Vitamin D Sulfate (found in breast milk) - several studies
Summary as of March 2023 - Vitamin D3 sulfate does exist.
It is processed by many of the same genes at Vitamin D3.
The genes are not as efficient at processing the sulfate version.
Very little data exists at this time to indicate that the sulfate version is important.
It has been discussed for decades,
Patent application 2018
CHOLECALCIFEROL SULFATE SALTS AND THEIR USE FOR THE TREATMENT OF VITAMIN D DEFICIENCY
Patent Application - 20180200269
“[0009] Surprisingly, it was found that cholecalciferol sulfate salts and thus, vitamin D.sub.3 in its native form can be supplied to the body in a way without immediate metabolization thereof into water-insoluble cholecalciferol or (25OH)-cholecalciferol. The lymph system is able to distribute the water-soluble cholecalciferol sulfate salts as such in the body. The water-soluble cholecalciferol sulfate salts are of course also soluble in blood. However, there they may potentially undergo rapid, e.g. enzymatic, decomposition.
[0012] Cholecalciferol sulfate salts are usually soluble in water or an aqueous-alcoholic mixture. Examples for pharmaceutically acceptable cholecalciferol sulfate salts with an inorganic cation are
sodium cholecalciferol sulfate ,
magnesium cholecalciferol sulfate ,
calcium cholecalciferol sulfate and
ammonium cholecalciferol sulfate .
Examples for pharmaceutically acceptable cholecalciferol sulfate salts with organic cations are
trimethylammonium cholecalciferol sulfate and
L-lysine cholecalciferol sulfate
[0013] The method of administration in accordance with the present invention is chosen such that it effects that at least 5% of the administrated amount of cholecalciferol sulfate salt is transported from the site of administration into a systemic fluid transport system of the human body without metabolization. Depending on the method of administration, preferably at least 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45% or 50% of the administered cholecalciferol sulfate salt reach a systemic fluid transport system of the human body from the site of administration as such without experiencing metabolization.
Transported by Blood and/orLymph
Synthesis and biological activity of vitamin D3-sulfate
J Biol Chem . 1981 Jan 25;256(2):823-6.
L E Reeve, H F DeLuca, H K Schnoes
Vitamin D3-3 beta-sulfate has been synthesized using pyridine sulfur trioxide as the sulfate donor. It has been shown to be pure by high performance liquid chromatography and spectral methods. Unlike previous reports, the product has been identified unambiguously as the 3 beta-sulfate ester of vitamin D3 by its ultraviolet, nuclear magnetic resonance, infrared, and mass spectra. The biological activity of vitamin D3-sulfate was then determined in vitamin D-deficient rats. Vitamin D3-sulfate has less than 5% of the activity of vitamin D3 to mobilize calcium from bone and approximately 1% of the ability of vitamin D3 to stimulate calcium transport, elevate serum phosphorus, or support bone calcification. These results disprove previous claims that vitamin D3-sulfate has potent biological activity, and they further do not support the contention that vitamin D-sulfate represents a potent water-soluble form of vitamin D in milk.
📄 Download the PDF from VitaminDWiki
Sunlight and Vitamin D: They're Not the Same Thing! - Seneff (March 2021?)
Foundation for Alternative and Integrative Medicine
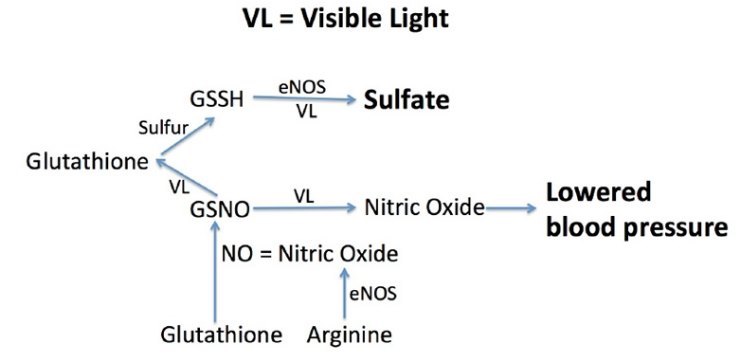
- "Those who are familiar with my research will know that I believe that keratinocytes in the skin, endothelial cells lining the walls of surface veins, and red blood cells are able to exploit the energy in sunlight by oxidizing hydrogen sulfide to make sulfate.3 In the skin, the sulfate is conjugated with both vitamin D and cholesterol, and this makes these otherwise water-insoluble molecules water soluble."
Vitamin D SULFATE - a lesser form, might be important for CVD - Masterjohn Feb 2023
Harnessing the Power of Nutrients Substack
"Several roles of cholesterol sulfate are noted, but no discussion of direct comparisons with cholesterol or with sulfate, making it unclear how important the specific compound cholesterols sulfate is."
"It is a worthy hypothesis but does not have clear evidence supporting it as a conclusion."
4 - Biochemistry of Human Milk (behind paywall) - 2022
Breastfeeding (Ninth Edition) A Guide for the Medical Profession 2022, Pages 93-144
Vitamin D3 sulfate and Vitamin D genes – March 2023
Analysis of the ability of vitamin D3-metabolizing cytochromes P450 to act on vitamin D3 sulfate and 25-hydroxyvitamin D3 3-sulfate
The Journal of Steroid Biochemistry and Molecular Biology Volume 227, March 2023, https://doi.org/10.1016/j.jsbmb.2022.106229
Robert C. Tuckey, Chloe Y.S. Cheng, Lei Li, Yuhan Jiang
Abstract
25-Hydroxyvitamin D3 (25(OH)D3) is present in the human circulation esterified to sulfate with some studies showing that 25(OH)D3 3-sulfate levels are almost as high as unconjugated 25(OH)D3. Vitamin D3 is also present in human serum in the sulfated form as are other metabolites. Our aim was to determine whether sulfated forms of vitamin D3 and vitamin D3 metabolites can be acted on by vitamin D-metabolizing cytochromes P450 (CYPs), one of which (CYP11A1) is known to act on cholesterol sulfate . We used purified, bacterially expressed CYPs to test if they could act on the sulfated forms of their natural substrates. Purified CYP27A1 converted vitamin D3 sulfate to 25(OH)D3 3-sulfate with a catalytic efficiency (kcat/Km) approximately half that for the conversion of vitamin D3 to 25(OH)D3. Similarly, the rate of metabolism of vitamin D3 sulfate was half that of vitamin D3 for CYP27A1 in rat liver mitochondria. CYP2R1 which is also a vitamin D 25-hydroxylase did not act on vitamin D3 sulfate. CYP11A1 was able to convert vitamin D3 sulfate to 20(OH)D3 3-sulfate but at a considerably lower rate than for conversion of vitamin D3 to 20(OH)D3. 25(OH)D3 3-sulfate was not metabolized by the activating enzyme, CYP27B1, nor by the inactivating enzyme, CYP24A1. Thus, we conclude that 25(OH)D3 3-sulfate in the circulation may act as a pool of metabolically inactive vitamin D3 to be released by hydrolysis at times of need whereas vitamin D3 sulfate can be metabolized in a similar manner to free vitamin D3 by CYP27A1 and to a lesser degree by CYP11A1.
Introduction
Vitamin D and its metabolites are present in humans not only in their free form but also as esters. These conjugated forms include sulfates, glucuronides and fatty acid esters [1], [2], [3]. The detection of 25-hydroxyvitamin D3 3-sulfate (25(OH)D3 3-sulfate) in human plasma was first reported in 1985 [4] and it is now well established that it is present in the human circulation at appreciable concentrations, often almost as high as 25(OH)D3 [1], [2], [4], [5], [6], [7], [8], [9]. The plasma of newborns has a higher concentration of 25(OH)D3 3-sulfate than 25(OH)D3 [7], [8], [9]. Recently, Jenkinson et al. [10] reported that the proportion of conjugated 25(OH)D3 was 46–48% of the total in men over the age of 70 both with and without a vitamin D3 supplement. The proportion of sulfate was much higher than the glucuronide with the serum 25(OH)D3 3-sulfate levels averaging 50–55 nM. Some vitamin D3 is also present as the sulfate with a mean serum concentration of 0.7 nM being reported [5]. Conjugated forms of 25-hydroxyvitamin D2, 24,25-dihydroxyvitamin D3 and 3-epi-25-hydroxyvitamin D3 are also present in proportions ranging from 30% to 60% of the total in human serum from men over 70, being highest for 24,25-dihydroxyvitamin D3 [10].
The enzymes responsible for the formation of secosteroid sulfates belong to the sulfotransferases superfamily of enzymes which are responsible for sulfation of steroids, neurotransmitters, drugs and xenobiotic compounds [11]. Sulfating activity on 25(OH)D3 has been found in the liver, kidney, lung and small intestine, and on D3 in the liver. In humans, SULT1A1, SULT2A1, SULT2B1a and SULT2B1b have been shown to sulfate vitamin D3-related compounds [11]. SULT2A1 is believed to be the most important sulfotransferases as it can sulfate 7-DHC, vitamin D3, 25(OH)D3 and 1,25(OH)2D3 [11], [12]. SULT2B1a and SULT2B1b can only use 7-DHC as a substrate and SULT1A1 shows activity only on 1,25(OH)2D3 [11].
7-Dehydrocholesterol (7DHC), the precursor vitamin D3, is present in the human skin where it is converted to vitamin D3 after exposure to UVB radiation [13], [14]. The subsequent major pathways of vitamin D3 activation and inactivation by CYPs are shown in Fig. 1. Vitamin D3 is transferred from the skin to the liver via the bloodstream primarily bound to vitamin D binding protein, and undergoes 25-hydroxylation by CYP27A1 or CYP2R1 to form 25(OH)D3 [1], [15], [16]. 25(OH)D3 can be further hydroxylated at C-1a by CYP27B1 in the kidney to produce 1a,25-dihydroxyvitamin D3 (1,25(OH)2D3) [1], [15], [16]. 1,25(OH)2D3 has a high binding affinity for the vitamin D receptor and is the major hormonal form of vitamin D which regulates the transcription of many target genes [1], [17], [18], [19], [20]. These include genes responsible for calcium and phosphate homeostasis [19] as well as many genes involved in non Musculo-skeletal actions such as inhibiting cell proliferation, promoting differentiation, inhibiting inflammation and modulating the immune system [1], [20]. CYP24A1 catalyzes the inactivation of 1,25(OH)2D3 by oxidizing its side chain, ultimately producing the excretion product, calcitroic acid [1], [16], [21]. The side chain of 25(OH)D3 is similarly oxidized by CYP24A1, with the first major intermediate being 24R,25-dihydroxyvitamin D3 (24,25(OH)2D3) (Fig. 1) [1], [16], [21].
An alternative pathway of vitamin D3 activation is catalyzed by CYP11A1 which produces biologically active metabolites with low calcemic activity such as 20S-hydroxyvitamin D3 (20(OH)D3) and 20S,23S-dihydroxyvitamin D3 (20,23(OH)2D3) [1], [22], [23], [24], [25], [26], [27]. CYP11A1 also catalyzes the first step in steroid biosynthesis, the oxidation of the cholesterol side chain to produce pregnenolone [1], [27]. As well as cholesterol, CYP11A1 cleaves the side chain of cholesterol sulfate [28], [29], indicating that the presence of a sulfate group in the 3ß-position does not prevent catalysis. We therefore analyzed the ability of all the major vitamin D metabolizing CYPs, CYP27A1, CYP2R1, CYP27B1, CYP24A1 and CYP11A1 to act on the sulfated forms of their substrates to test the effect of sulfation on the activation and inactivation of vitamin D and its metabolites.
Section snippets
Materials
The bacterial expression and purification of vitamin D-metabolizing CYPs was carried out using our previously described procedures for human CYP27A1 [30], human CYP2R1 [31], mouse and human CYP27B1 [32], [33], human CYP24A1 [34] and human and bovine CYP11A1 [26], [35], [36]. Human adrenodoxin [37], adrenodoxin reductase [26] and NADPH-P450 oxidoreductase [31] were also expressed and purified as before. Vitamin D3, 25-hydroxyvitamin D3 (25(OH)D3), vitamin D3 sulfate and 25-hydroxyvitamin D3 . . . .
Metabolism of vitamin D3 sulfate and vitamin D3 by CYP27A1
CYP27A1 can oxidize a number of sterols acting as a 27- and/or 25-hydroxylase [30], [38], [43], [44], [45]. It acts as a 25-hydroxylase on vitamin D3 and contributes to the initial activation of vitamin D3 to 25-hydroxyvitamin D3 [1], [16], [17], [46]. To test its ability to act on vitamin D3 sulfate, the sulfate was dissolved in 0.45% HP-ß-CD, and incubated with human CYP27A1 (Fig. 2 A, B). This resulted in the production of one major and several minor products that were not present in the . . . .
Discussion
This study shows that of the three CYPs that can metabolize vitamin D3, both CYP27A1 and CYP11A1 can also metabolize vitamin D3 sulfate, but no appreciable metabolism of the conjugate was observed for CYP2R1. The extent of metabolism of vitamin D3 sulfate by CYP11A1 was lower than for free vitamin D3, more so for the human enzyme than the bovine enzyme. Previously, it was reported that human CYP11A1 can metabolize cholesterol sulfate to pregnenolone sulfate with a comparable kcat to free . . .
Title change made April 2022 caused the visitor count to reset.
There have actually been visitors to this page since it was originally made
