Foot and Ankle Surgeries should benefit from Vitamin D
Surgical Considerations for Vitamin D Deficiency in Foot and Ankle Surgery
Orthop Clin N Am (2018) https://doi.org/10.10167j.od.2018.10.0080030-5898/18/
Kenneth DeFontes III, MD a,b, Jeremy T. Smith, MD a' jsmith42@bwh.harvard.edu
a Department of Orthopaedics, Brigham and Women's Hospital, 75 Francis Street, Boston, MA 02115, USA;
b Towson Orthopaedic Associates, Ruxton Professional Center, 8322 Bellona Avenue, Suite 100, Towson, MD 21204, USA
📄 Download the PDF from Sci-Hub via VitaminDWiki
Key Points
A substantial percentage of patients undergoing foot and ankle surgery are vitamin D deficient.
Consideration should be given for preoperative vitamin D testing in at-risk patients undergoing foot and ankle surgery.
Vitamin D repletion either preoperatively or postoperatively is a proposed way to optimize outcomes in orthopedic surgery, including foot and ankle surgery.
There may be a correlation between vitamin D deficiency and poor clinical outcomes.
Vitamin D supplementation is relatively safe and inexpensive.
INTRODUCTION
Vitamin D deficiency is extremely common, affecting more than one billion people world- wide.1 There are many known causes of vitamin D deficiency, including malnutrition, malabsorption syndromes, and inadequate sun exposure. Vitamin D level has been shown to correlate to both climate and geography, which is in part thought to be due to variations in sunlight, which affects vitamin D metabolism (Fig 1). Vitamin D deficiency is particularly prevalent in certain populations. For example, patients with darker skin living in areas with less sun exposure are at particular risk for low vitamin D.1,3,4 Certain medical conditions also predispose to vitamin D deficiency, including patients with gastrointestinal malabsorption syndromes and renal insufficiency.1
Relating to orthopedics, certain patient subgroups have been noted to be at particular risk for low vitamin D levels. These subgroups include orthopedic trauma patients,5-11 geriatric hip and fragility fracture patients,12-15 patients undergoing joint replacement surgery,16-25 spinal fusion patients,26-28 those undergoing scoliosis correction,29 and patients with foot and ankle conditions.30-35 This article summarizes the current literature regarding vitamin D deficiency in patients undergoing orthopedic surgery, focusing on patients with foot and ankle conditions.
VITAMIN D METABOLISM
A general understanding of vitamin D metabolism is necessary to appreciate the intricate relationship between vitamin D levels, calcium homeostasis, and bone health (see Fig. 1). Vitamin D is absorbed both through the skin and through the gastrointestinal tract. Vitamin D2, also known as ergocalciferol, is derived from plant sources and generated by UV radiation of ergosterol from yeast.1 Vitamin D3 is called cholecalciferol and is generated from the UV irradiation of 7-dehydrocholesterol from lanolin, which is found in animals.1,36 Both vitamin D2 and vitamin D3 are converted to 25-hydroxyvitamin D (25(OH)D) in the liver. This major circulating metabolite, also called cal- cidiol, is the most commonly measured vitamin D laboratory value.
Fig. 1. Photoproduction and metabolism of vitamin D and the various biologic effects of 1,25(OH)2D on calcium, phosphorus, and bone metabolism. DBP, Vitamin D Binding Protein. (From McCabe MP, Smyth MP, Richardson DR. Current concept review: vitamin D and stress fractures. Foot Ankle Int 2012;33(6):527; with permission.)
Calcidiol (25(OH)D) circulates in the blood and undergoes hydroxylation in the kidneys to become calcitriol (1,25(OH)D).1,36 As the active form of vitamin D, calcitriol affects both bone turnover and gastrointestinal absorption to regulate calcium and phosphate levels. In the setting of vitamin D deficiency, less calcium is absorbed from the gastrointestinal track, which upregulates parathyroid hormone (PTH).1,37,38 Increased PTH has the effect of increasing bone resorption, which can lead to osteoporosis, osteomalacia/rickets, and compromised bone healing.1,39
VITAMIN D AND MUSCULOSKELETAL HEALTH
Vitamin D levels are reported in either nanograms per milliliter or nanomoles per liter. Calcidiol (25(OH)D) is the value measured in most routine laboratory tests. Although there is no universally accepted classification of vitamin D deficiency or insufficiency, there is a consensusin the literature regarding normal values. The Endocrine Society defines vitamin D insufficiency as between 20 and 30 ng/mL and vitamin D deficiency as less than 20 ng/mL.40 Patients with values greater than 30 ng/mL are considered sufficient (Table 1). Most of the studies in the orthopedic and foot and ankle literature use the Endocrine Society definition of vitamin D adequacy, and unless otherwise noted, all articles referenced in this chapter use this definition.
Vitamin D is critical to bone health due to its regulation of serum calcium and phosphate. In the most basic sense, low levels of vitamin D result in low levels of calcium. In one of the most striking examples of vitamin D deficiency, rickets, bone is exceptionally soft and weakened, which results in delayed growth and skeletal deformity.1,3,39 In cases of renal osteodystrophy, effective vitamin D deficiency results in substantial bone weakness and fracture risk.1,37 These examples illustrate the integral role that vitamin D plays in bone health. Similarly, low vitamin D has been identified as a common cause of fragility fractures in patients without known medical underlying risk factors for vitamin D deficiency.12-14,41 Fragility fractures are not only an enormous burden to the elderly population, in which they most commonly occur, but also an economic burden to the health care system.1,42
In addition to the well-documented effects of vitamin D on bone homeostasis, vitamin D also directly affects muscle. Adequate vitamin D levels have been associated with minimizing risks of falls, suggesting a connection between vitamin D and muscle strength.1,43,44 Vitamin D interacts directly with muscle through the vitamin D receptor that is found in skeletal muscle.43,45,46 Additional research is needed to fully elucidate the role of vitamin D and skeletal muscle health and function.
For supplementation in those with adequate vitamin D levels, the Endocrine Society Clinical Practice Guidelines recommend at least 600 IU of vitamin D daily for patients age 1 to 70 and at least 800 IU daily for patients over the age of 70, with a general upper limit of 4000 IU daily for adults.40,47 For those found to be deficient of vitamin D, it is recommended that children aged
Table 1
Normal 25-hydroxyvitamin D values and cutoffs for insufficiency and deficiency
Diagnosis Values (ng/mL) Values (nmol/L)
Sufficient >30 >75
Insufficient 21-29 52.5-72.5
Deficient <20 <50
1 to 18 take 2000 IU daily or a 6-week course of 50,000 IU weekly followed by 400 to 1000 IU daily.40 Adults with deficiency are recommended 6000 IU daily or 50,000 IU weekly for 8 weeks followed by maintenance treatment of 1500 to 2000 IUs daily.40 The goal of treatment is to maintain levels greater than 30 ng/mL. Either D2 or D3 supplementation is acceptable.40
Vitamin D toxicity has been described and is extremely rare. Levels greater than 150 ng/mL are considered excessive and can result in intox- ication.48 Symptoms of vitamin D intoxication range from mild gastrointestinal distress (nausea, vomiting, and diarrhea) to more severe symptoms, including lethargy, headache, cardiac arrhythmia, muscle and joint pain, frequent urination, and kidney stones.48,49
VITAMIN D IN ORTHOPEDIC TRAUMA
The rate of vitamin D deficiency is particularly high in orthopedictrauma patients, including both low- energy and high-energy trauma.7,10,11,13,41 In the geriatric hip fracture population, vitamin D levels have been reported to be as high as 76%.13 Nevertheless, despite high rates of deficiency among those with fractures, it is not clear that vitamin D deficiency affects outcomes after fracture. A recent meta-analysis evaluated vitamin D supplementation in fracture patients and found that vitamin D supplementation was a safe way to increase vitamin D levels in all cases, but it was not clear whether supplementation affected outcomes.9 Similarly, a study from Singapore did not show any effect of vitamin D deficiency on clinical outcomes in 171 patients with geriatric hip fractures.15 Lee and colleagues13 in 2015 looked at vitamin D deficiency as a risk factor for mortality in older patients with a hip fracture and found no correlation between serum 25(OH)D levels and mortality. Although vitamin D levels may not affect clinical outcomes after trauma, some researchers think that vitamin D has an important role in fracture prevention. A large meta-analysis published in 2005 concluded that vitamin D supplementation (700-800 IU daily) reduced the risk of hip and any nonvertebral fractures in ambulatory or institutionalized elderly patients.50
Nonunions in trauma patients have been linked to vitamin D deficiency. In 2007, Brinker and colleagues5 investigated 683 patients who developed a nonunion from a fracture over a 7-year period. The investigators identified patients in whom a metabolic cause for nonunion was suspected by identifying those with adequate fracture stabilization and reduction, those who had a history of multiple low-energy fractures, and those with a nonunited pubic rami or sacral ala fracture. Thirty- seven patients were identified and referred to an endocrinologist. Of these, 68% were found to be vitamin D deficient. Although this study does not definitively identify a causal link between low vitamin D levels and the development of nonunion, most patients deemed to be at highest risk for nonunion had vitamin D deficiency.
The orthopedic trauma literature also sheds light on the effectiveness of vitamin D supplementation in patients with vitamin D deficiency. In 2015, Robertson and colleagues8 monitored vitamin D levels of 201 orthopedic trauma patients throughout the course of treatment. Before initiation of vitamin D therapy, the rate of vitamin D deficiency was 40% and the rate of insufficiency was 44%. All patients, regardless of preoperative levels, received 1000 IU D3 and 1500 mg of calcium daily, and those insufficient or deficient additionally received 50,000 IU D2 weekly for 8 weeks. Vitamin D levels in all patients improved throughout follow-up, but most patients did not normalize. These data suggest that supplementation can improve vitamin D levels, although attention should be given to dosing and duration of supplementation.
VITAMIN D IN ELECTIVE ORTHOPEDIC SURGERY
Vitamin D deficiency has been reported to be present in up to 86% of patients undergoing elective orthopedic procedures.16-18'27'28'31-51'52 Patients undergoing elective hip and knee replacement have been found to have a particularly high prevalence of vitamin D deficiency.16,22 For comparison, although rates of vitamin D deficiency tend to vary geographically, an analysis of healthy adults aged 18 to 29 in the United States reported 36% had 25(OH)D levels less than or equal to 20 ng/mL.53,54 A study of healthy patients in France reported a deficiency rate of 14%.55 Although these groups vary in terms of age, many studies evaluating elective orthopedic patients report substantially higher rates of deficiency than population norms.
Studies have shown that patients with lower preoperative vitamin D levels have lower preoperative functional status.21 Whether this is due to a causal effect of vitamin D or is representative of poorer general fitness is uncertain. Studies also suggest that patients with lower vitamin D levels have lower outcome scores after total knee and hip replacement.23,24,56 Shin and col- leagues24 analyzed a prospective cohort of 92 patients who underwent total knee arthroplasty and found that mean postoperative Knee
Society Scores and additional performance tests were significantly lower in the vitamin D deficient group. Unnanuntana and colleagues25 showed that early postoperative functional outcomes, at 6 weeks, were similar in patients who were deficient versus sufficient preoperatively, as long as adequate vitamin D supplementation was begun as soon as it was identified. Additional research is needed to fully elucidate the effects of vitamin D deficiency and supplementation in the arthroplasty population.
There also appears to be a link between vitamin D and periprosthetic infection rate, length of hospital stay, and overall complication rates after elective orthopedic surgery.17,19,20,22 Hegde and colleagues17 evaluated 6598 patients undergoing elective joint replacement in the Humana registry and found those with vitamin D deficiency to have a statistically higher rate of periprosthetic surgical site infection and prosthesis explantation as well as a higher overall complication rate. Similarly, Maier and colleagues19 found significant differences in vitamin D levels in patients who developed an infection after hip, knee, or shoulder arthroplasty when compared with patients who were either scheduled for arthroplasty or developed aseptic loosening. Maier and colleagues20 also reported longer hospital stays in those with vitamin D deficiency in 1083 patients undergoing elective hip or knee arthroplasty.
VITAMIN D IN FOOT AND ANKLE SURGERY
Much of the early literature looking at hypovita- minosis D and foot and ankle patients relates to bone marrow edema syndrome, also known as transient osteoporosis.57 Bone marrow edema syndrome is characterized by a sudden onset of severe periarticular pain without trauma and has a predilection for the lower extremities, including the hip, knee, foot, and ankle.57,58 Multiple studies have evaluated the association between vitamin D and bone marrow edema syndrome in the foot and have found vitamin D deficiency in 84% to 90% of these patients.57,59,60 Treatment of bone marrow edema syndrome is largely nonsurgical and includes off-loading, synthetic prostacyclin analogues, bisphosphonates, and vitamin D supplementation.57 When combined with other medical therapies, such as bisphosphonates, vitamin D supplementation seems to cause some improvement in pain, suggesting vitamin D supplementation may be an important part of treatment of these patients.59-61
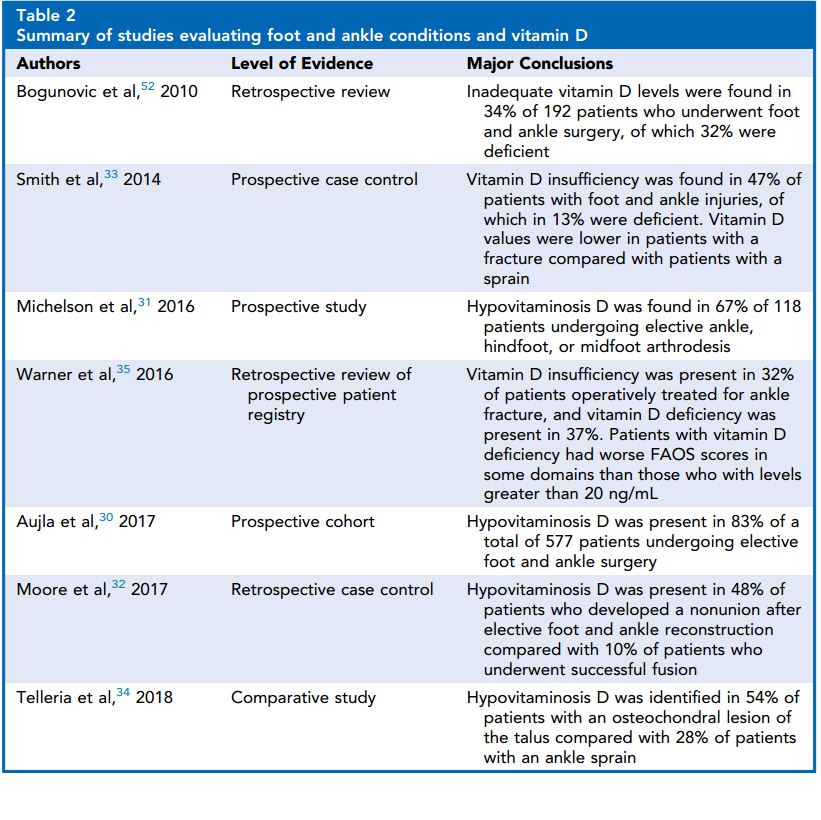
More recently, studies have examined rates of hypovitaminosis D in patients with foot and ankle injuries (Table 2). In 2014, Smith and colleagues33 evaluated the prevalence of vitamin D deficiency in patients who presented with acute low-energy foot and ankle injuries. The study cohort included patients with a low- energy ankle fracture, fifth metatarsal base fracture, or stress fracture of the foot and ankle. A group of patients with an ankle sprain were used as a control. Of patients with a low- energy foot or ankle fracture, 47% were vitamin D insufficient, with 13% of patients severely deficient. These values were statistically significantly lower than in the control group, of which 71% had normal vitamin D levels. Further analysis revealed an association between low vitamin D and smoking, obesity, and other medical riskfactors for hypovitaminosis D. This study demonstrated that vitamin D deficiency is particularly common among patients with a low-energy fracture of the foot or ankle. Although foot and ankle injuries are not typically classified as fragility fractures, the investigators suggest that clinicians consider these injuries to be related to overall bone health. Accordingly, consideration should be given to checking vitamin D levels in patients who present with low-energy foot or ankle injuries.
Vitamin D levels have also been evaluated in elective foot and ankle surgery populations. A study published in 2017 evaluated 577 patients in England undergoing elective foot and ankle surgery.30 This large cohort reported the levels of vitamin D to be normally distributed, yet with only 18% of patients within the normal range. The investigators noted an association between the season and vitamin D levels, which can likely be attributed to levels of sun exposure.
Table 2 Summary of studies evaluating foot and ankle conditions and vitamin D
67% of patients were found to have low vitamin D levels (<30 ng/mL). Interestingly, older patients had a statistically lower risk for hypovita- minosis D. The investigators also looked at the Charlson Co-Morbidity Index.62 Patients with a Charlson Co-Morbidity Index greater than or equal to 3, which corresponds to either patients with diabetes or multiple comorbid conditions, were at increased risk for vitamin D deficiency. To address the deficiency, all patients were placed on 2000 IU D3 and 750 mg calcium carbonate daily and those with hypovitaminosis D were also treated with 50,000 IU D2 three times a week for 2 to 3 months. Despite this repletion, only 56% of patients who were deficient corrected to normal levels. This study illustrates 2 important points. The prevalence of vitamin D deficiency in patients undergoing foot and ankle fusions, at least in Vermont, is quite high. Second, the repletion approach used in this study failed to adequately normalize levels in many patients.
An additional group of patients who have been recently studied are those with osteochondral lesions of the talus (OLTs). Patients with OLTs have a particularly high rate of vitamin D deficiency, as identified by Telleria and colleagues.34 In patients with a documented OLT, the prevalence of vitamin D insufficiency is 54%. This rate of hypovitaminosis contrasts with an acute ankle sprain. These data suggest that OLTs may be related to an underlying bone abnormality, and the investigators suggest that patients presenting with a talar osteochondral lesion should be evaluated for vitamin D sufficiency. Although repletion was not evaluated in this study, the implication is that vitamin D and overall bone health should be considered when treating patients with OLT. Other studies have looked at juvenile osteochondritis dissecans and have similarly found high rates of vitamin D deficiency, ranging from 60% to 78%.63,64
The relationship between vitamin D deficiency and nonunion has also been studied in patients undergoing foot and ankle surgery.32 Twenty- nine patients who underwent successful arthrodesis (forefoot, midfoot, hindfoot, ankle) were matched to 29 patients who developed a nonunion after an arthrodesis procedure. Vitamin D deficiency or insufficiency was identified in 48% of patients who developed a nonunion, compared with 10% in patients who united. Statistical analysis revealed that patients with a preoperative diagnosis of vitamin D insufficiency or deficiency were 8 times more likely to develop a nonunion than patients who had sufficient vitamin
D levels. Although this study is small and cannot definitively link hypovitaminosis D and nonunion, these data suggest that an association may exist between vitamin D and the development of nonunion in foot and ankle surgery. Further study into these relationships is needed.
With respect to treatment of hypovitaminosis D, there are very few studies that examine the benefits of treatment on outcomes after foot and ankle surgery. In a study examining hypovitaminosis D in patients undergoing ankle fracture fixation, Warner and colleagues35 reported improved outcomes in patients who were vitamin D sufficient. In this study, the investigators identified a hypovitaminosis D prevalence of 69% in a registry of 98 patients. Patients with vitamin D deficiency had statistically lower Foot and Ankle Outcome Score (FAOS), specifically in the symptoms and quality-of-life domains. Whether the poorer outcome was due to the vitamin D level or if this was an indication of poorer overall fitness is not known. If an association between vitamin D and outcomes does exist, one can theorize as to why vitamin D is beneficial. This benefit could be due to direct effects on bone healing or alternatively influencing balance and muscle strength and thus facilitating better rehabilitation and recovery.43,65,66
With respect to postoperative rehabilitation after foot and ankle surgery, vitamin D has been shown to benefit skeletal muscle and athletic performance. A recent review examines the role of vitamin D in muscle function.43 Skeletal muscle has a vitamin D receptor that, once activated, enhances the interaction between myosin and actin. In a murine knockout model, mice without functional vitamin D receptors have been shown to have smaller muscle fiber size and body weight compared with normal mice, even when calcium levels are normal- ized.43,67 Vitamin D is thus postulated to have a direct effect on muscle strength, performance, and recovery.43,45 Additional research is necessary to further develop the understanding of the complex interplay between vitamin D and muscle strength, particularly as it pertains to recovery after injury or surgery.
SUMMARY
In summary, vitamin D deficiency is common in patients undergoing orthopedic procedures, including patients with foot and ankle conditions and those undergoing foot and ankle surgery. As the understanding and awareness of vitamin D and bone health evolve, we continue to identify conditions in which vitamin D deficiency is clinically relevant. The limited data available to date suggest that patients who are vitamin D sufficient may have improved outcomes. The authors would thus recommend monitoring vitamin D levels as part of the preoperative evaluation for certain foot and ankle conditions. Because of its low cost and ease of administration, vitamin D optimization should be a routine consideration before orthopedic surgery.
REFERENCES
Holick MF. Vitamin D deficiency. N Engl J Med 2007;357:266-81.
Holick MF, Chen TC, Lu Z, et al. Vitamin D and skin physiology a D-lightful story. J Bone Miner Res 2007;22:V28-33.
Pettifor JM, Moodley GP, Hough FS, et al. The effect of season and latitude on in vitro vitamin D formation by sunlight in South Africa. S Afr Med J 1996;86:1270-2.
Vieth R. Why the optimal requirements for vitamin D3 is probably much higher than what is officially recommended for adults. J Steroid Biochem Mol Biol 2004;89-90:575-9.
Brinker MR, O'Conner DP, Monla YT, et al. Metabolic and endocrine abnormalities in patients with nonunions. J Orthop Trauma 2007;21:557-70.
Feng Y, Cheng G, Wang H, et al. The associations between serum 25-hydroxyvitamin D level and the risk of total fracture and hip fracture. Osteoporos Int 2017;28:1641-52.
Hood MA, Murtha YM, Della Rocca GJ, et al. Prevalence of low vitamin D levels in patients with orthopaedic trauma. Am J Orthop 2016;45:E522-6.
Robertson DS, Jenkins T, Murtha YM, et al. Effectiveness of vitamin D therapy in orthopaedic trauma patients. J Orthop Trauma 2015;29:e451-3.
Sprague S, Petrisor B, Scott T, et al. What is the role of vitamin D supplementation in acute fracture patients? A systematic review and meta-analysis of the prevalence of hypovitaminosis D and supplementation efficacy. J Orthop Trauma 2016;30:53-63.
Steele B, Serota A, Helfet DL, et al. Vitamin D deficiency: a common occurrence in both high- and low-energy fractures. HSS J 2008;4:143-8.
Zellner BS, Dawson JR, Reichel LM, et al. Prospective nutritional analysis of a diverse trauma population demonstrates substantial hypovitaminosis D. J Orthop Trauma 2014;28:e210-5.
Buchebner D, McGuigan F, Gerdhem P, et al. Association between hypovitaminosis D in elderly women and long- and short-term mortality - results from the Osteoporotic Prospective Risk Assessment Cohort. J Am Geriatr Soc 2016;64: 990-7.
Lee GH, Lim JW, Park YG, et al. Vitamin D deficiency is highly concomitant but not strong risk factor for mortality in patients aged 50 years and older with hip fracture. J Bone Metab 2015;22:205-9.
Maier GS, Seeger JB, Horas K, et al. The prevalence of vitamin D deficiency in patients with vertebral fragility fractures. Bone Joint J 2015;97-B:89-93.
Seng WR, Belani MH, Ramason R, et al. Functional improvement in geriatric hip fractures: does vitamin D deficiency affect the functional outcome of patients with surgically treated interochanteric hip fractures. Geriatr Orthop Surg Rehabil 2015;6:186-91.
Glowacki J, Hurwitz S, Thornhill TS, et al. Osteoporosis and vitamin-D deficiency among postmenopausal women with osteoarthritis undergoing total hip arthroplasty. J Bone Joint Surg Am 2003;85-A: 2371-7.
Hegde V, Arshi A, Wang C, et al. Preoperative vitamin D deficiency is associated with higher postoperative complication rates in total knee arthroplasty . Orthopedics 2018;27:1-7.
Inkrott BP, Koberling JL, Noel CR. Hypovitaminosis D in patients undergoing shoulder arthroplasty: a single-center analysis. Orthopedics 2016;39:e651-6.
Maier GS, Hora K, Seeger JB, et al. Is there an association between periprosthetic joint infection and low vitamin D levels? Int Orthop 2014;38:1499-504.
Maier GS, Maus U, Lazovic D, et al. Is there an association between low serum 25-OH-D levels and the length of hospital stay in orthopaedic patients after arthroplasty? J Orthop Traumatol 2016;17:297-302.
Maniar RN, Patil AM, Maniar AR, et al. Effect of preoperative vitamin D levels on functional performance after total knee arthroplasty . Clin Orthop Surg 2016;8:153-6.
Russell LA. Osteoporosis and orthopedic surgery: effect of bone health on total joint arthroplasty outcome. Curr Rheumatol Rep 2013;15:371.
Schwartz FH, Lange J. Factors that affect outcome following total joint arthroplasty: a review of the recent literature. Curr Rev Musculoskelet Med 2017;10:346-55.
Shin KY, Park KK, Moon SH, et al. Vitamin D deficiency adversely affects early post-operative functional outcomes after total knee arthroplasty . Knee Surg Sports Traumatol Arthrosc 2017;25:3424-30.
Unnanuntana A, Saleh A, Nguyen JT, et al. Low vitamin D status does not adversely affect shortterm functional outcome after total hip arthroplasty. J Arthroplasty 2013;28:315-22.
Kim TH, Yoon JY, Lee BH, et al. Changes in vitamin D status after surgery in female patients with lumbar spinal stenosis and its clinical significance. Spine 2012;37:E1326-30.
Mabey T, Singhatanadgige W, Yingsakmongkol W, et al. Vitamin D and spine surgery. World J Orthop 2016;18:726-30.
Stoker GE, Buchowski JM, Bridwell KH, et al. Preoperative vitamin D status of adults undergoing surgical spinal fusion. Spine 2013;15:507-15.
Mayes T, Anadio JM, Sturm PF. Prevalence of vitamin D deficiency in pediatric patients with scoliosis preparing for spinal surgery. Spine Deform 2017;5:369-73.
Aujla RS, Allen PE, RibbansWJ. Vitamin D levels in 577 consecutive elective foot & ankle surgery patients. Foot Ankle Surg 2017 [pii:S1268-7731 (17)31368-1].
Michelson JD, Charlson MD. Vitamin D status in an elective orthopaedic surgical population. Foot Ankle Int 2016;37:186-91.
Moore KR, Howell MA, Saltrick KR, et al. Risk factors associated with nonunion after elective foot and ankle reconstruction: a case-control study. Foot Ankle Surg 2017;56:457-62.
Smith JT, Halim K, Palms DA, et al. Prevalence of vitamin D deficiency in patients with foot and ankle injuries. Foot Ankle Int 2014;35:8-13.
Telleria JS, Ready LV, Bluman EB, et al. Prevalence of vitamin D deficiency in patients with talar osteochondral lesions. Foot Ankle Int 2018;39:471-8.
Warner SJ, Garner MR, Nguyen JT, et al. Perioperative vitamin D levels correlate with clinical outcomes after ankle fracture fixation . Arch Orthop Trauma Surg 2016;136:339-44.
Christakos S, Dhawan P, Verstuyf A, et al. Vitamin D: metabolism, molecular mechanisms of action, and pleiotrophic effects. Physiol Rev 2016;96:365-408.
Holick MF. Vitamin D for health in chronic kidney disease. Semin Dial 2005;18:266-75.
Thomas KK, Lloyd-Jones DM, Thadhani RI, et al. Hypovitaminosis D in medical inpatients. N Engl J Med 1998;338:777-83.
Holick MF. Ressurection of vitamin D deficiency and rickets. J Clin Invest 2006;116:2062-72.
Holick MF, Binkley NC, Bischoff-Ferarri HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2011;96:1911-30.
Leboff MS, Kohlmeier L, Hurwitz S, et al. Occult vitamin D deficiency in postmenopausal US women with acute hip fracture. JAMA 1999;281:1505-11.
Orsini LS, Rousculp MD, Long SR, et al. Healthcare utilization and expenditures in the United States: a study of osteoporosis-related fractures. Osteo- poros Int 2005;16:359-71.
Abrams GD, Feldman D, Safran MR. Effects of vitamin D on skeletal muscle and athletic performance. J Am Acad Orthop Surg 2018;26:278-85.
Bischoff-Ferrari HA, Giovannucci E, Willett WC, et al. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr 2006;84:18-28.
Costa EM, Blau HM, Feldman D. 1,25-dihydroxyvi- tamin D3 receptors and hormonal responses in cloned human skeletal muscle cells. Endocrinology 1986;199:2214-20.
Pojednic RM, Ceglia L. The emerging biomolecular role of vitamin D in skeletal muscle. Exerc Sport Sci Rev 2014;42:76-81.
Ross AC, Manson JE, Abrams SA, et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab 2011;96:53-8.
Alshahrani F, Aljohani N. Vitamin D: deficiency, sufficiency, and toxicity. Nutrients 2013;5:4605-16.
Schwalfenberg G. Not enough vitamin D: health consequences for Canadians. Can Fam Physician 2007;53:841-54.
Bischoff-Ferrari HA, Willet WC, Wong JB, et al. Fracture prevention with vitamin D supplementation: a meta-analysis of randomized controlled trials. JAMA 2005;293:2257-64.
Nawabi DH, Chin KF, Keen RW, et al. Vitamin D deficiency in patients with osteoarthritis undergoing total hip replacement: a cause for concern? J Bone Joint Surg Br 2010;92:496-9.
Bogunovic L, Kim AD, Beamer BS, et al. Hypovita- minosis D in patients scheduled to undergo orthopaedic surgery: a single-center analysis. J Bone Joint Surg Am 2010;92:2300-4.
Holick MF. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin Proc 2008;81:353-73.
Tangpricha V, Pearce EN, Chen TC, et al. Vitamin D insufficiency among free-living healthy young adults. Am J Med 2002;112:659-62.
Chapuy MC, Preziosi P, Maamer M, et al. Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos Int 1997;7:439-43.
Lavernia CJ, Villa JM, Iacobelli DA, et al. Vitamin D insufficiency in patients with THA: prevalence and effects on outcome. Clin Orthop Relat Res 2014; 472:681-6.
Mirghasemi SA, Trepman E, Sadeghi MS. Bone marrow edema syndrome in the foot and ankle. Foot Ankle Int 2016;37:1364-73.
Gigena LM, Chung CB, Lektrakul N, et al. Transient bone marrow edema of the talus: MR imaging findings in five patients. Skeletal Radiol 2002; 31:202-7.
Sprinchorn AE, O'Sullivan R, Beischer AD. Transient bone marrow edema of the foot and ankle and its association with reduced systemic bone mineral density. Foot Ankle Int 2011;32:508-12.
Horas K, Fraissler L, Maier G, et al. High prevalence of vitamin D deficiency in patients with bone marrow edema syndrome of the foot and ankle. Foot Ankle Int 2017;38:760-6.
Ringe JD, Dorst A, Faber H. Effective and rapid treatment of painful localized transient osteoporosis (bone marrow edema) with intravenous ibandronate. Osteoporos Int 2005;16:2063-8.
Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987;40:373-83.
Bruns J, Werner M, Soyka M. Is vitamin D insufficiency or deficiency related to the development of osteochondritis dissecans? Knee Surg Sports Traumatol Arthrosc 2016;24:1575-9.
Maier GS, Lazovic D, Maus U, et al. Vitamin D deficiency: the missing etiological factors in the development of juvenile osteochondrosis dissecans? J Pediatr Orthop 2016. [Epub ahead of print].
Houston DK, Cesari M, Ferruci L, et al. Association between vitamin D status and physical performance: the InCHANTI study. J Gerontol A Biol Sci Med Sci 2007;62:440-6.
Tomlinson PB, Joseph C, Angioni M. Effects of vitamin D supplementation on upper and lower body muscle strength levels in healthy individuals: a systematic review with meta-analysis. J Sci Med Sport 2015;18:575-80.
Endo I, Inoue D, Mitsui T, et al. Deletion of vitamin D receptor gene in mice results in abnormal skeletal muscle development with deregulated expression of myoregulatory transcription factors. Endocrinology 2003;144:5138-44.
