Following breast cancer 100K IU Vitamin D doses twice a month helped a lot – RCT
Impact of a tailored oral vitamin D supplementation regimen on serum 25-hydroxyvitamin D levels in early breast cancer patients: a randomized phase III study.
Ann Oncol. 2016 Jul;27(7):1235-41. doi: 10.1093/annonc/mdw145
Jacot W1, Firmin N2, Roca L3, Topart D4, Gallet S2, Durigova A2, Mirr S2, Abach L3, Pouderoux S2, D'Hondt V2, Bleuse JP3, Lamy PJ5, Romieu G2.
📄 Download the PDF from VitaminDWiki
100,000 IU = doubleheaded arrow
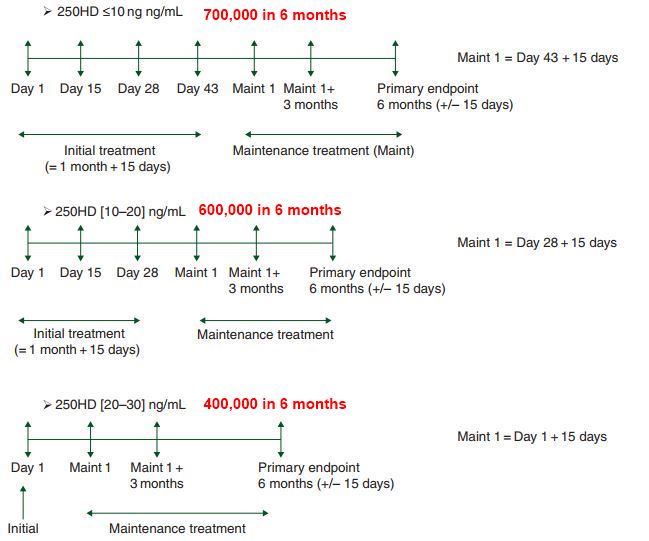
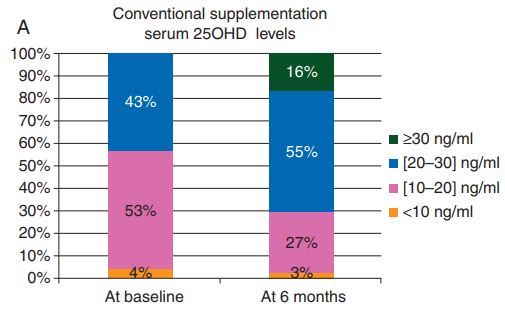
400 IU daily ==> 16% above 30ng
27% were still <20 ng
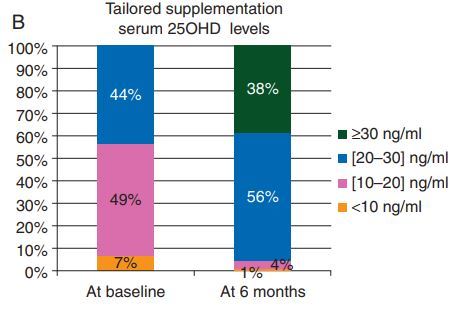
100,000 IU every two weeks(avg 7,000 IU daily) ==> 38% above 30 ng
only 4% were still <20 ng
BACKGROUND:
A minority of early breast cancer (EBC) patients treated with adjuvant or neoadjuvant chemotherapy have sufficient baseline vitamin D (vitD) level. This randomized phase III study assessed the safety and efficacy of a tailored, high-dose, oral vitD supplementation in restoring a normal 25-hydroxy vitD (25OHD) level in this population.
PATIENTS AND METHODS:
Participants received a 6-month conventional (C) vitD and calcium supplementation or a 6-month high-dose oral vitD regimen tailored on the deficiency (T) and a conventional calcium supplementation. The primary end point was the 6-month percentage of 25OHD serum level normalization.
RESULTS:
A total of 215 patients including 197 patients with vitD deficiency were recruited, and 195 patients were randomized (T, 100; C, 95). Compliance to the daily oral supplementation was 68.4% and 67% in the C and T arms, respectively. Discontinuous high-dose vitD compliance appeared higher in the T arm (77%). At 6 months, more patients presented with a normalized vitD level in the T arm (30% versus 12.6%; P = 0.003). Supplementation was well tolerated, and no significant difference in the treatment-related toxicity between the two arms was reported. Fifty-two patients without vitD normalization from the C arm switched to the T arm after 6 months. At 12 months, 44% of these patients achieved vitD normalization.
CONCLUSION:
A tailored high-dose oral vitD supplementation safely allows a higher percentage of the serum 25OHD level normalization compared with a conventional regimen in chemotherapy-treated EBC patients. As compliance to a daily oral supplementation remains poor in this setting, an adaptation of the treatment schedule is warranted.
CLINICAL TRIAL NUMBER: NCT01480869. PMID: 27029707 DOI: 10.1093/annonc/mdw145
