+ T-cell count is not clear cut. Some studies44,48,85 have described a positive correlation, some others74,79,82,83,90-92 have failed to demonstrate a significant association.
Vitamin D Status and HAART
Several in vitro and in vivo researches have evaluated the impact of antiretroviral drugs on vitamin D metabolism. Both protease inhibitors (PIs) and non-nucleoside reverse transcriptase inhibitors (NNRTIs) have been associated with the impairment of vitamin D metabolic pathways.
PIs have been shown to inhibit vitamin D 1 a-and 25a-hydroxylation in hepatocyte and monocyte cultures93: reduced conversion of 25OHD to its active metabolites may potentially explain the increased 25OHD levels found by some Authors in subjects with low 1,25(OH)2D74,79. As refers to NNRTIs, there is an increasing amount of data associating efavirenz (EFV) with compromised vitamin D homeostasis. EFV has been described indeed to increase 25OHD catabolism, through the induction of CYP2494,95 and reduced transcription of CYP2R1, a 25-hydroxylase96. A number of in vivo studies over the past years have supported this observation, describing an association between EFV assumption and low 25OHD levels. Welz et al76 found current EFV, but not nevirapine (NVP) use, to be associated with severe vitamin D deficiency (odds ratio (OR) 2 (C.I. 1.5-2.7), p < 0.001). In a recent re-port97, NNRTIs current use correlated with lower 25OHD levels: Pasquet et al found indeed an association between hypovitaminosis D and exposure to NNRTIs (p = 0.05), but not to EFV and NVP, probably because of a lack of statistical power of their analysis. However, considering the crude and adjusted coefficients for EFV and NVP in their regression models, the Authors suggested a NNRTI class effect, rather than a specific EFV impact, on vitamin D levels. Other stud-ies74,76,98-100 have reported opposing results, describing EFV, but not NVP influence on vitamin D homeostasis. Fux et al99 evaluated 25OHD levels in a cohort of 209 HIV-positive patients, before and one year after starting HAART. A significant reduction in 25OHD median values was observed among EFV-treated subjects (p < 0.001), but not among patients receiving a NVP-based or PI-based treatment. Consistent with these findings, the Authors also reported the results of a cross-sectional study performed on 262 individuals, showing 25OHD levels to be higher in patients exposed to NVP (p = 0.04) and PIs (p = 0.048), in comparison with those exposed to EFV. Nevertheless, after adjusting for confounding factors, 25OHD levels remained significantly lower in the EFV group, in comparison with the PI group (p = 0.006), but not the NVP group (p = 0.2). Of interest, a recent article of Boven et al101 showed that vitamin D levels did not significantly change in comparison with baseline values among patients receiving the new NNRTI rilpivirine over 48 weeks, whereas a significant decrease was observed among those starting a EFV-based regimen. Moreover, of the patients with baseline 25OHD insufficiency/deficiency, a smaller proportion developed severe 25OHD deficiency with rilpivirine than EFV. Analogously, Allavena et al77 found vitamin D deficiency to be associated with the administration of EFV [adjusted OR 1.89 (1.45-2.47)]; Brown et al95 showed a significant decline in 25OHD serum levels after the initiation of a EFV-based regimen, in comparison with a non-EFV-based regimen (p < 0.001). In addition, subjects receiving EFV had a 1.8 times increased Odds to develop vitamin D deficiency, if compared with those starting PIs. Similar results have been shown by other groups84,89: Conesa-Botella et al89, for instance, described a 3-fold increased risk to have 25OHD levels below 20 ng/ml among subjects receiving NNRTIs (p = 0.02) after 12 months of HAART. In the MONET study102, in which virologically controlled patients were switched to a darunavir-based regimen, lower baseline vitamin D levels were associated with efavirenz (p = 0.0062) and zidovudine (p = 0.015) use and a 27% increase in 25OHD values was observed in subjects discontinuing EFV (p = 0.007). Van Den Bout-Van Den Beukel et al79 reported higher 25OHD levels in white subjects receiving PIs in comparison with those receiving NNRTIs (p = 0.007) or treatment-naive (p = 0.049). Similarly, in the EuroSIDA cohort75, patients receiving a PI-based antiretroviral regimen were at low risk of hypovitaminosis D, whereas no significant association with EFV or TDF use was found.
One drawback of most studies is their cross-sectional design, so that causal relationships cannot be inferred.
Taken together, these data suggest the need for large prospective studies, properly designed to evaluate the specific effects and clinical impact of antiretroviral drugs on vitamin D status.
HIV Infection, Bone Disease and Vitamin D
Bone disease is one of the most common long-term complications in HIV-infected individuals35-37.
Many studies have shown low BMD values, including osteopenia and osteoporosis, among subjects with HIV, with an increased risk of fragility fractures38-41,103.
The pathophysiological mechanisms of bone disease in HIV infection are complex and partially unknown. In addition to the classical risk factors, shared with uninfected subjects (low BMI, corticosteroid use, increasing age, hypogo-nadism, including post-menopausal status in women, prolonged immobility and smoking), other factors are probably involved, such as the direct effects of HIV on osteoblast and osteoclast functions, chronic inflammation and antiretrovi-ral interference with bone turnover35. In particular, TDF assumption has been associated with low BMD, because of its capability to induce proximal renal tubular dysfunction, which may cause in turn renal phosphate wasting and finally BMD loss104. Patients starting a TDF-based regimen have a greater BMD loss in comparison with those treated with other antiretrovirals105,106. In addition, higher PTH levels have been described in subjects receiving TDF with suboptimal 25OHD levels107-109. Childs et al109 reported an independent association between PTH levels and TDF assumption (p = 0.017), with the highest PTH levels among TDF-treated subjects with 25OHD below 30 ng/ml (p = 0.045). A similar association between PTH concentration, TDF use and 25OHD levels has been described by Pocaterra et al110. The role of PIs on bone homeostasis appears controversial, since some studies have described an association between BMD loss and PI-based therapy38,41,111-114, but others have not37,47. When comparing HAART-naive and HAART-experienced subjects, most cross-sectional studies reported similar BMD val-ues36,37,46,111,113. On the contrary, a meta-analysis of pooled data coming from ten cross-sectional studies conducted over a 5-year period, between 2000 and 2005, none showing significant differences individually, reported a significant BMD reduction in HAART-treated patients (OR 2.5 (95% CI 1.8-3.7)39. Longitudinal studies evaluating the changes in BMD in subjects starting HAART have shown that the initiation of anti-retroviral therapy is associated with a 2%-6% decrease in BMD within the first year106,115. The negative impact of HAART on bone health has been confirmed in the SMART study, where subjects receiving continuous HAART were described to have a greater BMD loss in comparison with those on intermittent HAART116.
Hypovitaminosis D is considered a traditional risk factor for bone disease in the general population35; similarly, in the setting of vitamin D deficient HIV cohorts, low 25OHD values have been associated with BMD loss. In premenopausal HIV-positive women, Yin et al45 found that vitamin D levels below 20 ng/ml, but not antiretroviral therapy, were predictive of FN (femoral neck) bone loss; Stein et al44 described a weak association between serum 25OHD and LS (lower spine) BMD in Hispanic (r = 0.33, p = 0.01), but not African-American postmenopausal HIV-infected women.
Some prospective randomized trials117-119 have evaluated the effects of alendronate on BMD in HIV-positive subjects with osteopenia and osteoporosis, showing that the combination of weekly alendronate with daily calcium and vitamin D supplementation was effective and safe in increasing LS BMD at one year; of interest, even though in the placebo group receiving vitamin D and calcium supplements alone BMD variations were not statistically significant, a trend towards BMD increase was seen. These data highlight the need for further prospective studies, specifically designed to evaluate the effect of vitamin D replacement on BMD in vitamin D deficient HIV-infected subjects; in addition, considering the low vitamin D dose administered in the trials mentioned above, further investigations looking at the effects of higher dose on BMD are warranted.
Association Between HIV, Hypovitaminosis D and Cardiovascular Disease (CVD)
Several studies have described the association between HIV and increased risk of CVD120-123. HIV infection itself is considered an independent risk factor for atherosclerosis: in fact, the prevalence of atherosclerosis is higher among HIV-positive subjects and it occurs earlier as compared with uninfected individuals124,125. In addition, antiretroviral drugs-related dyslipidemia, chronic inflammation and immune activation may also affect atherogenesis in the setting of HIV33,126.
In the general population, vitamin D deficiency has been linked to CVD60. Considering the high prevalence of both hypovitaminosis D and CVD in patients with HIV, the observation of a relationship between low 25OHD and atherosclerosis in HIV-positive subjects is not surprising. In a cross-sectional study of 139 patients, Choi et al127 have shown an independent association between vitamin D insufficiency and cIMT (carotid intima-media thickness), a common marker for atherosclerotic vascular disease. Furthermore, they found that mean cIMT was 0.13 mm greater in vitamin D insufficient subjects, as compared to individuals with normal 25OHD levels. Similarly, Ross et al128 observed a 10.62 higher odds to have cIMT above the median value among subjects with 25OHD values below 30 ng/ml (p = 0.01). A recent report by Shikuma et al129 has described a significant correlation between 25OHD and brachial artery flow-mediated dilation (FMD), a early marker of endothelial dysfunction (r = 0.3; p = 0.01), but not with cIMT, possibly because of the small sample size. Lai et al130 found that 25OHD deficiency was independently associated with a greater than two-fold increase in the risk of significant coronary stenosis in a cohort of cardiovascularly asymptomatic HIV-positive African Americans.
Other traditional risk factors for CVD, such as insulin resistance and diabetes mellitus, are frequently seen in HIV-positive individuals131,132 and, as in the general population133, an association between vitamin D status and type 2 diabetes has been described134. In a recent cross-sectional study, Szep et al134 have reported lower 25OHD levels among subjects with type 2 diabetes, in comparison with those without diabetes (p < 0.001), although vitamin D deficiency was highly prevalent in both groups. Furthermore, vitamin D deficiency was independently associated with diabetes (OR 1.85 (CI 1.03-3.32), p = 0.038), but not with metabolic syndrome. One explanation may be vitamin D involvement in the regulation of insulin secretion and insulin-mediated glucose trans-port135-137. In addition, in diabetic HIV-negative subjects vitamin D supplementation was able to reduce insulin-resistance138. In the setting of HIV, the effects of vitamin D supplementation on insulin sensitivity need to be evaluated with large, prospective studies since nowadays only few data are available. A small prospective study, conducted by Van den Bout et al139, has surprisingly shown that cholecalciferol supplementation (2000 IU/day for 14 weeks, 1000 IU/day until 48 weeks) led to increased insulin resistance, evaluated with HOMA index and fasting glucose levels, after 24 weeks, whereas no differences were seen after 48 weeks. An explanation may be the inhibition of the expression of PPAR-y receptor by 1,25(OH)2D140-142, considering that the activation of PPAR-y pathway leads to increased insulin sen-sitivity143. It remains to be clarified if the effect of cholecalciferol on insulin sensitivity seen by Van den Bout et al139 is dose- or time-dependent, but this report further suggests the importance of clinical trials extensively evaluating the pros and cons of supplementing HIV-infected individuals with vitamin D.
Chronic inflammation may contribute to the increased cardiovascular risk of HIV-positive subjects. In fact, hs-CRP and IL-6 have been shown to predict both cardiovascular and HIV disease progression and mortality34. Recent studies have described an association between hypovitaminosis D and increased inflammation markers in the setting of HIV infection. Ansemant et al144 found severe vitamin D deficiency to correlate with increased hs-CRP (p = 0.04) and IL-6 (p = 0.001) levels in a cohort of 263 HIV-infected patients; analogously, Poudel-Tan-dukar et al145 reported that the risk of having high inflammation (CRP > 3 mg/l) was greater among HIV-positive people with a 25(OH)D serum level of < 20 ng/ml. However, given the cross-sectional design and the small size of these researches, there is a need for large, prospective studies, evaluating the causal relationship between inflammation and hypovitaminosis D and the safety and efficacy of vitamin D supplementation.
Management of Hypovitaminosis D in HIV-Positive Individuals
Last European AIDS Clinical Society (EACS) guidelines suggest to screen for vitamin D status every HIV-positive subject having a history of low bone mineral density and/or fracture, high risk for fracture or chronic kidney disease. It is also suggested to test all HIV-infected individuals having other risk factors for hypovitaminosis D. If 25OHD levels are below 10 ng/ml, replacement is recommended, whereas in vitamin D insufficient individuals vitamin D supplementation is suggested in presence of osteoporosis, osteomalacia or increased PTH146.
In the general population, current recommended dietary allowances (RDA) of vitamin D are 700-800 IU cholecalciferol/day55. Serum 25OHD levels generally increase by approximately 1 ng/ml for every 100 IU of vitamin D intake147. Few data coming from small cohorts are available about the efficacy of vitamin D repletion in HIV-infected subjects. Some Authors have described vitamin D supplementation as a safe and well tolerated option to successfully increase 25OHD levels: Van den Bout et al139, for instance, treated 20 vitamin D deficient subjects with 2000 IU cholecalciferol/day for 14 weeks, after which the dosage was lowered to 1000 IU/day until 48 weeks. The evaluation of 25OHD3 and 1,25(OH)2D3 levels at 24 weeks showed normalized values, but after 48 weeks only serum 25OHD3 was significantly different from baseline, whereas 1,25(OH)2D3 returned to baseline levels, probably because of the reduction in cholecalciferol dose. Arpadi et al91 reported the benefits of orally administered bimonthly doses of 100,000 IU cholecalciferol and 1g/day calcium in a cohort of HIV-infected children and adolescents during a 12-month period: 25OHD levels were found to be significantly higher among supplemented subjects, in comparison with individuals receiving placebo; 44.4% of subjects in the group receiving cholecalciferol and calcium had 25OHD > 30 ng/ml after 1 year vs 11.1% observed in the placebo group. Havens et al148 demonstrated a rapid increase in 25OHD serum concentration in a cohort of 207 adolescents and young adults with HIV when orally administering vitamin D3 (50000 IU in three doses at monthly intervals). By contrast, other studies reported less encouraging results. In a small study, only 40% of patients receiving oral vitamin D supplements had 25OHD levels > 30 ng/ml after a median 16 week follow up149; similarly, just a 46% reduction in hypovitaminosis D was found in a cohort of HIV-positive young adults receiving 50000 IU vitamin D3/week for 12 weeks150. Lastly, a recent randomized clinical trial reported a significant increase in 25OHD after 12 weeks of daily supplementation with 4000 IU vitamin D3 only in subjects receiving a non-EFV-based regimen (p = 0.011)151,152.
Conclusions
Vitamin D deficiency is emerging as a matter of great concern among HIV-infected subjects.
Considering that hypovitaminosis D represents a risk factor for skeletal and extra-skeletal health, clinicians should consider screening all HIV patients at risk for vitamin D deficiency.
Prospective studies are necessary to determine the optimal dosage of vitamin D supplementation and to assess the impact of vitamin D supplementation on prevention of comorbidities among HIV-infected people. Furthermore, the exact association between exposure to antiretroviral drugs and hypovitaminosis D needs to be clarified.
Conflict of Interest None declared.
References
1) Palella FJ, Baker RK, Moorman AC, Chmiel JS, Wood KC, Brooks JT, Holmberg SD, HIV Outpatient Study Investigators. Mortality in the highly active antiretroviral therapy era: changing causes of death and disease in the HIV outpatient study. J Acquir Immune Defic Syndr 2006; 43: 27-34.
2) Deeks SG, Phillips AN. HIV infection, antiretroviral treatment, ageing, and non-AIDS related morbidity. BMJ 2009; 338: a3172.
3) Bhavan KP, Kampalath VN, Overton ET. The aging of the HIV epidemic. Curr HIV/AIDS Rep 2008; 5: 150-158.
4) Martellotta F, Berretta M, Vaccher E, Schioppa O, Zanet E, Tirelli U. AIDS-related Kaposi's sarcoma: state of the art and therapeutic strategies. Curr HIV Res 2009;7:634-638.
5) Berretta M, Cinelli R, Martellotta F Spina M, Vaccher E, Tirelli U. Therapeutic approaches to AIDS-related malignancies. Oncogene 2003; 22: 6646-6659.
6) Nunnari G, Berretta M, Pinzone MR, Di Rosa M, Cappellani A, Berretta S, Tirelli U, Malaguarnera M, Schnell JM, Cacopardo B. Hepatocellular carcinoma in HIV positive patients. Eur Rev Med Pharmacol Sci 2012; 16: 1257-1270.
7) Pinzone MR, Fiorica F, Di Rosa M, Malaguarnera G, Malaguarnera L, Cacopardo B, Zangh'i G, Nunnari G. Non-AIDS-defining cancers among HIV-infected people. Eur Rev Med Pharmacol Sci 2012; 16: 1377-1388.
8) Dornadula G, Nunnari G, Vanella M, Roman J, Babinchak T, DeSimone J, Stern J, Braffman M, Zhang H, Pomerantz RJ. Human immunodeficiency virus type 1-infected persons with residual disease and virus reservoirs on suppressive highly active antiretroviral therapy can be stratified into relevant virologic and immunologic subgroups. J Infect Dis 2001; 183: 1682-1687.
9) Nunnari G, Otero M, Dornadula G, Vanella M, Zhang H, Frank I, Pomerantz RJ. Residual HIV-1 disease in seminal cells of HIV-1-infected men on suppressive HAART: latency without on-going cellular infections. AIDS 2002; 16: 39-45.
10) Nunnari G, Leto D, Sullivan J, Xu Y, Mehlman Ke, Kulkosky J, Pomerantz RJ. Seminal reservoirs during an HIV type 1 eradication trial. AIDS Res Hum Retroviruses 2005; 21: 768-775.
11) Nunnari G, Sullivan J, Xu Y, Nyirjesy P, Kulkosky J, Cavert W, Frank I, Pomerantz RJ. HIV type 1 cervi-covaginal reservoirs in the era of HAART. AIDS Res Hum Retroviruses 2005; 21: 714-718.
12) Nunnari G, Argyris E, Fang J, Mehlman KE, Pomerantz RJ, Daniel R. Inhibition of HIV-1 replication by caffeine and caffeine-related methylxanthines. Virology 2005; 335: 177-184.
13) Smith JA, Nunnari G, Preuss M, Pomerantz RJ, Daniel R. Pentoxifylline suppresses transduction by HIV-1 -based vectors. Intervirology 2007; 50:377-386.
14) Pinzone MR, Di Rosa M, Cacopardo B, Nunnari G. HIV RNA suppression and immune restoration: can we do better? Clin Dev Immunol 2012; 2012: 515962.
15) Berretta M, Di Benedetto F, Dal Maso L, Cacopardo B, Nasti G, Facchini G, BearzA, Spina M, Garlassi E, De Re V, Fiorica F, Lleshi A, Tirelli U. Sorafenib for the treatment of unresectable hepatocellular carcinoma in HIV-positive patients. Anticancer Drugs 2013;24:212-218.
16) Zanet E, Berretta M, Di Benedetto F, Talamini R, Ballarin R, Nunnari G, Berretta S, Ridolfo A, Lleshi A, Zanghi A, Cappellani A, Tirelli U. Pancreatic cancer in HIV-positive patients: a clinical case-control study. Pancreas 2012;41:1331-1335.
17) Zanet E, Berretta M, Martellotta F, Cacopardo B, Fisichella R, Tavio M, Berretta S, Tirelli U. Anal cancer: focus on HIV-positive patients In the HAART Era. Curr HIV Res 2011; 9: 70-81.
18) Bearz A, Vaccher E, Talamini R, Berretta M, Tirelli U. Comment on 'Lung cancer in the Swiss HIV Cohort Study: role of smoking, immunodeficiency and pulmonary infection'. Br J Cancer 2012; 106: 1899-1900.
19) Berretta M, Garlassi E, Cacopardo B, Cappellani A, Guaraldi G, Cocchi S, De Paoli P, Lleshi A, Izzi I, Torresin A, Di Gangi P, Pietrangelo A, Ferrari M, Bearz A, Berretta S, Nasti G, Di Benedetto F, Balestreri L, Tirelli U, Ventura P. Hepatocellular carcinoma in HIV-infected patients: check early, treat hard. Oncologist 2011; 16: 1258-1269.
20) Di Benedetto F De Ruvo N, Berretta M, Masetti M, Montalti R, Di Sandro S, Ballarin R, Codeluppi M,
Guaraldi G, Gerunda GE. Hepatocellular carcinoma in HIV patients treated by liver transplantation. Eur J Surg Oncol 2008; 34: 422-427.
21) Berretta M, Tirelli U. Colorectal cancer screening in HIV-infected patients 50 years of age and older: missed opportunities for prevention. Am J Gastroenterol 2006; 101: 907.
22) Berretta M, Lleshi A, Cappellani A, Bearz A, Spina M, Talamini R, Cacopardo B, Nunnari G, Montesar-chio V, Izzi I, Lanzafame M, Nasti G, Basile F Berretta S, Fisichella R, Schiantarelli C, Garlassi E, Ridol-fo A, Guella L, Tirelli U. Oxaliplatin based chemotherapy and concomitant highly active anti-retroviral therapy in the treatment of 24 patients with colorectal cancer and HIV infection. Curr HIV Res 2010;8:218-222.
23) Nunnari G, Smith JA, Daniel R. HIV-1 Tat and AIDS-associated cancer: targeting the cellular anti-cancer barrier. J Exp Clin Cancer Res 2008; 27:3.
24) Berretta M, Cappellani A, Di Benedetto F, Lleshi A, Talamini R, Canzonieri V, Zanet E, Bearz A, Nasti G, Lacchin T, Berretta S, Fisichella R, Balestreri L, Tor-resin A, Izzi I, Ortolani P, Tirelli U. Clinical presentation and outcome of colorectal cancer in hiv-positive patients: a clinical case-control study. Onkologie 2009; 32: 319-324.
25) Spina M, Berretta M, Tirelli U. Hodgkin's disease in HIV. Hematol Oncol Clin North Am 2003; 17: 843858.
26) Otero M, Nunnari G, Leto D, Sullivan J, Wang FX, Frank I, Xu Y, Patel C, Dornadula G, Kulkosky J, Pomerantz RJ. Peripheral blood Dendritic cells are not a major reservoir for HIV type 1 in infected individuals on virally suppressive HAART. AIDS Res Hum Retroviruses 2003; 19:1097-1103.
27) Pinzone MR, Cacopardo B, Condorelli F, Di Rosa M, Nunnari G. Sirtuin-1 and HIV-1: an overview. Curr Drug Targets 2013 Apr 3 [Epub ahead of print].
28) Nunnari G, Xu Y, Acheampong EA, Fang J, Daniel R, Zhang C, Zhang H, Mukhtar M, Pomerantz RJ. Exogenous IL-7 induces Fas-mediated human neu-ronal apoptosis: potential effects during human immunodeficiency virus type 1 infection. J Neu-rovirol 2005; 11:319-328.
29) Nunnari G, Pomerantz RJ. IL-7 as a potential therapy for HIV-1-infected individuals. Expert Opin Biol Ther 2005; 5: 1421-1426.
30) Kulkosky J, Bouhamdan M, Geist A, Nunnari G, Phinney DG, Pomerantz RJ. Pathogenesis of HIV-1 infection within bone marrow cells. Leuk Lymphoma 2000;37:497-515.
31) Pomerantz RJ, Nunnari G. HIV and GB virus C--can two viruses be better than one? N Engl J Med 2004; 350: 963-965.
32) Pinzone MR, Celesia BM, Di Rosa M, Cacopardo B, Nunnari G. Microbial translocation in chronic liver diseases. Int J Microbiol 2012; 2012: 694629.
33) Appay V, Sauce D. Immune activation and inflammation in HIV-1 infection: causes and consequences. J Pathol 2008; 214: 231 -241.
34) Nxon DE, LandayAL Biomarkers of immune dysfunction in HIV. Curr Opin HIV AIDS 2010; 5: 498-503.
35) Stone B, Dockrell D, Bowman C, McCloskey E. HIV and bone disease. Arch Biochem Biophys 2010; 503: 66-77.
36) Knobel H, Guelar A, Vallecillo G, Nogues X, Diez A. Osteopenia in HIV-infected patients: is it the disease or is it the treatment? AIDS 2001; 15: 807-808.
37) Cazanave C, Dupon M, Lavignolle-Aurillac V, Barthe N, Lawson-Ayayi S, Mehsen N, Mercie P, Morlat P, Thiebaut R, Dabis F, Groupe d'Epidemi-ologie Clinique du SIDA en Aquitaine. Reduced bone mineral density in HIV-infected patients: prevalence and associated factors. AIDS 2008; 22: 395-402.
38) Tebas P, Powderly WG, Claxton S, Marin D, Tan-tisiriwat W, Teitelbaum SL, Yarasheski KE. Accelerated bone mineral loss in HIV-infected patients receiving potent antiretroviral therapy. AIDS 2000; 14: F63-67.
39) Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS 2006; 20: 2165-2174.
40) Triant VA, Brown TT, Lee H, Grinspoon SK. Fracture prevalence among human immunodeficiency virus (HIV)-infected versus non-HIV-infected patients in a large U.S. healthcare system. J Clin Endocrinol Metab 2008; 93: 3499-3504.
41) CalmyA, Fux CA, Norris R, Vallier N, Delhumeau C, Samaras K, Hesse K, Hirschel B, Cooper DA, Carr A. Low bone mineral density, renal dysfunction, and fracture risk in HIV infection: a cross-sectional study. J Infect Dis 2009; 200: 1746-1754.
42) Overton ET, Mondy K, Bush T. Factors Associated with Low Bone Mineral Density in a Large Cohort of HIV-infected US Adults: Baseline Results from the SUN Study. Proceedings of the 14th Conference on Retroviruses and Opportunistic Infections 2007. Los Angeles, CA: Abstract 836.
43) Arnsten JH, Freeman R, Howard AA, Floris-Moore M, Lo Y, Klein RS. Decreased bone mineral density and increased fracture risk in aging men with or at risk for HIV infection. AIDS 2007; 21: 617-623.
44) Stein EM, Yin MT, McMahon DJ, Shu A, Zhang CA, Ferris DC, Colon I, Dobkin JF, Hammer SM, Shane E. Vitamin D deficiency in HIV-infected post-menopausal Hispanic and African-American women. Osteoporos Int 2010; 22: 477-487.
45) Yin MT, Lu D, Cremers S, Tien PC, Cohen MH, Shi Q, Shane E, Golub ET, Anastos K. Short-term bone loss in HIV-infected premenopausal women. J Ac-quir Immune Defic Syndr 2010; 53: 202-208.
46) Seminari E, Castagna A, Soldarini A, Galli L, Fusetti G, Dorigatti F, Hasson H, Danise A, Guffanti M, Lazzarin A, Rubinacci A. Osteoprotegerin and bone turnover markers in heavily pretreated HIV-infected patients. HIV Med 2005; 6: 145-150.
47) Garcia Aparicio AM, Fernandez SM, Gonzalez J, Ar-ribasJR, PenaJM, Vazquez JJ, Martinez ME, CoyaJ, Martin Mola E. Abnormalities in the bone mineral metabolism in HIV-infected patients. Clin Rheumatol 2006; 25: 537-539.
48) Teichmann J, Stephan E, Lange U, Discher T, Friese G, Lohmeyer J, Stracke H, Bretzel RG. Os-teopenia in HIV infected women prior to highly active antiretroviral therapy. J Infect 2003; 46: 221-227.
49) Bikle D. Nonclassic actions of vitamin D. J Clin Endocrinol Metab 2009; 94: 26-34.
50) Di Rosa M, Malaguarnera G, De Gregorio C, Palumbo M, Nunnari G, Malaguarnera L. Im-muno-modulatory effects of vitamin D3 in human monocyte and macrophages. Cell Immunol 2012; 280: 36-43.
51) Di Rosa M, Malaguarnera L, Nicolosi A, Sanfilippo C, Mazzarino C, Pavone P, Berretta M, Cosentino S, Cacopardo B, Pinzone MR, Nunnari G. Vitamin D3: an ever green molecule. Front Biosci (Schol Ed) 2013;5:247-260.
52) Nunnari G, Coco C, Pinzone MR, Pavone P, Berretta M, Di Rosa M, Schnell M, Calabrese G, Cacopardo B. The role of micronutrients in the diet of HIV-infected individuals. Front Biosci (Elite Ed) 2012;4:2442-2456.
53) Cacopardo B, Camma C, Petta S, Pinzone MR, Cappellani A, Zanghi A, Nicolosi A, Nunnari G. Diagnostic and therapeutical role of vitamin D in chronic hepatitis C virus infection. Front Biosci (Elite Ed). 2012; 4: 1276-1286.
54) Mozolowsk W. Jedrzej Sniadecki (1768-1838) on the cure of rickets. Nature 1939; 143:121.
55) Holick MF. Vitamin D Deficiency. N Engl J Med 2007;357:266-281.
56) Prosser DE, Jones G. Enzymes involved in the activation and inactivation of vitamin D. Trends Biochem Sci 2004; 29: 664-673.
57) Stoffels K, Overbergh L, Bouillon R, Mathieu C. Immun e regulation of 1 alpha-hydroxylase in murine peritoneal macrophages: unravelling the IFN-gamma pathway. J Steroid Biochem Mol Biol 2007; 103: 567-571.
58) Omdahl JL, Morris HA, May BK. Hydroxylase enzymes of the vitamin D pathway: expression, function, and regulation. Annu Rev Nutr 2002; 22: 139-166.
59) Hypponen E, LAarA E, Reunanen A, Jarvelin MR, Vir-tanen SM. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet 2001; 358: 1500-1503.
60) Wang TJ, Pencina MJ, Booth SL Jacques PF, Ingels-son E, Lanier K, Benjamin EJ, DAgostino RB, Wolf M, Vasan RS. Vitamin D deficiency and risk of cardiovascular disease. Circulation 2008; 117: 503511.
61) Hines SL, Jorn HK, Thompson KM, Larson JM. Breast cancer survivors and vitamin D: a review. Nutrition 2010;26:255-262.
62) Gorham ED, Garland CF, Garland FC, Grant WB, Mohr SB, Lipkin M, Newmark HL, Giovannucci E,
Wei M, Holick MF. Vitamin D and prevention of colorectal cancer. J Steroid Biochem Mol Biol 2005; 97: 179-194.
63) Liu PT, Stenger S, Tang DH, Modlin RL. Cutting edge: vitamin D-mediated human antimicrobial activity against Mycobacterium tuberculosis is dependent on the induction of cathelicidin. J Immunol 2007; 179: 2060-2063.
64) Krutzik SR, Hewison M, Liu PT, Robles JA, Robles JA, Stenger S, Adams JS, Modlin RL. IL-15 links TLR2/1-induced macrophage differentiation to the vitamin D-dependent antimicrobial pathway. J Immunol 2008; 181: 7115-7120.
65) Nnoaham KE, Clarke A. Low serum vitamin D levels and tuberculosis: a systematic review and meta-analysis. Int J Epidemiol 2008; 37:113-119.
66) Ginde AA, Mansbach JM, Camargo Jr CA. Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the Third National Health and Nutrition Examination Survey. Arch Intern Med 2009; 169: 384-390.
67) Adams JS, Hewison M. Unexpected actions of vitamin D: new perspectives on the regulation of innate and adaptive immunity. Nat Clin Pract Endocrinol Metab 2008; 4: 80-90.
68) Provvedini DM, Tsoukas CD, Deftos LJ, Manolagas SC. 1,25-Dihydroxyvitamin D3 receptors in human leukocytes. Science 1983; 221: 1181 -1183.
69) Campbell GR, Spector SA. Hormonally active vitamin D3 (1,25-Dihydroxycholecalciferol) triggers autophagy in human macrophages that inhibits HIV-1 infection. J Biol Chem 2011; 286: 1889018902.
70) Adams JS, Hewison M. Update in Vitamin D. J Clin Endocrinol Metab 2010; 95: 471-478.
71) Arabi A, El Rassi R, El-Hajj Fuleihan G. Hypovita-minosis D in developing countries-prevalence, risk factors and outcomes. Nat Rev Endocrinol 2010; 6: 550-561.
72) GindeAA, Liu MC, Camaargo CA. Demographic differences and trends of vitamin D insufficiency in the US population, 1988-2004. Arch Intern Med 2009; 169: 626-632.
73) Yetley EA. Assessing the vitamin D status of the US population. Am J Clin Nutr 2008; 88: 558S-564S.
74) Dao CN, Patel P, Overton ET, Rhame F, Pals SL, Johnson C, Bush T, Brooks JT, and the Study to Understand the Natural History of HIV and AIDS in the Era of Effective Therapy (SUN) Investigators. Low vitamin D among HIV-infected adults: prevalence of and risk factors for low vitamin D Levels in a cohort of HIV-infected adults and comparison to prevalence among adults in the US general population. Clin Infect Dis 2011; 52: 396-405.
75) Viard JP, Souberbielle JC, Kirk O, Reekie J, Knysz B, Losso M, Gatell J, Pedersen C, Bogner JR, Lund-gren JD, Mocroft A, for the EuroSIDA Study Group. Vitamin D and clinical disease progression in HIV infection: results from the EuroSIDA study. AIDS 2011; 25: 1305-1315.
76) Welz T, Childs K, Ibrahim F, Poulton M, Taylor CB, Moniz CF, Post FA. Efavirenz is associated with severe vitamin D deficiency and increased alkaline phosphatase. AIDS 2010; 24: 1923-1928.
77) Allavena C, Delpierre C, Cuzin L, Rey D, Viget N, Bernard J, Guillot P, Duvivier C, Billaud E, Raffi F. High frequency of vitamin D deficiency in HIV-in-fected patients: effects of HIV-related factors and antiretroviral drugs. J Antimicrob Chemother 2012;67:2222-2230.
78) Vescini F Cozzi-Lepri A, Borderi M, Re MC, Maggio-lo F, De Luca A, Cassola G, Vullo V, Carosi G, Anti-nori A, Tozzi V, Monforte AD, for the Icona Foundation Study Group. Prevalence of hypovitaminosis d and factors associated with vitamin D deficiency and morbidity among HIV-infected patients enrolled in a large italian Cohort. J Acquir Immune Defic Syndr 2011; 58: 163-172.
79) Van Den Bout-Van Den Beukel CJ, Fievez L, Michels M, Sweep FC, Hermus AR, Bosch ME, Burger DM, Bravenboer B, Koopmans PP, Van Der Ven AJ. Vitamin D deficiency among HIV type 1-infected individuals in the Netherlands: effects of antiretroviral therapy. AIDS Res Hum Retroviruses 2008; 24: 1375-1382.
80) Stephensen CB, Marquis GS, Kruzich LA, Douglas SD, Aldrovandi GM, Wilson CM. Vitamin D status in adolescents and young adults with HIV infection. Am J Clin Nutr 2006; 83: 1135-1141.
81) Rodriguez M, Daniels B, Gunawardene S, Robbins GK. High frequency of vitamin D deficiency in ambulatory HIV-Positive patients. AIDS Res Hum Retroviruses 2009; 25: 9-14.
82) Bang UC, Shakar SA, Hitz MF, Jespersen MS, Andersen O, Nielsen SD, Jensen JE. Deficiency of 25-hy-droxyvitamin D in male HIV-positive patients: a descriptive cross-sectional study. Scand J Infect Dis 2010;42:306-310.
83) Crutchley RD, Gathe JC, Mayberry C, Trieu A, Abughosh S, Garey KW. Risk factors for vitamin D deficiency in HIV-infected patients in the South Central United States. AIDS Res Hum Retroviruses 2012;28:454-459.
84) Mueller NJ, Fux CA, Ledergerber B, Elzi L Schmid P, Dang T, Magenta L CalmyA, Vergopoulos A, Bischoff-Ferrari HA, Swiss HIV Cohort Study. High prevalence of severe vitamin D deficiency in combined antiretroviral therapy naive and successfully treated Swiss HIV patients. AIDS 2010; 24:1127-1134.
85) Haug CJ, Aukrust P, Haug E, Morkrid L, Muller F, Froland SS. Severe deficiency of 1,25-dihydrox-yvitamin D3 in human immunodeficiency virus infection: Association with immunologic hyper-activity and only minor changes in calcium homeostasis. J Clin Endocrinol Metab 1998; 83: 3832-3838.
86) Holick MF. Resurrection of vitamin D deficiency and rickets. J Clin Invest 2006; 116: 2062-2072.
87) Willis CM, Laing EM, Hall DB, Hausman DB, Lewis RD. A prospective analysis of plasma 25-hydroxyvitamin D concentrations in white and black prepubertal females in the southeastern United States. Am J Clin Nutr 2007; 85: 124130.
88) Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF. Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr 2000; 72: 690-693.
89) Conesa-Botella A, Florence E, Lynen L, Colebun-ders R, Menten J, Moreno-Reyes R. Decrease of vitamin D concentration in patients with HIV infection on a non nucleoside reverse transcriptase inhibitor-containing regimen. AIDS Res Ther2010; 7: 1-6.
90) Mehta S, Mugusi FM, Spiegelman D, Villamor E, Finkelstein JL, Hertzmark E, Giovannucci EL, Msamanga Gl, Fawzi WW. Vitamin D status and its association with morbidity including wasting and opportunistic illnesses in HIV-infected women in Tanzania. AIDS Patient Care STDS 2011;25:579-585.
91) Arpadi SM, McMahon D, Abrams EJ, Bamji M, Purswani M, Engelson ES, Horlick M, Shane E. Effect of bimonthly supplementation with oral cholecalciferol on serum 25-hydroxyvitamin D concentrations in HI V-infected children and adolescents. Pediatrics 2009; 123: e121-126.
92) Mehta S, Giovannucci E, Mugusi FM, Spiegelman D, Aboud S, Hertzmark E, Msamanga Gl, Hunter D, Fawzi WW. Vitamin D status of HIV-infected women and its association with HIV disease progression, anemia, and mortality. PLoS One 2010; 5: e8770.
93) Cozzolino M, Vidal M, Arcidiacono MV, Tebas P, Yarasheski KE, Dusso AS. HIV protease inhibitors impair vitamin D bioactivation to 1,25-dihydrox-yvitamin D. AIDS 2003; 17: 513-520.
94) Hariparsad N, Nallani SC, Sane RS, Buckley DJ, Buckley AR, Desai PB. Induction of CYP3A4 by efavirenz in primary human hepatocytes: comparison with rifampin and phenobarbital. J Clin Pharmacol 2004; 44: 1273-1281.
95) Brown T, Mc Comsey G. Association between initiation of antiretroviral therapy with efavirenz and decreases in 25-hydroxyvitamin D. Antivir Ther2010; 15: 425-429.
96) Ellfolk M, Norlin M, Gyllensten K, Wikvall K. Regulation of human vitamin D(3) 25-hydroxylases in dermal fibroblasts and prostate cancer LNCaP cells. Mol Pharmacol 2009; 75: 1392-1399.
97) Pasquet A, Viget N, Ajana F, de la Tribonniere X, Dubus S, Paccou J, Legroux-Gerot l, Melliez H, Cortet B, Yazdanpanah Y. Vitamin D deficiency in HIV infected patients: associated with NNRTI or efavirenz use? AIDS 2011; 25: 873-874.
98) Welz T, Childs K, Post FA. Do nevirapine and efavirenz affect vitamin D homeostasis similarly? AIDS 2011;25:875-876.
99) Fux CA, Baumann S, Furrer H, Mueller NJ. Is lower serum 25-hydroxy vitamin D associated with efavirenz or the non-nucleoside reverse transcriptase inhibitor class? AIDS 2011; 25: 876-878.
100) Lattuada E, Lanzafame M, Zoppini G, Concia E, Vento S. No influence of nevirapine on vitamin D deficiency in HIV-infected patients. AIDS Res Hum Retroviruses 2009; 25: 849-850.
101) Wohl D, Doroana M, Orki C, Pilotto JH, Sungkanuparph S, Yeni P, Vanveggel S, Deckx H, Boven K. Change in vitamin D levels smaller, and risk of development of severe vitamin D deficiency lower, among HIV-1-infected, treatment-naive adults receiving TMC278 compared with efavirenz: 48-week results from the Phase III ECHO trial. Abstracts of the 17th Annual Conference of the British HIV Association (BHIVA), Bournemouth, UK, 2011.
102) Fox J, Peters B, Prakash M, Arribas J, Hill A, Moecklinghoff C. Improvement in Vitamin D deficiency following antiretroviral regime change: results from the MONET trial. AIDS Res Hum Retroviruses 2010; 26: 1 -6.
103) Torti C, Mazziotti G, Soldini PA, Foca E, Maroldi R, Gotti D, Carosi G, GiustinaA. High prevalence of radiological vertebral fractures in HIV-infected males. Endocrine 2012; 41: 512-517.
104) Labarga P, Barreiro P, Martin-Carbonero L, Ro-driguez-Novoa S, Solera C, Medrano J, Rivas P, Albalater M, Blanco F, Moreno V, Vispo E, Soriano V. Kidney tubular abnormalities in the absence of impaired glomerular function in HIV patients treated with tenofovir. AIDS 2009; 23: 689-696.
105) Martin A, Bloch M, Amin J, Baker D, Cooper DA, Emery S, Carr A. Simplification of antiretroviral therapy with tenofovir-emtricitabine or abacavir-lamivudine: a randomized, 96-week trial. Clin Infect Dis 2009;49:1591-1601.
106) Gallant JE, Staszewski S, Pozniak AL, DeJesus E, Suleiman JM, Miller MD, Coakley DF, Lu B, Toole JJ, Cheng AK, 903 Study Group. Efficacy and safety of tenofovir DF vs stavudine in combination therapy in antiretroviral naive patients: a 3-year randomized trial. JAMA 2004; 292: 191 -201.
107) Labarga P, Barreiro P, Martin-Carbonero L Alvarez E, Solera C, Pinilla J. Prevalence and mechanisms of hyperparathyroidism in HIV+ patients treated with TdF. Abstracts from the 18th Conference on Retroviruses and Opportunistic Infections Boston, MA, 2011.
108) Rosenvinge MM, Gedela K, Copas AJ, Wilkinson A, Sheehy CA, Bano G, Hay PE, Pakianathan MR, Sadiq ST. Tenofovir-linked hyperparathyroidism is independently associated with the presence of vitamin D deficiency. J Acquir Immune Defic Syndr 2010;54:496-499.
109) Childs KE, Fishman SL, Constable C, Gutierrez JA, Wyatt CM, Dieterich DT, Mullen MP, Branch AD. Short communication: inadequate vitamin D exacerbates parathyroid hormone elevations in tenofovir users. AIDS Res Hum Retroviruses 2010;26:855-899.
110) Pocaterra D, Ricci E, Carenzi L, Schiavini M, Mer-aviglia P, Minisci D. Prevalence of hypovitaminosis D and factors associated in a sample of HIV patients. Abstracts from the 18th International AI DS Conference Vienna, Austria, 2010.
111) Fernandez-Rivera J, Garcia R, Lozano F, Macias J, Garcia-Garcia JA, Mira JA, Corzo JE, G6mez-Ma-teos J, Rueda A, Sanchez-Burson J, Pineda JA. Relationship between low bone mineral density and highly active antiretroviral therapy including protease inhibitors in HIV-infected patients. HIV Clin Trials 2003; 4: 337-346.
112) Duvivier C, Kolta S, Assoumou L, Ghosn J, Rozenberg S, Murphy RL, Katlama C, Costagliola D, ANRS 121 Hippocampe study group. Greater decrease in bone mineral density with protease inhibitor regimens compared with nonnucleoside reverse transcriptase inhibitor regimens in HIV-1 infected naive patients. AIDS 2009; 23: 817-824.
113) Madeddu G, Spanu A, Solinas P, Calia GM, Lovigu C, Chessa F, Mannazzu M, Falchi A, Mura MS, Madeddu G. Bone mass loss and vitamin D metabolism impairment in HIV patients receiving highly active antiretroviral therapy. Q J Nucl Med Mol Imaging 2004; 48: 39-48.
114) Foca E, Motta D, Borderi M, Gotti D, Albini L, Calabresi A, Izzo l, Bellagamba R, Narciso P, Sighi-nolfi L, Clo A, Gibellini D, Quiros-Roldan E, Bri-anese N, Cesana BM, Re MC, Torti C. Prospective evaluation of bone markers, parathormone and 1,25-(OH)2 vitamin D in HIV-positive patients after the initiation of tenofovir/emtricitabine with atazanavir/ritonavir or efavirenz. BMC Infect Dis 2012; 12:38.
115) Tebas P, Umbleja T, Dube M. Initiation of ART is associated with bone loss independent of the specific ART regimen. The results of ACTG A5005s Proceedings of the 14th Conference on Retro-viruses and Opportunistic Infections, Los Angeles, CA, 2007.
116) Grund B, Peng G, Gibert CL, Hoy JF, Isaksson RL, Shlay JC, Martinez E, Reiss P, Visnegarwala F Carr AD, INSIGHT SMART Body Composition Substudy Group. Continuous antiretroviral therapy (ART) decreases bone mineral density. AIDS 2009; 23: 1519-1529.
117) Mondy K, Powderly WG, Claxton SA, Yarasheski KH, Royal M, Stoneman JS, Hoffmann ME, Tebas P. Alendronate, vitamin D, and calcium for the treatment of osteopenia/osteoporosis associated with HIV infection. J Acquir Immune Defic Syndr 2005; 38: 426-431.
118) McComsey GA, Kendall MA, Tebas P, Swindells S, Hogg E, Alston-Smith B, Suckow C, Gopalakrishnan G, Benson C, Wohl DA. Alendronate with calcium and vitamin D supplementation is safe and effective for the treatment of decreased bone mineral density in HIV. AIDS 2007; 21: 2473-2482.
119) Guaraldi G, Orlando G, Madeddu G, Vescini F, Ventura P, Campostrini S, Mura MS, Parise N, Cau-darella R, Esposito R. Alendronate reduces bone resorption in HIV-associated osteopenia/osteoporosis. HIV Clin Trials 2004; 5: 269-277.
120) Triant VA, Lee H, Hadigan C, Grinspoon SK. Increased acute myocardial infarction rates and cardiovascular risk factors among patients with human immunodeficiency virus disease. J Clin Endocrinol Metab 2007; 92: 2506-2512.
121) Currier JS, Taylor A, Boyd F, Dezii CM, Kawabata H, Burtcel B, Maa JF, Hodder S. Coronary heart disease in HIV-infected individuals. J Acquir Immune Defic Syndr 2003; 33: 506-512.
122) Grinspoon SK, Grunfeld C, Kotler DP, Currier JS, Lundgren JD, Dube MP, Lipshultz SE, Hsue PY, Squires K, Schambelan M, Wilson PW, Yarasheski KE, Hadigan CM, Stein JH, Eckel RH. State of the Science Conference Initiative to Decrease Cardiovascular Risk and Increase Quality of Care for Patients Living With HIV/AIDS. Circulation 2008; 118: 198-210.
123) DAD Study Group, Friis-M0ller N, Reiss P, Sabin CA, Weber R, Monforte Ad, El-Sadr W, Thiebaut R, De Wit S, Kirk O, Fontas E, Law MG, Phillips A, Lundgren JD. Class of antiretroviral drugs and the risk of myocardial infarction. N Engl J Med 2007; 356: 1723-1735.
124) Grunfeld C, Delaney JA, Wanke C, Currier JS, Scherzer R, Biggs ML, Tien PC, Shlipak MG, Sidney S, Polak JF, O'Leary D, Bacchetti P, Kronmal RA. Preclinical atherosclerosis due to HIV infection: carotid intima-medial thickness measurements from the FRAM study. AIDS 2009; 23: 1841 -1849.
125) Hsue PY, Hunt PW, SchnellA, Kalapus SC, Hoh R, Ganz P, Martin JN, Deeks SG. Role of viral replication, antiretroviral therapy, and immunodeficiency in HIV-associated atherosclerosis. AIDS 2009; 23: 1059-1067.
126) Crowe SM, Westhorpe C, Mukhamedova N, Ja-worowski A, Sviridov D, Bukrinsky M. The macrophage: the intersection between HIV infection and atherosclerosis. J Leukoc Biol 2010; 87: 589-598.
127) ChoiAI, LoJC, Mulligan K, SchnellA, Kalapus SC, Li Y, Hunt PW, Martin JN, Deeks SG, Hsue PY. Association of vitamin D insufficiency with carotid intima-media thickness in HIV-infected persons. Clin Infect Dis 2011; 52: 941-944.
128) Ross A, Judd SE, Kumari M, Hileman C, Storer N, Labbato D, TangprichaV, McComsey GA. Vitamin D is linked to carotid intima-media thickness and immune reconstitution in HIV-infected individuals. Antivir Ther 2011; 16: 555-563.
129) Shikuma CM, Seto T, Liang CY, Bennett K, DeGrut-tolaV, Gerschenson M, Stein JH, Budoff M, Hodis HN, Delaney JA, Ogata-Arakaki D, Pramyothin P, Chow D. Vitamin D levels and markers of arterial dysfunction in HIV. AIDS Res Hum Retroviruses 2012;28:793-797.
130) Lai H, Gerstenblith G, Fishman EK, Brinke J, Kickler T, Tong W, Bhatia S, Hong T, Chen S, Li J, Detrick B, Lai S. Vitamin D deficiency is associated with silent coronary artery disease in cardiovascular-ly asymptomatic African Americans with HIV infection. Clin Infect Dis 2012; 54: 1747-1755.
131) Tebas P. Insulin resistance and diabetes mellitus associated with antiretroviral use in HIV-infected patients: pathogenesis, prevention, and treatment options. J Acquir Immune Defic Syndr 2008; 49(S2): S86-S92.
132) Brown TT, Cole SR, Li X, Kingsley LA, Palella FJ, Riddler SA, Visscher BR, Margolick JB, Dobs AS. Antiretroviral therapy and the prevalence and incidence of diabetes mellitus in the multicenter AIDS cohort study. Arch Intern Med 2005; 165: 1179-1184.
133) Mitri J, Muraru MD, AG Pittas. Vitamin D and type 2 diabetes: a systematic review. Eur J Clin Nutr 2011; 65:1005-1015.
134) Szep Z, Guaraldi G, Shah SS, Lo Re V 3rd, Rat-cliffe SJ, Orlando G, Carli F Rossi R, Rochira V, Tebas P. Vitamin D deficiency is associated with type 2 diabetes mellitus in HIV infection. AIDS 2011;25:525-529.
135) Maestro B, Campion J, Davila N, Calle C. Stimulation by 1,25-dihydroxyvitamin D3 of insulin receptor expression and insulin responsiveness for glucose transport in U-937 human promonocytic cells. Endocr J 2000; 47: 383-391.
136) Norman AW, Frankel JB, Heldt AM, Grodsky GM. Vitamin D deficiency inhibits pancreatic secretion of insulin. Science 1980; 209(4458): 823-825.
137) Chertow BS, Sivitz Wl, Baranetsky NG, Clark SA, Waite A, Deluca HF. Cellular mechanisms of insulin release: the effects of vitamin D deficiency and repletion on rat insulin secretion. Endocrinology 1983; 113: 1511-1518.
138) Pittas AG, Harris SS, Stark PC, Dawson-Hughes B. The effects of calcium and vitamin D supplementation on blood glucose and markers of inflammation in non-diabetic adults. Diabetes Care 2007; 30: 980-986.
139) Van den Bout-van den Beukel CJ, Van den Bos M, Oyen WJ, Hermus AR, Sweep FC, Tack CJ, Bosch ME, Burger DM, Koopmans PP, van der Ven AJ. The effect of cholecalciferol supplementation on vitamin D levels and insulin sensitivity is dose related in vitamin D deficient HIV-1-infected patients. HIV Med 2008; 9: 771-779.
140) Hida Y Kawada T, Kayahashi S, Ishihara T, Fushiki T. Counteraction of retinoic acid and 1,25-dihydrox-yvitamin D3 on up-regulation of adipocyte differentiation with PPARgamma ligand, an antidiabetic thiazolidinedione, in 3T3-L1 cells. Life Sci 1998; 62: L205-211.
141) Blumberg JM, Tzameli l, Astapova l, Lam FS, Flier JS, Hollenberg AN. Complex role of the vitamin D receptor and its ligand in adipogenesis in 3T3-L1 cells. J Biol Chem 2006; 281: 11205-11213.
142) Kong J, Li YC. Molecular mechanism of 1,25-di-hydroxyvitamin D3 inhibition of adipogenesis in 3T3-L1 cells. Am J Physiol Endocrinol Metab 2006; 290: E916-924.
143) Evans RM, Barish GD, Wang YX. PPARs and the complex journey to obesity. Nat Med 2004; 10: 355-361.
144) Ansemant T, Mahy S, Piroth C, Ornetti P, Ewing S, 149) Guilland JC, Croisier D, Duvillard L Chavanet P, Maillefert JF, Piroth L. Severe hypovitaminosis D correlates with increased inflammatory markers in HIV infected patients. BMC Infectious Diseases 2013; 13: 7.
145) Poudel-Tandukar K, Poudel KC, Jimba M, Kobayashi 150) J, Johnson CA, Palmer PH. Serum 25-hydroxyvitamin D levels and C-reactive protein in persons with human immunodeficiency virus infection. AIDS Res Hum Retroviruses [Epub ahead of print]
146) European AIDS Clinical Society. Guidelines: prevention and management of non-infectious co-morbidities in HIV. Available at http://www.euro-peanaidsclinicalsociety.org/images/stories/EACS -Pdf/EacsGuidelines-v6.1-2edition.pdf. Ac- 151) Acessed January 2013.
147) Heaney RP Davies KM, Chen TC, Holick MF, Barger-Lux MJ. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am J Clin Nutr 2003; 77:204-210.
148) Havens PL Mulligan K, Hazra R, Flynn P, Rutledge B, Van Loan MD, Lujan-Zilbermann J, Kapogiannis 152) BG, Wilson CM, Stephensen CB, and the Adolescent Medicine Trials Network for HIV/AlDS Interventions (ATN) 063 Study Team. Serum 25-Hy-droxyvitamin D response to vitamin D3 supplementation 50,000 IU monthly in youth with HIV-1 infection. J Clin Endocrinol Metab 2012; 97: 4004-4013.
Childs K, Fishman S, Factor S, Dieterich D, Mullen M, Branch A. First report of dose/response data of HIV-infected men treated with vitamin D3 Supplement. 17th Conference on Retroviruses and Opportunistic Infections San Francisco, California, 2010.
Havens P, Hazra R, Stephensen C, van Loan M, Rut-ledge B, Bethel J, Pan C, Liu N, Lujan-Zilbermann J, Mulligan K, and ATN 063 Study Team. Vitamin D3 supplementation decreases PTH in HIV-in-fected youth being treated with TDF-containing combination ART: a randomized, double-blind, placebo-controlled multicenter trial: Adolescent Trials Network Study 063. Abstracts from the 18th Conference on Retroviruses and Opportunistic Infections Boston, MA, 2011.
Buccisano F Rossi FM, Venditti A, Del Poeta G, Cox MC, Abbruzzese E, Rupolo M, Berretta M, De-gan M, Russo S, Tamburini A, Maurillo L, Del Principe Ml, Postorino M, Amadori S, Gattei V. CD90/Thy-1 is preferentially expressed on blast cells of high risk acute myeloid leukaemias. Br J Haematol 2004; 125: 203-212.
Longenecker C, Hileman C, Carman C, Ross AA, Seydafkan S, Brown TT, Labbato DE, Storer N, Tangpricha V, Mccomsey GA. Vitamin D supplementation and endothelial function among vitamin D-deficient HIV-infected persons: a randomized placebo-controlled trial. Abstracts from the 18th Conference on Retroviruses and Opportunistic Infections Boston, MA, 2011.
Another HIV and Vitamin D paper a few months later, behind a paywall
Vitamin D and HIV infection: a review of the clinical evidence
Future Virology, June 2013, Vol. 8, No. 6, Pages 589-606 , DOI 10.2217/fvl.13.40 (doi:10.2217/fvl.13.40)
Ali Tafazoli1 & Hossein Khalili *2
The positive effects of vitamin D in different acute and chronic diseases (e.g., bone and renal disorders, acute and chronic respiratory tract infections, and diabetes mellitus), and regulation of immune system function have been shown. In this review vitamin D status and the effects of its supplementation alone or in combination with other bone-modifying substances like calcium and bisphosphonates on the different aspects of human health have been investigated in HIV+ individuals. Three scientific electronic databases have been investigated for extracting related articles.
Searching only PubMed yielded 59 results with ‘HIV OR AIDS’ and ‘Vitamin D’ keywords. Because many of the studies in this field are observational or cross-sectional, designing comprehensive and eligible randomized clinical trials has been recommended by several authors in order to develop evidence-based clinical practice guidelines to determine the best regimen of vitamin D supplementation in HIV-infected patients.
