Vitamin K2 (as MK-7) is needed for bone quality – Review
Vitamin K2 and Bone Quality
Toshiro Sato oshiro.sato@j-oil.com
Fine Chemical Laboratory, Nakashinden, Fukuroi-city, Shizuoka, Japan
Introduction
Vitamin K acts as a cofactor for the endoplasmic enzyme y-glutamyl carboxylase, during post-translational conversion of glutamic acid residues to y-carboxyglutamic acid (Gla) in specific proteins. These proteins are referred to as vitamin K-dependent proteins, and include several blood coagulation factors and anti-coagulation factors, which are synthesized in the liver; Osteocalcin(OC), a bone-specific protein synthesized by osteoblasts; and Matrix Gla Protein (MGP), which is synthesized in several organs. Recently, considerable attention has been directed towards these vitamin K-dependent Gla proteins; their role in bone metabolism; and their inhibitory effect on artery calcification. The current average intake of vitamin K from a normal diet in healthy adults is greater than that required for normal blood coagulation, but is insufficient for extra hepatic tissue requirements [1,2].
Vitamin K2 has been found to be highly effective in bone metabolism compared to vitamin K1.
In Japan, a high dose of vitamin K2 (45 mg/day), as menaquinone-4 (MK-4) , is used as therapeutic treatment for osteoporosis. The principle effect of vitamin K2 on osteoporosis is prevention of bone fracture by improving bone quality, and not increasing bone mineral density. Recently, attention has been directed towards another vitamin K2 homolog; menaquinone-7 (MK- 7) extracted from Bacillus subtilis natto. This has been found to be highly effective in carboxylation of osteocalcin at nutritional doses. This review focuses on the effects of vitamin K2 as MK-7 on bone quality.
Structure of Vitamin K and its Distribution in Foods
There are two naturally occurring forms of vitamin K: vitamin K1 (phylloquinone), derived from green plants; and vitamin K2 (menaquinones, MK-n), which are a series of vitamers with multi-isoprene units at position 3 of the common 2-methyl-1,4- naphthoquinone ring structure.
In food, vitamin K1 is bound to the chloroplast membrane of leafy green vegetables; whereas, MK-4 is found in animal products, such as eggs, meat, and liver. MK-4 is derived from menadione (a synthetic analog of vitamin K, consisting only of the 2-methyl-1,4- naphthoquinone ring structure), which is given to animals as a feed additive nutrient and converted to MK-4 in animal tissues. Long chain menaquinones (e.g., MK-7, MK-8, and MK-9) are found in fermented foods such as cheese, curd, and sauerkraut [3]. The Japanese fermented food “natto” contains MK-7 at an exceptionally high concentration [3]. Vitamin K1, MK-4, and MK-7 are currently used as nutritional supplements and by the food industry (Figure 1).
Osteocalcin and Vitamin K
OC is produced by osteoblasts and forms bone matrix. Fully- carboxylated OC binds to calcium and shows affinity for hydroxyl apatite in bone [4]. The exact function of OC in bone is unclear, but is thought to be involved in calcium modulation. The first study using OC-knockout mice found that bone formation was accelerated in the knockout mice [5]; in contrast, bone structure turned fragile in OC- knockout mice after ovariectomy treatment [6]. This suggested that OC is important for bone maturation and bone quality.
OC has been used as a biomarker for bone metabolism: deficiency in vitamin K, elevates serum undercarboxylated OC (ucOC) levels; and high serum ucOC has been associated with hip fracture [7,8], and has been recognized as independent risk factor of fracture. In Japan, serum ucOC has been used as diagnostic marker to evaluate vitamin K deficiency in bone, since 2007.
Bone Quality and Vitamin K2
Murasawa et al. [9] conducted a study on ovariectomized rats fed with MK-7 (30 mg/kg bw per day) for 5 months. They observed a significant reduction in the bone mineral density of femurs in ovariectomized rats compared to sham-operated rats. MK-7 was modestly protective against this decrease (Figure 2); but markedly improved bone strength. Therefore, the benefits of MK-7 on bone strength were thought to be by maintaining and improving bone quality, and not by increasing bone mineral density.
A clinical study on post-menopausal women treated with MK-4 (45 mg/day) for 3 years, exhibited no effect on bone mineral density; in contrast, its effects on the maintenance of bone mass and bone quality indices of the femur were significant over the 3 year period [10]. Other intervention studies on vitamin K1 (5 mg/day; duration: 2 or 4 years) also found that vitamin K1 had no effect on bone mineral density, but significantly decreased the fracture rates [11].
Another possible mechanism by which vitamin K maintains and improves bone quality has been suggested. In addition to carboxylation and activation of OC, MK-4 may also increase collagen accumulation [12]. We confirmed this finding by showing that MK-7 increased collagen production through osteoblastic cells (Figure 3). Collagen occupies more than half the volume of bone and makes the foundations on which calcium and other minerals accumulate; and collagen accumulation contributes to bone flexibility and elasticity. Therefore, along with calcium and other minerals, collagen accumulation is critically important for high quality bone formation.
MK-7 has been reported to activate bone formation by osteoblastic cells [13], and suppress bone resorption [14]. The mechanism was recently demonstrated, and showed that MK-7 stimulates osteoblastogenesis and suppresses osteoclastogenesis by inhibiting NF- kB activation [15].
In addition to OC, many vitamin K-dependent proteins, such as MGP; protein S [16]; and periostin [17], are contained within the bone matrix; therefore, vitamin K-dependent proteins are thought to have, unknown functions in bone until now.
Observational studies of vitamin K intake and many observational studies on the relationship between bone metabolism, bone fracture, and vitamin K intake have been reported: MK-7 intakes from natto were inversely associated with bone fracture rates in Japan [18,19]; high serum ucOC was associated with hip fracture [7,8]; and serum vitamin K levels were inversely associated with hip fracture rates [20]. Furthermore, serum vitamin K levels and ucOC levels were inversely associated [21]. These findings clearly showed the close correlation between carboxylation of OC by vitamin K and fracture rates. Therefore, it is believed that an increased nutritional intake of vitamin K could reduce the risk of osteoporosis.
Vitamin K2 for Children
It is widely recognized that increasing peak bone mass before 20-30 years of age is very important for the prevention of osteoporosis, since bone mass gradually decreases with age. However, decreasing bone mass in children has been reported [22]. Bone metabolism is highly active in children, serum OC and ucOC levels are also extremely high compared to adults [23]. Furthermore, serum ucOC levels in children and bone health indices are inversely correlated [24,25]; and 45 pg of MK-7 has been shown to activate OC in children [26]. Therefore, it is expected that a sufficient intake of vitamin K may contribute children’s bone health.
Calcium Paradox and Vitamin K2
Ectopic artery calcification is frequently accompanied by osteoporosis in patients. This contradictory association is known as the “Calcium Paradox” or “Calcification Paradox” [27]. Vascular calcification is associated with increased cardiovascular mortality and morbidity [28,29], and is recognized as an independent risk factor for cardiovascular death [30]. A vitamin K-dependent protein, MGP, is a strong inhibitor of vascular calcification [31,32], and an intake of vitamin K, sufficient to fully activate MGP, is thought to prevent ectopic artery calcification. Two large epidemiological studies have shown inverse associations between vitamin K2 intake and cardiovascular death and/or disease [33,34]. Long chain vitamin K2 such as MK-7 mainly contributed to the result, while vitamin K1 showed no associations. Another study has also shown an inverse relationship between vitamin K2 intake and artery calcification [35]. The difference between vitamin K1 and K2 are due to differences in their bioavailability and cofactor activity. Recently, nutritional intake of MK-7 has been found to activate MGP [36,37]. Thus, it is expected that MK-7 may have therapeutic functions that contribute to both artery health and bone metabolism, and may solve the symptoms of “Calcium Paradox” [27].
Recent studies indicated that calcium supplements may increase cardiovascular rates [38,39]; it was concluded that this potentially detrimental effect on cardiovascular health needs to be balanced against the probable benefits of calcium on bone health [38]. It is widely recognized that calcium alone is not sufficient for bone health; therefore, other minerals, such as magnesium and vitamin D, are normally added. Vitamin K, through two vitamin K-dependent proteins; OC and MGP, has an essential role in modulating calcium homeostasis. As such, vitamin K2 should be recommended as an addition to calcium supplements.
Nutritional Features of Menaquinone-7
The effect of long chain vitamin K2, such as MK-7, on normal blood coagulation in rats was found to be higher and long lasting, than vitamin K1 and MK-4 [40,41]. A human study demonstrated that MK-7 derived from natto has a very long half-life time in the serum, and induced more complete carboxylation of OC compared to vitamin K1 [2]. It was also demonstrated that a high dose of MK-4 has a very short-half life in humans [42]. Our study found that a nutritional dose of MK-4 (420 |rg) was not absorbed; whereas, MK-7 was absorbed well in healthy women (Figure 4) [43]. The minimum dose of 1500 qg/day of MK-4 is required to activate OC in healthy subjects [44]; in contrast, 45-150 qg MK-7 was able to activate OC in healthy subjects [2,26,45].
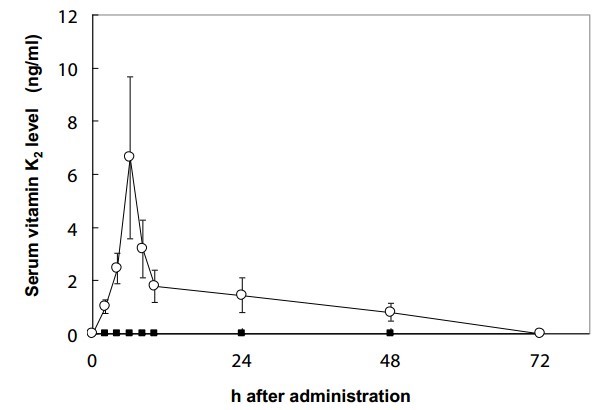
Figure 4: Change in serum vitamin K2 levels following a single oral dose (420 μg) of MK-4 or MK-7. Each point represents the mean ± SEM of 5 subjects, at 0, 2, 4, 6, 10, 24, 48, and 72 h. ■=MK-4; ○=MK-7 [43].
Because all vitamin K homologs are converted to MK-4 in organs, MK-4 has been thought to have specific functions, other than y-carboxylation of vitamin K-dependent proteins [46,47]. However, our previous rat study [48] found that an adequate nutritional intake of MK-4 did not increase the MK-4 levels in extrahepatic organs ; whereas, MK-7 increased MK-4 significantly in organs, such as the femur, brain, testis, kidney, and pancreas, indicating that MK-7 is better than MK-4 as an MK-4 precursor in vivo (Figure 5).
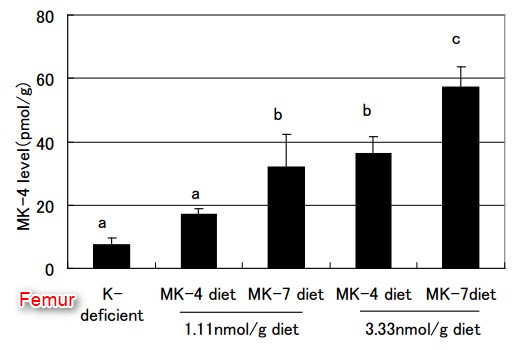
MK-4 appears to be the logical choice for the pharmacological use of vitamin K (at high dose; 45 mg/day) for the treatment for osteoporosis. The long half-life and better bioavailability of MK-7 would mean that a regular intake of physiological or nutritional doses of MK-7 (50-150 pg/day) would lead to the accumulation of vitamin K2 in extrahepatic tissues, at levels that could only be achieved by MK-4 at much higher doses.
Conclusion
The principle effects of vitamin K on bone health are to maintain and promote bone quality, and not to increase bone mineral density. Possibly the mechanisms may be activating osteocalcin, increasing collagen matrix, and regulating differentiation of osteoblasts and osteoclasts. Among the vitamin K homologs, MK-7 shows highest activity and bioavailability in humans. Therefore, it is believed that MK-7 at nutritional doses will promote bone health. A larger intervention trial for MK-7 is justified.
References
Cranenburg EC, Schurgers LJ, Vermeer C (2007) Vitamin K: The coagulation vitamin that became omnipotent. Thromb Haemost 98: 120-125.
Schurgers LJ, Teunissen KJ, Hamulyak K, Knapen MH, Vik H, et al. (2007) Vitamin K-containing dietary supplements: comparison of synthetic vitamin K1 and natto-derived menaquinone-7. Blood 109: 3279-3283.
Schurgers LJ, Vermeer C (2000) Determination of phylloquinone and menaquinones in food. Effect of food matrix on circulating vitamin K concentrations. Haemostasis 30: 298-307.
Hoang QQ, Sicheri F, Howard AJ, Yang DS (2003) Bone recognition mechanism of porcine osteocalcin from crystal structure. Nature 425: 977-980.
Ducy P, Desbois C, Boyce B, Pinero G, Story B, et al. (1996) Increased bone formation in osteocalcin-deficient mice. Nature 382: 448-452.
Boskey AL, Gadaleta S, Gundberg C, Doty SB, Ducy P, et al. (1998) Fourier transform infrared microspectroscopic analysis of bones of osteocalcin-deficient mice provides insight into the function of osteocalcin. Bone 23: 187-196.
Seibel MJ, Robins SP, Bilezikian JP (1997) Serum undercarboxylated osteocalcin and the risk of hip fracture. J Clin Endorcinol Metab 82: 717-718.
Vergnaud P, Garnero P, Meunier PJ, Breart G, Kamihagi K, et al. (1997) Undercarboxylated osteocalcin measured with a specific immunoassay predicts hip fracture in elderly women: the EPIDOS Study. J Clin Endocrinol Metab 82: 719-724.
Murasawa H, Araki S, Mitsui M, Shirota S, Ikeda R, et al. (1999) Studies on high-vitamin K containing natto and its effect on osteoporosis: Shokuhinsozai no Kinousei Souzou.Seigyo Gijutsu. (1st edn) The Japanese Research and Development Association for New Functional Foods, Koseisha Koseikaku 131146.
Knapen MH, Schurgers LJ, Vermeer C (2007) Vitamin K2 supplementation improves hip bone geometry and bone strength indices in postmenopausal women. Osteoporos Int 18: 963-972.
Cheung AM, Tile L, Lee Y, Tomlinson G, Hawker G, et al. (2008) Vitamin K supplementation in postmenopausal women with osteopenia (ECKO trial): a randomized controlled trial. PLoS Med 14: e196.
Ichikawa T, Horie-Inoue K, Ikeda K, Blumberg B, Inoue S (2006) Steroid and xenobiotic receptor SXR mediates vitamin ^-activated transcription of extracellular matrix-related genes and collagen accumulation in osteoblastic cells. J Biol Chem 281: 16927-16934.
Yamaguchi M, Sugimoto E, Hachiya S (2001) Stimulatory effect of
menaquinone-7 (vitamin K2) on osteoblastic bone formation in vitro. Mol Cell Biochem 223: 131-137. 2
Yamaguchi M, Ma ZJ (2001) Inhibitory effect of menaquinone-7 (vitamin K2) on osteoclast-like cell formation and osteoclastic bone resorption in rat bone tissues in vitro. Mol Cell Biochem 228: 39-47.
Yamaguchi M, Weitzmann MN (2011) Vitamin K2 stimulates osteoblastogenesis and suppresses osteoclastogenesis by suppressing NF-kB activation. Int J Mol Med 27: 3-14.
Maillard C, Berruyer M, Serre CM, Dechavanne M, Delmas PD (1992) Protein-S, a vitamin K-dependent protein, is a bone matrix component synthesized and secreted by osteoblasts. Endocrinology 130: 1599-1604.
Coutu DL, Wu JH, Monette A, Rivard GE, Blostein MD, et al. (2008) Periostin, a member of a novel family of vitamin K-dependent proteins, is expressed by mesenchymal stromal cells. J Biol Chem 283: 17991-18001.
Kaneki M, Hodges SJ, Hosoi T, Fujiwara S, Lyons A, et al. (2001) Japanese fermented soybean food as the major determinant of the large geographic difference in circulating levels of vitamin K2: possible implications for hip- fracture risk. Nutrition 17: 315-321.
Yaegashi Y, Onoda T, Tanno K, Kuribayashi T, Sakata K, et al. (2008) Association of hip fracture incidence and intake of calcium, magnesium, vitamin D, and vitamin K. Eur J Epidemiol 23: 219-225.
Hodges SJ, Pilkington MJ, Stamp TC, Catterall A, Shearer MJ, et al. (1991) Depressed levels of circulating menaquinones in patients with osteoporotic fractures of the spine and femoral neck. Bone 12: 387-389.
Tsugawa N, Shiraki M, Suhara Y, Kamao M, Tanaka K, et al. (2006) Vitamin K status of healthy Japanese women: age-related vitamin K requirement for gamma-carboxylation of osteocalcin. Am J Clin Nutr 83: 380-386.
Clark EM, Ness AR, Bishop NJ, Tobias JH (2006) Association between bone mass and fractures in children: a prospective cohort study. J Bone Miner Res 21: 1489-1495.
van Summeren M, Braam L, Noirt F, Kuis W, Vermeer C (2007) Pronounced elevation of undercarboxylated osteocalcin in healthy children. Pediatr Res 61: 366-370.
O’Connor E, M0lgaard C, Michaelsen KF, Jakobsen J, Lamberg-Allardt CJ, et al. (2007) Serum percentage undercarboxylated osteocalcin, a sensitive measure of vitamin K status, and its relationship to bone health indices in Danish girls. Br J Nutr 97: 661-666.
van Summeren MJ, van Coeverden SC, Schurgers LJ, Braam LA, Noirt F, et al. (2008) Vitamin K status is associated with childhood bone mineral content. Br J Nutr 100: 852-858.
van Summeren MJ, Braam LA, Lilien MR, Schurgers LJ, Kuis W, et al. (2009) The effect of menaquinone-7 (vitamin K2) supplementation on osteocalcin carboxylation in healthy prepubertal children. Br J Nutr 102: 1171-1178.
Persy V, D’Haese P (2009) Vascular calcification and bone disease: the calcification paradox. Trends Mol Med 15: 405-416.
Doherty TM, Asotra K, Fitzpatrick LA, Qiao JH, Wilkin DJ (2003) Calcification in atherosclerosis: bone biology and chronic inflammation at the arterial crossroads. Proc Natl Acad Sci 100: 11201-11206.
Zieman SJ, Melenovsky V, Kass DA (2005) Mechanisms, pathophysiology, and therapy of arterial stiffness. Arterioscler Thromb Vasc Biol 25: 932-943.
Rosenhek R, Binder T, Porenta G, Lang I, Christ G, et al. (2000) Predictors of outcome in severe, asymptomatic aortic stenosis. N Engl J Med 343: 611-617.
Luo G, Ducy P, McKee MD, Pinero GJ, Loyer E, et al. (1997) Spontaneous calcification of arteries and cartilage in mice lacking matrix GLA protein. Nature 386: 78-81.
Chatrou ML, Reutelingsperger CP, Schurgers LJ (2011) Role of vitamin K-dependent proteins in the arterial vessel wall. Hamostaseologie 31: 251-257.
Geleijnse JM, Vermeer C, Grobbee DE, Schurgers LJ, Knapen MH, et al. (2004) Dietary intake of menaquinone is associated with a reduced risk of coronary heart disease: the Rotterdam Study. J Nutr 134: 3100-3105.
Gast GC, de Roos NM, Sluijs I, Bots ML, Beulens JW, et al. (2009) A high menaquinone intake reduces the incidence of coronary heart disease. Nutr Metab Cardiovasc Dis 9: 504-510.
Beulens JW, Bots ML, Atsma F, Bartelink ML, Prokop M, et al. (2009) High dietary menaquinone intake is associated with reduced coronary calcification. Atherosclerosis 203: 489-493.
Westenfeld R, Krueger T, Schlieper G, Cranenburg EC, Magdeleyns EJ, et al. (2012) Effect of vitamin K2 supplementation on functional vitamin K deficiency in hemodialysis patients: a randomized trial. Am J Kidney Dis 59: 186-195.
Theuwissen E, Cranenburg EC, Knapen MH, Magdeleyns EJ, Teunissen KJ, et al. (2012) Low-dose menaquinone-7 supplementation improved extra-hepatic vitamin K status, but had no effect on thrombin generation in healthy subjects. Br J Nutr 108: 1652-1657.
Bolland MJ, Barber PA, Doughty RN, Mason B, Horne A, et al. (2008) Vascular events in healthy older women receiving calcium supplementation: randomised controlled trial. BMJ 336: 262-266.
Bolland MJ, Grey A, Avenell A, Gamble GD, Reid IR (2011) Calcium supplements with or without vitamin D and risk of cardiovascular events: reanalysis of the Women’s Health Initiative limited access dataset and metaanalysis. BMJ 342: d2040.
Groenen-van Dooren MM, Ronden JE, Soute BAM, Vermeer C (1995) Bioavailability of phylloquinone and menaquinones after oral and colorectal administration in vitamin K-deficient rats. Biochem Pharmacol 50: 797-801.
Sato T, Ohtani Y, Yamada Y, Saitoh S, Harada H (2001) Difference in the metabolism of vitamin K between liver and bone in vitamin K-deficient rats. Br J Nutr 87: 307-314.
Schurgers LJ, Vermeer C (2002) Differential lipoprotein transport pathways of K-vitamins in healthy subjects. Biochim Biophys Acta 1570: 27-32.
Sato T, Schurgers LJ, Uenishi K (2012) Comparison of menaquinone-4 and menaquinone-7 bioavailability in Japanese. Nutr J 11: 93.
Takeuchi A, Masuda Y, Kimura M, Marushima R, Matsuoka R, et al. (2005) Minimal effective dose of vitamin K2 (menaquinone-4) on serum osteocalcin concentration in Japanese subjects and safety evaluation of vitamin K2 supplemented in calcium tablet. J Jpn Soc Clin Nutr 26: 254-260.
Bruge F, Bacchetti T, Principi F, Littarru GP, Tiano L (2011) Olive oil supplemented with menaquinone-7 significantly affects osteocalcin carboxylation. Br J Nutr 106: 1058-1062.
Kimura S, Satoh H, Komai M (1992) The roles of intestinal flora and intestinal function on vitamin K metabolism. J Nutr Sci Vitaminol (Tokyo) 38: 425-428.
Nakagawa K, Hirota Y, Sawada N, Yuge N, Watanabe M, et al. (2010) Identification of UBIAD1 as a novel human menaquinone-4 biosynthetic enzyme. Nature 468: 117-121.
Sato T, Kawahara R, Kamo S, Saito S (2007) Comparison of menaquinone-4 and menaquinone-7 in rats. Vitamins (Japan) 81: 377-381.
PDF is attached at the bottom of this page
