Most vitamin D bought over-the-counter meet requirements
Comments by VitaminDWiki
- Many articles on the web alarmed people by telling the worst case from single pills when the average over 5 pills almost always meet the requirements and the label
- The reader is given the impression that the entire bottle may contain very little vitamin D
- Vitamin D Potency Varies Widely in Dietary Supplements, Analysis Finds Science Daily
- That Vitamin D You're Taking? It May Not Be What It Appears Forbes
- Vitamin D Supplements Found to Vary Widely in Potency Medscape
- Vitamin D doses often don't match labels, study says USA Today
- Vitamin D Supplement Potency Often Not What Label Says Medical News Today
- Vitamin D Supplement Labels May be Misleading Yahoo News
- The actual article meets both the requirments and our expectation.
- The processes which are made to make millions of pills are far more accurate than the process which involve humans to make a hundred pills.
- The requirement is for the average to be close what is specified on the label, and it was.
- Wonder how the JAMA press release was worded for this study.
- Was it as biased and distorted as the articles listed above?
Over-the-Counter and Compounded Vitamin D: Is Potency What We Expect?
JAMA Intern Med. 2013;():1-2. doi:10.1001/jamainternmed.2013.3812. Published online February 11, 2013
Erin S. LeBlanc, MD, MPH; Nancy Perrin, PhD; Jeffery D. Johnson, PhD; Annie Ballatore, MS; Teresa Hillier, MD, MS
Because vitamin D insufficiency can be harmful to health,1- 3 supplementation is often prescribed. However, the Food and Drug Administration (FDA) does not regulate vitamin D supplements, so potency may not be well evaluated. In a recent trial examining vitamin D in menopausal women, we found that compounded vitamin D3 (cholecalciferol) supplements varied significantly in potency. Only one-third of our compounded study pills met US Pharmacopeial (USP) Convention standards, which require that compounded pills contain 90% to 110% of the active ingredient.4 This variability in compounded cholecalciferol pills led us to additionally investigate over-the-counter (OTC) cholecalciferol pills, in which we also found variability.
End of abstract
Table (only 2 suppliers has less than 90% of the amount stated on the label)

Beginning of preview on the web
Because vitamin D Insufficiency can be harmful to health.1-3 supplementation is often prescribed. However, the Food and Drug Administration (FDA) does not regulate vitamin D supplements, so potency may not be well evaluated. In a recent trial examining vitamin D in menopausal women, we found that compounded vitamin D3 (cholecaldferol) supplements varied significantly in potency. (Only one-third of our compounded study pills met US Pharmaco-peial (USP) Convention standards, which require that compounded pills contain 90% to 110% of the active ingredient.4 This variability in compounded cholecaldferol pills led us to additionally investigate over-the-counter (OTC) cholecaldferol pills, in which we also found variability.
Methods. We randomly selected 5 pills from 15 sealed bottles of OTC cholecaldferol dietary supplements (1000 IU, 50001U, and 10 000IU) purchased at 5 stores in Portland, Oregon. We next randomly selected 1 pill from 5 bottles with the same lot number and 1 pill from 5 bottles with different lot numbers. We also analyzed compounded study cholecaldferol pills (1,000 IU and 50,000 IU), compounded on 3 occasions over 4 months. Testing was done at variable times (0-6 months) alter compounding.
Pill potency was analyzed using high performance liquid chromatography (HPLC) (Rapid Separation LC [RSLC] Ultimate 3000 UHPLC System; Dioncx) (see eAppendix; http://www.jamaintcrnalmcd.coin). To validate our laboratory testing accuracy, we split pills and sent them to a second laboratory. The values from the second laboratory were within 10% of the results of our laboratory.
We describe pill accuracy using the percentage of expected potency. We averaged overall potency of 5 pills from the same bottle and calculated the standard deviation. To standardize variability and compare across doses, we calculated the coefficient of variation (CV)-the ratio ol the standard deviation to the mean.
Results. Analysis of 5 Pills I torn the Same Bottle. The OTC pills contained 52% to 135% of expected dose (Table). When averaged over 5 pills, two-thirds of bottles met USP Convention standards for OTC cholecaldferol solution,1 which state that contents should be within 90% to 120% of the staled dose. In approximately one-fourth of bottles, all 5 pills met USP Convention standards. Two-thirds of bottles had less than 10% variability (CV). The one manufacturer that was USP verified (No. 4) was highly . . . . End of preview – the rest of the article in behind a paywall.
Letter to the Editor on the above study - similar variability in Multivitamins
Variability in Vitamin D Content Among Products for Multivitamin and Mineral Supplements Oct 2013
Karen W. Andrews, BS1; Pamela R. Pehrsson, PhD1; Joseph M. Betz, PhD2
JAMA Intern Med. 2013;173(18):1752-1753. doi:10.1001/jamainternmed.2013.8759.
To the Editor This letter is in response to the Research Letter by LeBlanc et al1 about vitamin D in dietary supplements. As scientists at the Agricultural Research Service of the US Department of Agriculture (USDA) and the Office of Dietary Supplements of the National Institutes of Health (NIH), we are also concerned about the variability and content of ingredients in dietary supplements. Results of our work show similar variability in vitamin D content among products and sometimes among lots of multivitamin and mineral (MVM) supplements. Overall, we find that most MVM products have vitamin D overages but with considerable variability . The mean percentage difference for vitamin D tested in a pilot study of 54 adult MVM products was +17% of the label (range, +172% to −93%).2 The predicted mean percentage difference from the label using weighted regression techniques for a national sampling of 64 children’s MVM products was +34%3 (range, +110% to −38%). This variability is higher than that reported by LeBlanc et al1 and may be due to the multiple matrices evaluated by the USDA (MVM products were defined as supplements with >3 vitamins) and a larger national sampling of MVM products for these studies that included multiple channels.
Other variations are far larger than the variations between manufacturers
- 300% in the response that an individual will have to the same dose than the variation in the dose inside of a pill
- 200% as much response from a dose of vitamin D
- 300% variation between individuals just due to weight. Many believe that the vitamin D dose should be based on weight.
Example of huge variation in the response from the same manufacturer
USANA: which prides itself on quality control (and costs about 3X more than the competition)
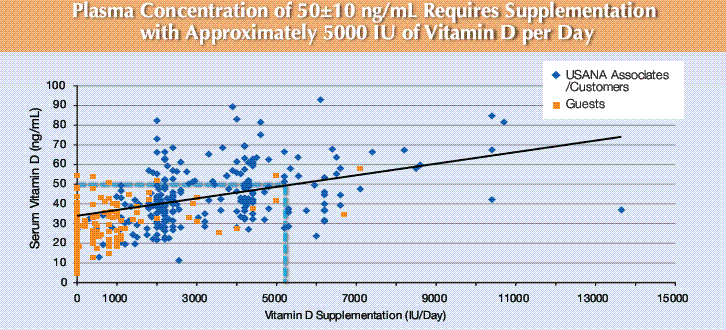
There was a lot of controversy about amount of vitamin D in pills back in 2010
Average vitamin D contained only 33% of what is on the label – June 2010
See also web about this study
- All Vitamin D Supplements Are the Same, Right? Uhm, Not Quite. OM Times Dec 2013
