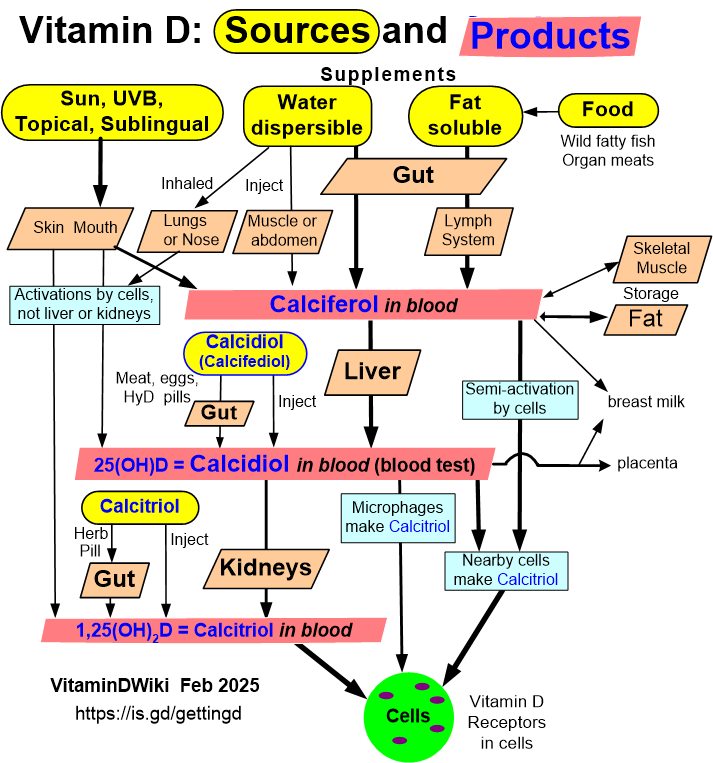
More than 30 ng of vitamin D is sometimes needed (Kidney needs 50 ng)
Rationale for Raising Current Clinical Practice Guideline Target for Serum 25-Hydroxyvitamin D in Chronic Kidney Disease.
Am J Nephrol. 2019;49(4):284-293. doi: 10.1159/000499187
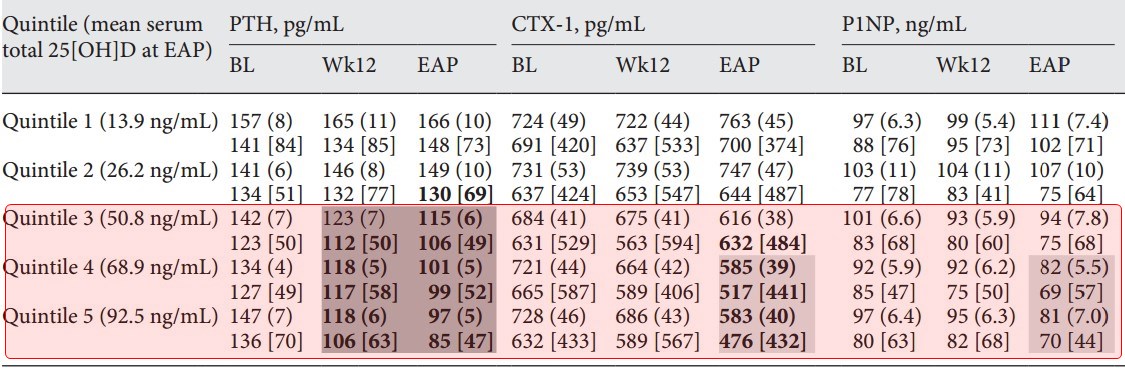
CTx-1 = Serum collagen type 1 C-telopeptide
P1NP = Intact procollagen type 1 N-terminal propeptide
Strugnell SA1, Sprague SM2, Ashfaq A3, Petkovich M4, Bishop CW3.
1 Renal Division, OPKO Health, Inc., Miami, Florida, USA, sstrugnell@opko.com.
2 Department of Medicine, NorthShore University HealthSystem, Evanston, Illinois, USA.
3 Renal Division, OPKO Health, Inc., Miami, Florida, USA.
4 Department of Biomedical and Molecular Sciences, Queen's University, Kingston, Ontario, Canada.
BACKGROUND:
Vitamin D repletion is recommended for secondary hyperparathyroidism (SHPT) and associated vitamin D insufficiency (VDI) in chronic kidney disease (CKD), but optimal levels of serum total 25-hydroxyvitamin D remain undefined. Clinical practice guidelines target sufficiency, whereas recent data indicate that higher levels are required to control the elevation of intact parathyroid hormone (iPTH) as CKD advances. This secondary analysis of 2 randomized controlled trials seeks to identify the minimum level of mean serum 25-hydroxyvitamin D required to control SHPT arising from VDI in stage 3 or 4 CKD.
METHODS:
Adult subjects (n = 429) with SHPT, VDI, and stage 3 or 4 CKD were stratified by stage and treated daily with either extended-release calcifediol (ERC) or placebo in 2 identical, parallel, randomized, double-blind studies. After treatment for 26 weeks, all subjects were ranked by the level of serum total 25-hydroxyvitamin D and divided into quintiles in order to examine the relationships between the degree of vitamin D repletion and the associated changes in plasma iPTH, serum bone turnover markers, calcium, phosphorus, intact fibroblast growth factor 23 (FGF23) and vitamin D metabolites, estimated glomerular filtration rate (eGFR), and urine calcium:creatinine (Ca:Cr) ratio.
RESULTS:
Progressive increases in serum 1,25-dihydroxyvitamin D and reductions in plasma iPTH and serum bone turnover markers were observed as mean posttreatment serum 25-hydroxyvitamin D rose from 13.9 ng/mL (in Quintile 1) to 92.5 ng/mL (in Quintile 5), irrespective of CKD stage. Mean serum calcium, phosphorus and FGF23, eGFR, and urine Ca:Cr ratio (collectively "safety parameters") did not significantly change from Quintile 1. Suppression of iPTH and bone turnover markers was not observed until serum 25-hydroxyvitamin D rose to at least 50.8 ng/mL (Quintile 3).
CONCLUSION:
ERC therapy produced exposure-dependent reductions in plasma iPTH and bone turnover markers only when mean serum total 25-hydroxyvitamin D reached at least 50.8 ng/mL, indicating that current targets for vitamin D repletion therapy in CKD are too low. Gradual elevation of mean serum 25-hydroxyvitamin D to 92.5 ng/mL was not associated with significant adverse changes in safety parameters.
