Boron is probably important to human health – 2003
The Physiological Effects of Dietary Boron
Critical Reviews in Food Science and Nutrition, 43(2):219–231 (2003)
Tara A. Devirian and Stella L. Volpe*
Department of Nutrition, University of Massachusetts, Amherst
Referee: Dr. Susan Meacham, Associate Professor and Department Head, University of Nevada, Las Vegas, College of Health
Sciences, Room 329, Rod Lee Bigelow Building, 4505 Maryland Parkway, Las Vegas, NV 89154-3019
- Please address all correspondence to: Stella L. Volpe, Ph.D., R.D., FACSM, University of Massachusetts, Department of Nutrition, 210 Chenoweth Lab, 100 Holdsworth Way, Amherst, MA 01003. Phone: (413) 545-1076. Fax: (413) 545-1074. Email: volpe@nutrition.umass.edu
📄 Download the PDF from VitaminDWiki
ABSTRACT:
Boron may be an essential nutrient for animals and humans. Dietary boron influences the activity of many metabolic enzymes, as well as the metabolism of steroid hormones and several micronutrients, including calcium, magnesium, and vitamin D. Boron supplementation in rats and chicks has been shown to increase bone strength. Boron may also play a role in improving arthritis, plasma lipid profiles, and brain function. Additional research is necessary to further clarify boron’s influence in human and animal physiology, as well as determine a dietary requirement for humans.
INTRODUCTION
Boron was accepted in 1923 as an essential nutrient for all vascular plants.1 Since then, evidence has been accumulating that boron may be required by animals and humans.2 Boron is known to influence a variety of metabolic actions. It interacts with calcium, vitamin D, and magnesium, which are all important in bone metabolism. Boron accumulates in bone in concentrations dependent on the amount of boron consumed.3 Boron has also been found to increase steroid hormone concentrations in postmenopausal women4 and to have antioxidant properties,5 which could make it beneficial in preventing atherosclerosis. Boron is abundant in nature as boric acid and borate and can be obtained in the diet through the consumption of fruits, vegetables, and legumes.6 This review article discusses past research that shows the nutritional importance of dietary boron in both animals and humans.
but contains characteristics between those of metals and nonmetals.7 A small atom with three electrons in its outer shell, boron uses covalent bonding to fill its outer shell. The boron atom does not donate protons, but acts as a Lewis acid by accepting hydroxyl ions.7 Boron binds with several elements, resulting in either a tetrahedral or trigonal-planar arrangement. It has a strong attraction to oxygen, resulting in borates and other oxo compounds. When boron binds to four oxygen atoms, the resulting compound in the tetrahedral arrangement is the borate anion (B(OH)4-).8 When boron binds to three oxygen atoms, the resulting compound in the trigonal-planar arrangement is orthoboric acid (B(OH)3).8
Boron has a strong tendency to form complexes with organic molecules that have adjacent hydroxyl groups. Boron is able to interact with important biological substances, including polysaccharides, pyridoxine, riboflavin, dehy- droascorbic acid, and the pyridine nucleotides.9 It binds strongly to furanoid cis-diols, which include erythritan, ribose, and apiose; apiose is present throughout the cell walls of vascular plants.8 Nicotinamide adenine dinucleotide (NAD+) and nicotinamide adenine dinucleotide
phosphate (NADP) contain ribose components; because these molecules are active in energy metabolism, binding to them would affect the processes of certain metabolic pathways.10 Boron’s unique chemistry allows it to react with many other metabolites and enzymes, and thus may be capable of modifying mineral and energy metabolism in humans and animals.2
BORON IN THE ENVIRONMENT
Boron is found commonly in nature and occurs naturally in the form of borates.11 Boron occurs in high concentrations in sedimentary rocks, seawater, coals, and soils.9 The global average concentration of seawater is estimated to be around 4.6 mg/L.9 Deposits with plentiful boron exist in California, the high Andes, and the Alpine Himalayas.11
Boron is released into the atmosphere from commercial use and from oceans, rock weathering, volcanos, coal combustion, and forest fires.11 The glass, ceramic, detergent, and fertilizer industries use boron for commercial applications, leading to a large release of boron into the environment.12 The amount of available boron in soils varies with the soil type, fertilizers used, pH, and temperature. The application of superphosphate and potash fertilizers inhibit the absorption of boron.13 Warm, moist soil has highly available boron due to the activity of microflora.13 Sixty-five to 85% of boron in the atmosphere is derived from oceans, and another 7 to 18% from the major “anthropogenic” sources, which include boron-containing irrigation water and fertilizers, fly ash containing boron from coal-fired power plants, and from wastewater treatment plant releases.11 About 1.8 to 5.3 billion kg of boron is released into the atmosphere each year.12
Hunt et al.14 examined boron concentrations in personal care products and found moderate amounts in estrogen supplements (0.812 ug/g) and very high concentrations in gastric antacids (34.7 ug/g). Laxatives had low to exceptionally high levels (0.323 to 52.0 |ug/g).14 Lipsticks, some skin creams, and shampoos had from high to exceptionally high amounts (1.23 to 59.6 ug/g), while soaps and douches contained negligible concentrations (< 0.015 ug/g).14
BORON LEVELS IN HUMAN TISSUES
Due to their high solubility, borates are found in animal and plant tissues, although they are more concentrated in plant than in animal tissues.12 Their concentration varies from less than 0.07 mg/kg in animal livers to 248 mg/kg in certain seaweeds.12 The average amount of boron in plants and animals is estimated to be between 30 to 50 ppm.12
Boron is mostly present in body tissues and fluids as boric acid (B(OH)2).6 Boron exists in human blood as 98.4% boric acid and 1.6% as the borate anion (B(OH)4);6 total blood boron concentrations range from 15.3 to 79.5 ng/g of wet weight.15
Boron content differs among the various bodily organs, which suggests that boron carries out particular functions in each organ.16 Table 117,18,19 lists the total amount of boron in mg/organ or tissue based on two human cadavers . It is estimated that the total boron content in the human body ranges from 3 to 20 mg, with the highest concentrations found in bone, fingernails, toenails, and hair (4.3 to 17.9 ppm).20 Research in 50 healthy humans showed mean boron concentrations in whole blood, serum, and urine to be 0.06, 0.02, and 0.75 ppm, respectively.21 In addition, boron content differs in arthritic and healthy bone: there is about 3 ppm boron in arthritic bones, compared with 56 ppm boron in healthy bones. 16
Boron accumulates in bone but not in soft tissues.6 Studies with male rats consuming high boron diets (9000 ppm/day) indicated that soft tissue boron concentration plateaued at about 15 ppm, while that of bone increased to 47 ppm without reaching a plateau.20
BORON TOXICITY
Boric acid is toxic only when consumed in excessive amounts, because animals and humans excrete excess boron.22 Although an adult dose of 18 to 20 mg of boron has been shown to be fatal,22
TABLE 1 Boron Concentration per Organ or Tissue (ppm Boron/Organ or Tissue)
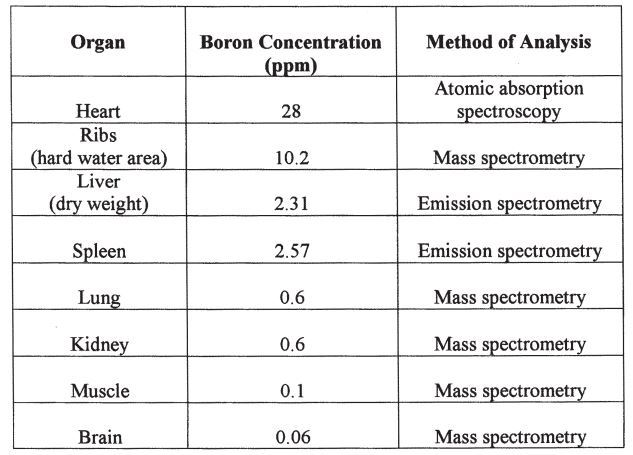
Organ death from boron toxicity is unusual, and some adults have tolerated 80 to 297 g.23 It is unlikely that a single ingestion of boron will be fatal; however, death may occur several days after ingestion from renal injury, circulatory collapse, or shock.23 The process by which boron causes death remains unclear.23
Boron, boric acid, and boron oxide are primarily irritants under exposure conditions.23 The clinical symptoms of boron toxicity are similar to pellagra symptoms. Biochemical symptoms of toxicity include riboflavinuria and riboflavin deficiency, along with the inhibition of the dehydrogenase enzymes.22 Toxic ingestions of boron may cause nausea, vomiting, and diarrhea. The vomi- tus and feces may also appear blue-green in color,23 and renal injury, hypothermia, erythema, restlessness, and weariness may occur.22 Chronic boron toxicity results in poor appetite, nausea, weight loss, and decreased seminal volume and sexual activity.22 Dogs and rats fed 1170 to 2000 ppm boron for 2 years showed stunted growth, less productive food use, skin rashes, and gonadal deterioration.23 Chronic exposures can lead to a high death rate in infants and children.23
BORON DEFICIENCY
Boron deficiency symptoms have been seen in rats, chickens, and humans.24 Boron deprivation in animals leads to impaired growth and abnormal bone development.7 Deprivation in humans and animals causes increased urinary calcium excretion.9
Other boron deficiency symptoms may result from an altered macromineral status. For example, boron deficiency exacerbates the signs of vitamin D3 deficiency (e.g., abnormal bone formation and poor growth).2 A low-magnesium diet exacerbated the deficiency symptoms of boron in rats, resulting in reduced growth and enlarged kidney and spleen as determined by ratio of organ to body weight.25
ABSORPTION AND EXCRETION OF BORON
Boron is accessible to all plants and animals.26 Boron is easily absorbed across the gastrointestinal epithelia in humans and animals,26 and across mucous membranes, such as the mouth, eyes, vagina, and anus. Hunt10 reported that humans and animals absorb nearly 100% of supplemental inorganic boron. Some organic forms of supplemental boron may be inaccessible to animals because plants can only absorb organic forms of boron in soils after mineralization.10
Boron is primarily excreted in the urine, with about 2% lost in the feces, and lesser amounts lost in bile, sweat, and breath.9,15 Tissue boron concentrations are generally kept steady by a homeostatic mechanism, primarily through renal excretion, and higher boron intakes do not significantly increase plasma levels.6 A 167-day metabolic study of 11 postmenopausal women showed a rapid increase in urinary boron when boron intake increased from 0.36 mg/day to 3.22 mg/day.29 Naghii and Samman30 studied the effect of boron supplementation on urinary excretion in healthy male subjects. When 18 healthy males remained on a habitual diet, urinary boron excretion measured on two separate occasions ranged from 0.3 to 3.53 mg/day. The difference in boron values between the two 24-h urinary collections was not statistically significant, but slight variations within and between some subjects suggested differences in their daily boron consumption. In a second study, when subjects were administered 10 mg/day of supplementary boron for 4 weeks, urinary boron increased from an average of 1.64 ± 0.30 (at baseline) to 10.16 ± 0.92 mg/day. This increase in urinary excretion, which occurred in every individual, was significant (p < 0.0001) and represented 84% of the supplemented dose. These findings provide evidence that urinary boron mirrors boron intake.
Daily boron intakes differ according to individual food selection, the amount obtained from personal care products, and the amount of boron in the drinking water.14 While the average daily boron intake in Finland was estimated to be 1.7 mg,32 boron intakes in the U.S. generally are between 1.7 to 7 mg/day.4 When Samman et al.9 analyzed 7-day food records, daily boron intake for males and females in Australia averaged 2.28 ± 1.3 mg/ day and 2.16 ± 1.1 mg/day, respectively. Boron consumption was higher when subjects ate foods with more energy, fiber, and plant protein.9 Diets containing a variety of foods should provide between 1.5 to 3.0 mg/day.2 Estimating boron intake, however, may best be accomplished by examining urinary excretion.
DIETARY SOURCES OF BORON
The primary sources of boron are fruits and vegetables. Most fruits, seeds, stalks, or barks have high concentrations of boron except for citrus fruits, berries, and pineapple.14 Of the vegetables, leafy greens have the highest amount of boron, especially when grown without chemical fertilizers .13 In addition, fruits, vegetables, tubers, and legumes have much greater amounts of boron than the grasses (corn, rice, and wheat) (Table 2).2,26 Dried fruits, legumes, nuts and avocado contain from 1.0 mg to 4.5 mg boron/100 g.7 Fresh fruits, vegetables, honey, and bee pollen contain from 0.1 to 0.6 mg boron/100 g, whereas foods from animal sources provide between 0.01 and 0.06 mg/100 g.7,13 Other major sources of boron are drinking water and commercial fertilizers. The amount of boron in water differs according to location, containing anywhere from negligible to high amounts.14
ESSENTIALITY OF BORON IN PLANTS AND ANIMALS
Boron is needed for plant growth and cell division, although plant scientists have yet to determine the exact mechanism by which it functions. Boron plays a role in carbohydrate metabolism,
TABLE 2 Boron Content in Selected Foods
hormone action, and nucleic acid synthesis11 and is known to enhance RNA production in plants.33 It is also involved with the structure and function of membranes, as well as the functioning of the plant’s reproductive tissue.11 Boron plays a role in cell wall cross-linking and binds to particular fibrous constituents of the cell wall.11
Various plants require different boron levels for best growth. Boron deficiency is much more common in nature than boron toxicity. Among plants, the deficiency of boron is the most common deficiency for any micronutrient . It is common in light-textured, acidic soils, in which boron can easily be leached.11 It is possible that deficiency in plants may be due to alterations in membrane permeability.24
The number of species in which the essentiality of boron is observed has been increasing in the last decade, providing additional evidence that boron may be an essential nutrient for humans.34 Fort et al.35 studied the effects of inadequate boron intakes in adult male frogs, and found that low dietary boron hindered normal Xenopus development during organ formation. It also interfered with normal reproductive function in adult frogs. In frogs fed a low boron diet, the death rate and number of developmental malformations in offspring were significantly higher than for frogs fed a normal amount of boron.35
HUMAN BORON REQUIREMENT
Past research has shown that boron may be an essential dietary component for animals and humans. Hunt10 claims that boron meets most of the criteria for a necessary nutrient. It has a light atomic weight and can bind to organic compounds in ways that influence biological function.10 Boron is ubiquitous in nature; it exists abundantly in the earth, in seawater, and in animals. It is generally nontoxic, and animals have a homeostatic control for maintaining boron concentrations within certain limits.10 Low levels of dietary boron among certain animals have been observed to result in developmental abnormalities. Studies on humans have not demonstrated that low boron intakes cause developmental abnormalities, but there is evidence that dietary boron benefits human health .10
A dietary requirement for boron among humans has yet to be determined. Before a requirement can be determined, additional studies are required on the biological function of boron and its homeostatic regulation.6 It has been determined that chicks require close to 1 ppm/day of dietary boron; thus, it is estimated that humans need at least 0.5 mg/day.2 In chicks, a boron intake of 0.3 to 0.4 mg/day led to beneficial alterations in mineral metabolism. Therefore, Nielsen2 suggests that humans need at least 0.2 mg boron/day, and a dietary requirement could possibly be near 1 to 2 mg/day. The Food and Nutrition Board of the Institute of Medicine has recently established a Tolerable Upper Intake Level (UL) for boron of 20 mg/day for adults over 18 years of age.36
BORON AND ENZYMATIC ACTIVITY
Hunt10 reported that boron influences the activity of at least 26 different enzymes seen in animal, plant, cultured, and chemical reaction systems. Boron plays a role in regulating enzymatic activity in pathways involved in energy substrate metabolism, insulin release, and the immune system. Diets low in boron (< 0.3 mg/g) supplemented with about 2 ug/g appear to be sufficient to modify “several aspects of animal and human physiology”.10 The mechanisms by which it binds to enzymes or to cofactors, such as NAD+, are not presently known and must be investigated further.10 One hypothesis is that boron alters metabolic reactions by binding to substrate compounds.7
Boron may be essential for controlling certain pathways that use serine proteases or oxidoreductases. These enzymes require pyridine or flavin nucleotides (NAD+, NADP, or FAD), and boron reversibly inhibits their activity by forming transition state analogs and by competing for NAD or FAD.10 Serine proteases, such as thrombin, function in regulating blood and coagulation sys- tems.10 Other enzymes, such as phosphoglucomutase, u-glutamyl transpeptidase (GGT), and glyceraldehyde-3-phosphate dehydrogenase (GPD) are also inhibited by boron.10
Boron inhibits glycolytic enzyme activity in vitro and may control energy substrate use.10 It modifies insulin release by altering the metabolism of NADPH. Changes in NADPH metabolism cause changes in cell membrane dynamics, leading to insulin secretion.10 Boron inhibits the activity of glucose-6-phosphate dehydrogenase and GPD, which are two key enzymes in the Pentose Phosphate Pathway (PPP).10 Insulin release is enhanced during boron deficiency conditions due to increased activity of the PPP, suggesting that boron modifies ion transport across the cell membrane.10
The level of NADPH may be modified due to boron’s inhibitory effect on enzymes in the PPP, decreasing the demand for oxygen. Boron may also lower the level of oxidative damage, which is accomplished by decreasing the production of NADPH and the activity of λglutamyl transpeptidase.10 This action could possibly increase the amount of glutathione (GSH) in the body,10 which plays a role in protecting cells from toxic oxygen radicals.37
BORON AND MICRONUTRIENT METABOLISM
Boron plays a regulatory role in the metabolism of several micronutrients, such as
calcium,
phosphorus,
aluminum,
magnesium, and
molybdenum.27
The micronutrients of particular interest with regard to bone health are magnesium, vitamin D, phosphorus, and calcium . Boron is observed to positively influence the metabolism of these dietary substances, which all play a role in maintaining bone health. As a result, boron may be nutritionally significant in preventing osteoporosis in humans .33 Boron’s likely role in the maintenance of bones is most obvious during magnesium deficiency, or other states in which there is an increase in urinary calcium.33
Magnesium Metabolism
Hunt and Nielsen38 studied magnesium-deficient chicks and found that supplemental boron decreased the level of abnormalities caused by inadequate magnesium intake. Subsequent boron supplementation enhanced growth and increased plasma calcium and magnesium concentrations, as well as inhibited the calcification of cartilage.39 Nielsen et al.25 found that in magnesium-deficient rats, inadequate dietary boron led to more severe symptoms of magnesium deficiency, and signs of boron deprivation were of greater significance when the diet was low in both magnesium and methionine.25
Vitamin D Metabolism
Hunt and Nielsen40 found a possible interaction between boron and vitamin D in chicks. When vitamin D-deficient chicks consumed basal diets containing 1 mg/kg boron, boron supplementation of up to 3 mg/kg significantly enhanced growth by 38% , increased plasma ionized calcium concentrations, lowered plasma alkaline phosphatase levels, and decreased the number of ricket cases.40 This study found that boron deprivation depressed growth; the effect was most evident when vitamin D intake was low.40 When chicks were fed adequate levels of vitamin D, boron supplementation improved growth rate by 11% .40 Hunt and Nielsen38 suggested an indirect effect of boron on vitamin D metabolism through an alteration in calcium, phosphorus, or magnesium metabolism.
Hunt et al.41 studied day-old cockerel chicks and found that dietary boron decreased the adverse effects of vitamin D deficiency, which included increased plasma glucose, triglyceride, and cholesterol levels. Chicks fed inadequate vitamin D for 26 days displayed decreased food utilization and plasma calcium levels, as well as increased plasma glucose, beta-hydroxybutyrate, triglyceride, triiodothyronine, cholesterol, and plasma alkaline phosphatase concentrations. Supplementation with 1.4 mg/kg boron as orthoboric acid decreased the elevated levels of plasma glucose and triglyceride levels to similar concentrations seen in the chicks fed adequate vitamin D levels.41 Boron supplementation at physiological amounts also increased food utilization and growth plate maturation in all chicks. In chicks fed adequate vitamin D, the incidence of rachitic long bones was greater in boron-deficient chicks compared with boron-supplemented chicks.41 Thus, it appears that boron plays a significant role in normalizing the disturbed energy substrate utilization caused by vitamin D deficiency, as well as improving the macromineral content of bone.41
Conflicting results were obtained by Elliot and Edwards42 when studying the effect of dietary boron on broiler chickens. The addition of boron at 5 to 20 mg/kg, 20 to 80 mg/kg, and 40 mg/kg to basal diets containing 9 to 14 mg/kg boron did not increase growth rate or food consumption. In subsequent experiments, chickens were fed basal diets containing 1 mg/kg boron and were supplemented with 3 mg/kg boron (a supplementation level comparable to that used by Hunt and Nielsen40). Boron supplementation at these lower levels also failed to increase growth rate or food consumption. However, boron supplementation of 3 mg/kg as boric acid tended to increase weight gain in chicks fed insufficient vitamin D (110 IU/ kg) for 16 days.42 Supplementation also tended to decrease weight gain in chicks fed adequate vitamin D (1100 IU/kg) for 16 days.42 Food consumption was not affected by these dietary modi- fications.42
Hunt and Nielsen38 also studied the outcomes of boron, magnesium, and molybdenum interactions on bone structure in vitamin D-deficient chicks. Day-old chicks were fed a basal diet of 125 IU of vitamin D (inadequate), 0.420 ug molybdenum/kg, 0.465 ug boron/kg, and a calcium:phosphorus ratio of 1:0.56. Supplementation included boric acid at 0 or 3 mg/kg, magnesium acetate at 300 (inadequate) or 500 mg/kg (adequate), and ammonium molybdenate at 0 or 20 mg/kg. After 28 days, dietary boron increased growth in chicks fed inadequate magnesium. These findings indicated a boron-vitamin D interaction, and led to the hypothesis that boron may play a role in either hydroxylating or extending the half-life of vitamin D3, based on its ability to complex with hydroxyl groups on organic compounds.38 This role would most likely affect bone metabolism and improve bone strength.
Calcium Metabolism
Nielsen33 suggested that boron also plays a role in calcium metabolism, because higher dietary levels of boron have been shown to decrease calcium excretion and increase plasma ionized calcium levels.33 There is considerable evidence that large intakes of calcium alone do not prevent bone loss.43 Based on this evidence, Nielsen et al.4 researched the influence of certain nutrients on major mineral metabolism other than calcium, vitamin D, and flouride. Thirteen postmenopausal women were given a basal diet supplying low amounts of boron (0.25 mg/day) for 23 days and supplemented with 200 mg of magnesium/day. All women also participated in four dietary periods of 24 days, which included: basal diet only; basal diet supplemented with 1000 mg aluminum as aluminum hydroxide/ day; basal diet supplemented with 200 mg magnesium as magnesium gluconate/day; and basal diet supplemented with 1000 mg of aluminum and 200 mg of magnesium/day. By the end of these four periods, all women had consumed a diet low in boron for 119 days. Twelve women then consumed the basal diet supplemented with 3 mg of boron/ day as sodium borate for two additional 24-day dietary periods. Seven of the women were fed a diet low in magnesium for the full 48 days. The other five were given 200 mg magnesium/day for the first period followed by 200 mg magnesium plus 1000 mg aluminum/day for the second period. Results showed that dietary boron had a significant effect on macromineral metabolism in postmenopausal women. The effect was altered by magnesium intake, but not by dietary aluminum. Supplementation with 3 mg boron/day significantly lowered urinary calcium and magnesium excretion in postmenopausal women ; the differences in excretion were more marked in the women fed inadequate levels of magnesium than in the women fed adequate levels of magnesium. Results indicated that magnesium status significantly influences the action of dietary boron, and that dietary boron causes changes, which favor an increase in plasma calcium levels and bone mineralization.4
BORON AND BONE STRENGTH
. Animal Studies
In addition to its interaction with other nutrients, supplemental boron as boric acid has been shown to increase bone strength measures in rats.3 Boric acid was added to the diets of 48 rats (42 males and 6 females) in doses of 200, 1000, 3000, or 9000 ppm/ day. The levels consumed were < 0.2 (control), 1.7, 8.5, 26, and 68 mg of boron/kg/day. Bone strength was measured by bending the tibia and femora and crushing the first four lumbar vertebrae, at 1, 2, 3, 4, 5, 8, and 12 weeks after consuming the diet. Significant reductions were observed in serum calcium, phosphorus, and magnesium concentrations compared with the controls, which was attributed to boron intake. However, lack of urinary and bone measurements of these elements do not indicate whether there was increased excretion or movement into bone. Results also showed a 10% increase in vertebral crush resistance at all dose levels. There was no dose-response of the vertebral crush resistance, which may have been due to the saturation of boron at all “pertinent sites” in bone at all dose levels used in the study.3 Although boric acid consumption tended to increase crush resistance of the vertebrae, it showed no effect on the ability of the tibia or femora to resist a bending force.3
Wilson and Ruszler44 found that supplemental boron increased measures of long bone strength in young hens. Supplementation with 50 and 100 mg boron/kg from day one until 16 weeks of age significantly increased the shear force and shear stress of the tibia in the hens and increased the shear fracture energy of the femur. Supplementation with 50, 100, and 200 mg boron/kg for 16 weeks increased the amount of tibial bone ash, with the greatest increase seen at 50 mg/kg.44
The effects of boron on tibial and femoral strength characteristics were also determined in hens fed supplemental boron since birth.45 From day one, all hens were fed a basal diet containing 3 mg/kg of boron. At 18 weeks of age, the hens were supplemented with 50, 100, 200, and 400 mg/kg daily of boric acid. Other hens were supplemented with 200 mg/kg of boron starting at 16 and 32 weeks of age. When bone strength measures were taken at 72 weeks of age, it was found that boron supplementation at 200 mg/kg significantly strengthened the shear fracture energy of the tibia and radius in hens started at 32 weeks on supplemental boron.45 Compared with the control group, the values for the shear fracture energy were 45% higher in the tibia and 47% higher in the radius for these hens. Thus, dietary boron may decrease bone brittleness in hens at levels of 200 mg/kg.45
Human Studies
Results from human studies have also shown that dietary boron may be useful in preventing osteoporosis . When 12 postmenopausal women consumed a low boron diet for 17 weeks, then took a daily supplement of 3 mg of boron for 7 more weeks, serum electrolyte concentration and steroid hormone balance both changed.4 Within 8 days of beginning the boron supplement action, urinary calcium and urinary magnesium excretion decreased by 40% and 33% , respectively.4 Calcium and magnesium losses were lower after taking the supplement than before the study. These findings are significant, because calcium and magnesium are necessary elements for maximum bone health.4
Nielsen33 found that dietary boron had the same effect as estrogen supplementation, based on a study with 15 human subjects. Boron satiety increased serum ionized calcium, lowered serum calcitonin concentrations, and increased the serum levels of 1,25-dihydroxycholecalciferol. The effects indicated that dietary boron might benefit calcium metabolism and help prevent bone deterioration. The fact that adequate dietary levels of boron decreased serum calcitonin levels is important, because calcitonin has been shown to increase calcium loss in humans, and serum calcitonin concentrations are higher in women with postmenopausal osteoporosis.33
To assess if boron had an impact in premenopausal women, Volpe et al.46 examined boron’s effect on bone mineral density in female college athletes. Twenty-eight athletic and nonathletic females took either a daily tri-boron supplement of 3 mg or a placebo for 10 months. Results indicated that dietary boron had no effect on bone mineral density. In menstruating females, boron supplementation did not increase steroid hormone concentrations, or did it stimulate bone calcification.46 Volpe et al.46 suggested that the dietary intake of calcium, magnesium, or vitamin D may need to be lower in order for dietary boron to benefit bone mineral density.
The mechanism by which boron acts has not yet been established. Present data give evidence that boron alters calcium levels by different mechanisms. Boron’s influence on vitamin D and magnesium metabolism (and thus calcium homeostasis) may be controlled by its influence on cell membrane activity.9 What is known is that large amounts of dietary boron benefit calcium and vitamin D status in humans and animals.7
BORON AND STEROID HORMONES
A number of studies have demonstrated that boron influences steroid hormone metabolism,4,5,9 ,30,47 and support the hypothesis that boron is necessary for the hydroxylation step in the formation of specific steroid hormones.47 Boron’s chemistry allows the mineral to complex with organic compounds containing hydroxyl groups, and the formation of testosterone and 17-P estradiol from precursors involves adding hydroxyl groups to a steroid ring. Both 17-P estradiol and testosterone significantly increased in postmenopausal women consuming 3 mg boron/day in the form of sodium borate for 7 weeks.4 Boron supplementation resulted in a twofold increase in 17-P estradiol levels, a greater than twofold increase in testosterone concentrations (from 0.31 to 0.83 ng/mL), and a significant increase in calcium retention in postmenopausal women.4 Men given 10 mg boron/ day in the form of sodium tetraborate for 4 weeks had significant increases in 17-P estradiol levels and tended toward increased plasma testosterone concentrations.30
An increase in steroid hormone levels has also been demonstrated in animals. Rats that consumed an equivalent of 2 mg boron/day in the form of boric acid had significant increases in plasma testosterone levels after 4 weeks.47 In the same study, plasma levels of 1,25-dihydroxyvitamin D were also measured in order to test the specificity of the action of boron on steroid hormones;47 dietary boron led to a marked increase in plasma 1,25- dihydroxyvitamin D concentrations. The increased levels of two unrelated steroid hormones support the hypothesis that boron enhances the hydroxyla- tion of steroid rings.47
Other reports contradict boron’s effects on plasma steroid hormone concentrations in humans. Ferrando and Green28 studied the effects of boron supplementation in 19 male body builders, ages 20 to 27 years. After 7 weeks of supplementation with 2.5 mg boron/day, no significant effects of boron were observed on plasma total or free testosterone concentrations, plasma boron levels, lean body mass, or strength measurements.28 Volpe et al.46 assessed the effect of boron supplementation in 28 females, 18 to 24 years of age. After 10 months of consuming 3 mg boron/day in the form of tri-boron, no significant changes in plasma 17-P estradiol, progesterone, testosterone, or 1,25-dihydroxyvitamin D levels were observed.46 It is possible that boron plays a role in steroid hormone and mineral metabolism when either vitamin D, magnesium, or both are deficient in
the diet.24
EFFECT ON ARTHRITIS
Because boron can complex with hydroxyl groups on organic compounds, it can form corticosteroids , and thus alleviate symptoms of rheumatoid arthritis .2 In areas of Australia where water and soil levels of boron are high, half the cases of musculoskeletal diseases have been reported when compared with areas containing lower boron levels in water and soil.16 A comparison between countries indicated that the occurrence of arthritis is negatively correlated with the level of boron in the soil, and subsequently in the food supply.16 In addition, arthritic bones are found to contain less boron than healthy bones.16
One study48 showed that high levels of dietary boron (20 ug/g) compared with very low levels (< 0.2 ug/g) postpones the onset and lessens the severity of arthritis in rats. Between 0 and 20 ug/mL boron in vitro prevented the growth of splenic cells taken from boron-deficient rats, which led to the hypothesis that boron may alleviate adjuvant-induced arthritis by preventing T-cell activity.48 A subsequent report stated that 3 ug/g boron added to a low boron diet (0.2 ug/g) more than doubled serum antibody levels.49 Further distinctions need to be made on how boron regulates the inflammatory response, but it is possible that boron helps alleviate arthritis by restricting T-cell activity and modifying the levels of serum antibodies.10
BORON AND PLASMA LIPIDS
Dietary boron may play a role in lowering plasma lipid levels. The administration of two boron-derived hypolipidemic agents (Tetrakis-u-fe- dicopper (II) and AA-dimethyl-n-octadecylamine borane) to rats significantly lowered serum low- density lipoprotein (LDL) cholesterol and triglyceride levels after 14 days.50 In addition, both drugs inhibited LDL binding and entrance into liver cells, fibroblasts, and aorta cells, and promoted high- density lipoprotein (HDL) binding and deterioration in liver cells. Results from this study indicate that boron-containing drugs may be of therapeutic benefit to atherosclerosis, because the compounds promoted cholesterol removal from tissues and decreased lipid accumulation.50
Nearly a decade later, Naghii and Samman30,47 studied the effect of boron on lipid metabolism in both humans and animals. Boric acid added to the drinking water of adult male rats at levels of 2 mg boron/day for 2 weeks resulted in a trend for lower plasma total cholesterol and HDL3-cholesterol levels, as well as significant decreases in plasma triglyceride and total HDL-cholesterol concentrations (p < 0.05).47 These effects were transient, because only HDL3-cholesterol concentrations were significantly reduced at week 4 (p < 0.002).47 Boron supplementation for 4 weeks in humans showed no alterations in plasma lipid concentrations, oxidizability, or the distribution among LDL and HDL fractions.31
BORON AND BRAIN/ PSYCHOLOGICAL FUNCTION
A study with rats51 suggested that a low-boron diet in combination with other nutrient deficiencies could possibly have a negative effect on brain function. Vitamin D-deficient rats fed a low-boron diet containing 0.158 ppm boron had a diminished calcium and phosphorus balance, in contrast to rats fed a higher amount of boron (2.72 ppm). Boron deficiency in conjunction with vitamin D deficiency showed an increase in brain cortex calcium and a decrease in cerebellum phosphorus, which provided a direct measure of the changes taking place in brain tissue as a result of boron deficiency.51
Penland 47 conducted several studies using rats and humans to discover any possible relationships between dietary boron and psychological function. In adult rats, boron deprivation was shown to decrease high-frequency and increase low-frequency brain electrical activity, indicating depressed mental alertness. Experiments with humans measuring brain and psychological function indicated that inadequate dietary boron led to lower frequency activity in the electroencephalogram (EEG), as well as less favorable performance in cognitive and motor tests.52 These studies provide further evidence that dietary boron can alter the function of an organ system, and therefore may be an essential nutrient for humans and animals.52
CONCLUSION/ SUMMARY
Evidence is accumulating that dietary boron has a significant effect on certain metabolic processes, such as coronary heart disease, arthritis, and osteoporosis.30 Boron may prevent these chronic diseases by increasing the production of certain steroid hormones .
When boron is increased in the diets of humans, it has been shown by some researchers to
increase estrogen, testosterone, and plasma ionized calcium levels, and
decrease calcium excretion and the negative effects of vitamin D and magnesium deficiency.4,30,33,40,41
All of these effects could have a profound influence on increasing bone mineral density in humans, particularly in postmenopausal women. Before a dietary requirement for boron can be established, additional research on homeostatic control and the functional importance of boron should be performed, in addition to epidemiological research on health conditions related to inadequate dietary boron.6
REFERENCES
Warington, K., The effect of boric acid and borax on the broad bean and certain other plants, Ann. Bot., 1923; 37: 629-672.
Nielsen, F.H., Boron- an overlooked element of potential nutritional importance, Nutr. Today (Jan/Feb), 1988; 4-7.
Chapin, R.E., Ku, W.W., Kenney, M.A., and McCoy, H., The effects of dietary boric acid on bone strength in rats, Biol. Tr. Elem. Res., 1998; 66: 395-399.
Nielsen, F.H., Hunt, C.D., Mullen, L.M., and Hunt, J.R., Effect of dietary boron on mineral, estrogen, and testosterone metabolism in postmenopausal women, FASEB. J, 1987; 87: 394-397.
Lee, I.P., Sherins, R.J., and Dixon, R.L., Evidence for induction of germinal aplasia in male rats by environmental exposure to boron, Tox. Appl. Pharmacol., 1978; 45: 577-590.
Sutherland, B., Strong, P., and King, J.C., Determining human dietary requirements for boron, Biol. Tr. Elem. Res., 1998; 66: 193-204.
Naghii, M.R., The significance of dietary boron, with particular reference to athletes, Nutr. Health, 1999; 13: 31-37.
Loomis, W.D, and Durst, R.W., Review: chemistry and biology of boron, Biofactors, 1992; 3: 229-239.
Samman, S., Naghii, M.R., Lyons Wall, P.M., Verus, A.P., The nutritional and metabolic effects of boron in humans and animals, Biol. Tr. Elem. Res., 1998; 66: 227-235.
Hunt, C.D., One possible role of dietary boron in higher animals and humans, Biol. Tr. Elem. Res., 1998; 66: 205-225.
- {ATTACH(inline="1" id="7363" icon="1")}Download the Hunt PDF from VitaminDWiki{ (from SciHub)ATTACH}
Howe, P.D., A review of boron effects in the environment, Biol. Tr. Elem. Res., 1998; 66: 153-166.
Argust, P., Distribution of boron in the environment, Biol. Tr. Elem. Res., 1998; 66: 131-143.
Newnham, R.E., Mineral imbalance and boron deficiency. In: Underwood, E.J., Ed. Trace Elements in Human and Animal Nutrition, 4th ed. New York: Academic Press, Inc., 1977, 400-402.
Hunt, C.D., Shuler, T.L., and Mullen, L.M., Concentration of boron and other elements in human foods and personal care products, J. Am. Diet Assoc., 1991; 91: 558-568.
Clark, W.B., Koekebakker, M., Barr, R.D., Dowining,R. G., and Fleming, R.F., Analysis of ultratrace lithium and boron by neutron activation and mass-spectro- metric measurement of 3He and 4He, Appl. Radiat. Isot., 1987; 38: 735-734.
Newnham, RE., Agriculture practices affect arthritis, Nutr. Health, 1991; 7: 89-100.
Hamilton, E.I., Minsky, M.J., and Cleary, J.J., The concentration and distribution of some stable elements in healthy human tissues from the United Kingdom, Sci. Total Environ, 1972; 1: 341-374.
Indraprasit, S., Alexander, G.V., and Gonick, H.C., Tissue composition of major and trace elements in uremia and hypertension, J. Chron. Dis., 1974; 27: 135-161.
Massie, H.R., Whitney, S.J., Aiello, V.R., and Sternick,
S. M., Changes in boron concentration during development and aging of Drosophilia and effect of dietary boron on life span, Mech. Ageing Dev., 1990; 53: 1-7.
Moseman, R.F., Chemical disposition of boron in animals and humans. Environ. Health Perspect., 1994; 102(Suppl 7): 5-11.
Abou-Shakra, F.R., Havercroft, J.M., and Ward, N.I., Lithium and boron in biological tissues and fluids, Trace Elem. Med, 1989; 6: 142.
Hunt, C.D., Boron. In: Macrae, R., Robinson, R.K., and Sadler, M.J., Eds. Encyclopedia of Food Science, Food Technology and Nutrition. London: Academic Press, 1993, 440-447.
Von Burg, R., Toxicology update, J. Appl. Tox., 1992; 12: 149-152.
Volpe, S.L., Taper, L.J., and Meacham, S., The relationship between boron and magnesium status and bone mineral density in the human: a review, Magnes. Res, 1993(a); 6: 291-296.
Nielsen, F.H., Shuler, T.R., Zimmerman, T.J., and Uthus, E.O., Magnesium and methionine deprivation affect the response of rats to boron deprivation, Biol. Tr. Elem. Res, 1988; 17: 91-107.
Vanderpool, R.A. and Johnson, P.E., Boron isotope ratios in commercial produce and boron-10 foliar and hydroponic enriched plants, J. Agric. Food Chem., 1992; 40: 462-466.
Wilson, J.H. and Ruszler, P.L., Effects of dietary boron supplementation on laying hens, Br. Poul. Sci., 1996; 37: 723-729.
Ferrando, A.A. and Green, N.R., The effect of boron supplementation on lean body mass, plasma testosterone levels, and strength in male bodybuilders, Int. J. Sport Nutr, 1993; 3: 140-149.
Hunt, C.D., Herbel, J.L., and Nielsen, F.H., Metabolic responses of postmenopausal women to supplemental dietary boron and aluminum during usual and low magnesium intake: boron, calcium, and magnesium absorption and retention and blood mineral concentrations, Am. J. Clin. Nutr., 1997; 65: 803-813.
Naghii, M.R. and Samman, S., The effect of boron supplementation on its urinary excretion and selected cardiovascular risk factors in healthy male subjects, Biol. Tr. Elem. Res, 1997a; 56: 273-286.
Green, N.R. and Ferrando, A.A., Plasma boron and the effects of boron supplementation in males, Environ. Health Perspect., 1994; 102: 73-77.
Koivistoinen, P., Mineral element composition of Finnish foods, Acta. Agric. Scand., 1980; 22(Suppl.): 7-165.
Nielsen, F.H., Studies on the relationship between boron and magnesium which possibly affects the formation and maintenance of bones, Magnes. Trace Elem., 1990; 9: 61-69.
Mertz, W., Review of the scientific basis for establishing the essentiality of trace elements, Biol. Tr. Elem. Res, 1998; 66: 185-191.
Fort, D.L., Propst, T.L., Stover, E.L., et al., Adverse reproductive and developmental effects in Xenopus from insufficient boron, Biol. Tr. Elem. Res., 1998; 66: 237-259.
Food and Nutrition Board, Institute of Medicine, Panel on micronutrients, Subcommittees on Upper Reference Levels of nutrients and of interpretation and use of dietary reference intakes, and the standing committee on the scientific evaluation of dietary reference intakes. Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicone, vanadium, and zinc, Washington D.C.: National Academy Press, 2001.
Groff, J.L. and Gropper, S.S., Advanced Nutrition and Human Metabolism, 3rd ed., Belmont, CA: Wadsworth/ Thomson Learning, 2000.
Hunt, C.D. and Nielsen, F.H., Dietary boron affects bone calcification in magnesium- and cholecalciferol- deficient chicks. In: Underwood, E.J., Ed. Trace Elements in Human and Animal Nutrition, 5th ed. New York: Academic Press, Inc., 1986, 275-277.
Hunt, C.D., Dietary boron modified the effect of magnesium and molybdenum on mineral metabolism in the cholecalciferol-deficient chick, Biol. Tr. Elem. Res, 1989; 22: 201-220.
Hunt, C.D. and Nielsen, F.H., Interaction between boron and cholecalciferol in the chicks. In: McHowell, J., Gawthorne, J.H., and White, C.L., Eds. Trace Element Metabolism in Man and Animals, vol. 4. Canberra City: Australian Academy of Science, 1981, 97-600.
Hunt, C.D., Herbel, J.L., and Idso, J.P., Dietary boron modifies the effects of vitamin D3 nutriture on indices of energy substrate utilization and mineral metabolism in the chick, J. Bone Min. Res., 1994; 9: 171— 182.
Elliot, M.A. and Edwards, H.M., Studies to determine whether an interaction exists among boron, calcium, and cholecalciferol on the skeletal development of broiler chickens, Poul. Sci. 1992; 71: 677-690.
Gordon, G.S. and Vaughan, C., Calcium and osteoporosis, J. Nutr, 1986; 116: 319.
Wilson, J.H. and Ruszler, P.L., Effects of boron on growing pullets, Biol. Tr. Elem. Res., 1997; 56: 287294.
Wilson, J.H. and Ruszler, P.L., Long-term effects of boron on layer bone strength and production parameters, Br. Poul. Sci, 1998; 39: 11-15.
Volpe, S.L., Taper, L.J., and Meacham, S., The effect of boron supplementation on bone mineral density and hormonal status in college female athletes, Med. Exerc. Nutr. Health, 1993(b); 2: 323-330.
Naghii, M.R. and Samman, S., The effect of boron on plasma testosterone and plasma lipids in rats, Nutr. Research, 1997b; 17: 523-531.
Bai, Y. and Hunt, C.D., Dietary boron alleviates adjuvant- induced arthritis (AIA) in rats, FASEB J. 1995; 9: A576.
Bai, Y. and Hunt, C.D., Dietary boron (B) increases serum antibody concentrations in rats immunized with heat-killed Mycobacterium Tuberculosis (MT), FASEB J., 1996; 10: A819.
Hall, I.H., Spielvogal,B.F., Griffin, T.S., et al., The effects of boron hyperlipidemic agents on LDL and HDL receptor binding and related enzyme activities of rat hepatocytes, aorta cells and human fibroblasts, Res. Comm. Chem. Pathol. Pharmocol., 1989; 65: 297-317.
Hegsted, M., Keenan, M.J., Siver, F., and Wozniak, P., Effect of boron on vitamin D deficient rats, Biol. Tr. Elem. Res., 1991; 28: 243-255.
Penland, J.G., The importance of boron nutrition for brain and psychological function, Biol. Tr. Elem. Res., 1998; 66: 299-317.
