Mouthwashes temporarily reduce COVID infectiousness - many studies
100's of studies have documented that Iodine reduces COVID infectiousness in dental offices
Povidone Iodine Mouthwash, Gargle, and Nasal Spray to Reduce Nasopharyngeal Viral Load in Patients With COVID-19 - A Randomized Clinical Trial Feb 2021
JAMA Otolaryngol Head Neck Surg. 2021;147(4):400-401. doi:10.1001/jamaoto.2020.5490
Jeremy Guenezan, MD1; Magali Garcia, MD, PhD2; Deidre Strasters, MD1; et alClément Jousselin, PhD2; Nicolas Lévêque, PharmD, PhD2; Denis Frasca, MD, PhD3; Olivier Mimoz, MD, PhD1
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is primarily transmitted person-to-person through the aerosolization of droplets containing contaminated nasopharyngeal secretions.1 Povidone iodine (PI) solutions at concentrations as low as 0.5% rapidly inactivate SARS-CoV-2 in vitro with contact times as short as 15 seconds.2 We investigated whether nasopharyngeal application of PI could reduce the viral load of patients with nonsevere coronavirus disease 2019 (COVID-19) symptoms.
Methods
We included adult outpatients (≥18 years old) having tested highly positive (cycle threshold ≤20) for SARS-CoV-2 ribonucleic acid (RNA) by reverse transcription-polymerase chain reaction (RT-PCR) in nasopharyngeal swabs within the previous 48 hours. Patients with a history of thyroid disease were excluded. The study was approved by the South Mediterranean Institutional Review Board, and written informed consent was obtained from all participants. The trial protocol is included in Supplement 1.
Patients underwent an additional nasopharyngeal swab for viral quantification at baseline before being randomly assigned (1:1) to a control group (no intervention, n = 12) or an intervention group (n = 12). Intervention consisted of 4 successive mouthwashes and gargles with 25 mL of 1% aqueous PI solution each (Mylan, Merignac, France), followed by one 2.5-mL nasal pulverization of the same solution into each nostril using an intranasal mucosal atomization device (MAD Nasal, Teleflex, Morrisville, North Carolina) connected to a 5-mL syringe while sniffing and 1 application on each nasal mucosa of a dab of 10% PI ointment followed by a massage of the nostril to help spread the ointment. Patients were trained during the first decolonization session, and received the necessary materials and a guide to help them perform the following sessions 4 times a day for 5 days.
Follow-up was done on day 1 and then every 2 days until day 7 to assess the efficacy (viral quantification) and safety of the decolonization. Almost all (>95%) of the nasopharyngeal swabs were taken by the same skilled nurse at least 3 hours after the last PI application for quantification of viral RNA using RT-PCR,3 and viral titer using the dilution limit method on Vero cells and the Spearman-Karber approach with a limit of detection of 102.5 tissue culture infectious dose (TCID50) per mL.4 Changes in viral load over time were compared between study groups using a linear mixed model for repeated measures.
Results
All patients completed the study, and none required hospital admission. Compared with the control group, patients in the intervention group were younger (Table). Median (IQR) age of patients in the control group was 57 (45-68) years and in the intervention group was 33 (23-46) years. A total of 4 patients (33%) in each group were male. All patients but 1 had negative viral titer by day 3. Use of PI had no influence on changes of viral RNA quantification over time (Figure). Mean relative difference in viral titers between baseline and day 1 was 75% (95% CI, 43%-95%) in the intervention group and 32% (95% CI, 10%-65%) in the control group (Figure). All patients exposed to PI experienced unpleasant nasal tingling but completed the study. Thyroid stimulating hormone elevation (median [IQR], 3.4 [2.6-4.3] mIU/L vs 2.1 [1.4-3.1] mIU/L at baseline) was observed in all patients after 5 days of PI exposure, exceeding the upper normal value in 5 patients, with a return to baseline values 7 to 12 days later. No modification in thyroid hormone (T3, T4) or creatinine levels was observed.
Discussion
Nasopharyngeal decolonization may reduce the carriage of infectious SARS-CoV-2 in adults with mild to moderate COVID-19.5 Thyroid dysfunction occurred in 42% of the patients exposed to PI, with spontaneous resolution upon treatment discontinuation, as previously reported.6 Strengths of this study include assessment of viral titer to determine whether the virus was viable and thus potentially transmissible. Limitations include the small number of patients and the single-center design. These data call for a larger clinical trial to confirm the benefit of PI in limiting the excretion and resulting human-to-human transmission of SARS-CoV-2, using lower PI concentrations to minimize adverse effects.
📄 Download the PDF from VitaminDWiki
Differential Effects of Antiseptic Mouth Rinses on SARS-CoV-2 Infectivity In Vitro - March 2021
Pathogens 2021, 10(3), 272; https://doi.org/10.3390/pathogens10030272
by Chuan Xu 1,Annie Wang 1,Eileen R. Hoskin 2,Carla Cugini 3ORCID,Kenneth Markowitz 3,Theresa L. Chang 1,4,ORCID andDaniel H. Fine 3,
1 Public Health Research Institute, Rutgers, New Jersey Medical School, 225 Warren Street, Newark, NJ 07103, USA
2 Department of Restorative Dentistry, School of Dental Medicine, Rutgers, The State University of New Jersey, Newark, NJ 07103, USA
3 Department of Oral Biology, School of Dental Medicine, Rutgers, The State University of New Jersey, Newark, NJ 07103, USA
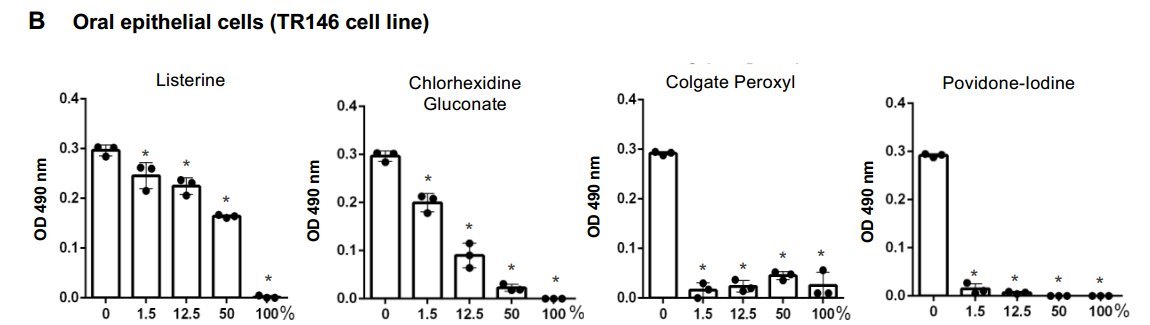
Severe acute respiratory syndrome-related coronavirus (SARS-CoV-2) is detectable in saliva from asymptomatic individuals, suggesting a potential benefit from the use of mouth rinses to suppress viral load and reduce virus spread. Published studies on the reduction of SARS-CoV-2-induced cytotoxic effects by mouth rinses do not exclude antiseptic mouth rinse-associated cytotoxicity. Here, we determined the effect of commercially available mouth rinses and antiseptic povidone-iodine on the infectivity of replication-competent SARS-CoV-2 viruses and of pseudotyped SARS-CoV-2 viruses. We first determined the effect of mouth rinses on cell viability to ensure that antiviral activity was not a consequence of mouth rinse-induced cytotoxicity. Colgate Peroxyl (hydrogen peroxide) exhibited the most cytotoxicity, followed by povidone-iodine, chlorhexidine gluconate (CHG), and Listerine (essential oils and alcohol). The potent antiviral activities of Colgate Peroxyl mouth rinse and povidone-iodine were the consequence of rinse-mediated cellular damage when the products were present during infection. The potency of CHG was greater when the product was not washed off after virus attachment, suggesting that the prolonged effect of mouth rinses on cells impacts the antiviral outcome. To minimalize mouth rinse-associated cytotoxicity, mouth rinse was largely removed from treated viruses by centrifugation prior to infection of cells. A 5% (v/v) dilution of Colgate Peroxyl or povidone-iodine completely blocked viral infectivity. A similar 5% (v/v) dilution of Listerine or CHG had a moderate suppressive effect on the virus, but a 50% (v/v) dilution of Listerine or CHG blocked viral infectivity completely. Mouth rinses inactivated the virus without prolonged incubation. The new infectivity assay, with limited impacts of mouth rinse-associated cytotoxicity, showed the differential effects of mouth rinses on SARS-CoV-2 infection. Our results indicate that mouth rinses can significantly reduce virus infectivity, suggesting a potential benefit for reducing SARS-CoV-2 spread.
📄 Download the PDF from VitaminDWiki
The Use of Povidone Iodine Nasal Spray and Mouthwash During the Current COVID-19 Pandemic May Reduce Cross Infection and Protect Healthcare Workers - Aug 2021
Preprint
Justin Kirk-Bayley Royal Surrey County Hospital
San Sunkaraneni Royal Surrey County Hospital
Stephen Challacombe King's College London
In late 2019 a novel coronavirus, SARS-CoV-2 causing Coronavirus disease 2019 (COVID-19) appeared in Wuhan China, and on 11th March 2020 the World Health Organisation declared it to have developed pandemic status. In early SARS-CoV-2 infection, viral titres of greater than 107/mL in saliva and nasal mucous can be found; minimisation of these titres should help to reduce cross-infection. Povidone-iodine (PVP-I) disinfectant has better anti-viral activity than other antiseptics and has already been proven to be an extremely effective virucide in vitro against severe acute respiratory syndrome and Middle East respiratory syndrome coronaviruses (SARS-CoV and MERS-CoV). Its in vivo virucidal activity is unknown, but it retains its antimicrobial activity against bacteria in vivo intraorally and one application can reduce oral microbial flora for greater than 3 hours.
PVP-I disinfectant has been shown to be safe when administered to the nasal cavity and as a mouthwash. We propose a protocolised intra-nasal and oral application of PVP-I for both patients and their attendant healthcare workers (HCWs) during the current COVID-19 pandemic to help limit the spread of SARS-CoV-2 from patients to healthcare workers and vice versa. The aim is to reduce the viral ‘load’ in two of the key areas from where droplets and aerosols containing the virus are expectorated (the lower respiratory tract being the other). The aim of use in HCWs is to destroy virus that has entered the upper aerodigestive tract before it has the opportunity to infect the host.
We suggest the protocol is considered for routine use during the care of COVID-19 patients, particularly before any procedure that involves the upper aerodigestive tract, including intubation, nasal and oral procedures, endoscopy and bronchoscopy. We suggest it should be considered when such procedures are carried out in all patients during the pandemic regardless of COVID-19 status, due to the reported significant rates of asymptomatic infection
The total iodine exposure proposed is well within previously recorded safe limits in those without contraindications to its use. The intervention is inexpensive, low risk and potentially easy to deploy at scale globally.
We acknowledge that the proposal we present extrapolates in vitro finding into the in vivo setting and that assumptions are made that under normal circumstances we would confirm with in vivo data prior to recommendations for use. However, given the strength of in vitro evidence and the low risk, minimal cost and global applicability of the proposed intervention, which amounts to disinfection of the oro/nasal cavities, we feel that there is little to lose and potentially much to gain.
📄 Download the PDF from VitaminDWiki
A randomized trial on the regular use of potent mouthwash in COVID-19 treatment - April 2021
Preprint doi: https://doi.org/10.1101/2020.11.27.20234997
Khalid Mukhtar, Suelen Qassim, Shaikha Ali Al Qahtani, Mohamed Ibn-Masud Danjuma, Mohamed Mohamedali, Housamaddeen Al Farhan, Mohammed F. Khudair, Abdel Rehim El Tayeh, Mohammed Al-Dosari, Mohamed Elhassan Babiker, Ahmed Hassib, Rumaisa Mohamed Elmustafa, Wesal Elhadary, Morwan Abdulkarim, Rajvir Singh, Muna Al.Maslamani
In this work we tried to study the effect of the regular use of potent mouthwash in COVID19 cases, on the premise that it may speedup the recovery, through the repeated reduction of microbial load, of both, the 2019-nCOV and oral microbiota; thus slowing the disease progression and lowering the incidence of superinfections.
Through a randomized controlled trial, a mixed solution of Hydrogen peroxide 2% and chlorhexidine gluconate, to be used for oral rinsing and gargling three times daily, was tested in cases admitted to COVID treatment facility, versus the standard (only) COVID19-treatment protocol, starting with 46 cases in each group, matched in terms of disease severity, of symptoms, and average cycle threshold value (CT-value) for the COVID PCR test on diagnosis.
Our findings showed statistically significant improvement in terms of a higher conversion rate to “COVID19-negative PCR” by five days of treatment (6/46 Vs 0/46), improvement in “symptoms severity” after two days of treatment, and less intubation and mortality (0/46 Vs 3/46) with all P-value < 0.05. There was also a trend of improvement in other outcome variables, though with no statistically significant difference; namely “shorter hospital stays,” “less progression in Oxygen requirements”, “less rate of plasma transfusion”, and better “gross extent of improvement”.
Our findings support a beneficial role in treating active cases (Disease) and anticipates better outcome should implemented earlier in course of the disease; thus, suggest a role in limiting the spread (Pandemic), as an additional preventive method. Additionally, we think the repeated reduction in the microbial load might have been sufficient to induce a strain in a possible viral-microbial interaction, resulting in slowing down of the disease progress.
Introduction / Background
The current treatment policies for COVID19 are rather “supportive”, including simultaneous approaches to maintain adequate oxygen supply, and preventing secondary infections, in addition to targeting the immunity itself, to optimize the outcome, and prevents overt reactions.
Considering the risk of exposure; the oral route is likely to pose and equal, if not a higher risk than the nasal; given the reported GIT symptoms in some cases, the lack of natural filtering capacity and the protection provided by the high levels of nitrous oxide produced (1, 2), which proved to inhibit viral replication, (3)
An existing state of dysbiosis reflects an impaired immune response, hence can facilitate developing the disease and subsequently the progression to worse outcomes.
It follow thus, the regular use of a potent mouthwash, that can consistently reduce the oral microbial load, including 2019-nCOV, aiding a faster recovery, as the immune systems is likely to struggle less to overcome the infection and consequently reduces the infection transmission rate. Additionally, this should lower the potential risk of other microbial superinfections. However, the sought benefits could also be achieved through another mechanism; invoking the evolutionary game theory, 2019-nCOV might be relying on an accomplice; be it a specific organism (e.g. bacterial species) or the state of dysbiosis in general, as the repeated and consistent reduction in the microbial load -oral microbiota and 2019-nCOV-might be sufficient to induce a strain in the viral-microbial interaction, inhibit reciprocal altruism, and negatively affecting the survival chance. The current literature indicates that 2019-nCOV is utilizing ACE2 receptors as access to the target cells (4), demonstrating the ability to induce both its upregulation through interferon-gene stimulation (5) possibly as a mean to increase its own replication, as well as down-regulation through direct binding (6) with effects that alter the natural lungs and gut microbiota in the direction of dysbiosis (7, 8), and an increased bacterial co-infection risk (9-16). ACE2 expression is identified in sites linked to the virus’s isolation and the disease complications, including the vascular endothelium, the respiratory system (10), and the intestines.
Additionally, both ACE and ACE2 have a structural similarity within their active-site region to other metalloproteases in some bacteria, including some of the URT pathogens; such as the M32 carboxypeptidase from the Bacillus subtilis (11), a probiotic and a possible agent of dental caries, and Paenibacillus sp. B38 with demonstrated ability to lower angiotensin II levels in mice (12), suggesting another possible direct viral -bacterial interaction. Thus, ACE2 can be utilized as a medium for microbial-viral interaction, and can be dialed up or down to maintain reciprocal altruism, especially among species with the potential to benefit from dysbiosis, as each side can affect the other, either directly or through inducing the host’s immune response.
Mouth Wash and oral Microbiota
Chlorhexidine mouthwash (CHX) (0.12%-0.24%) is frequently used in dental practice, as it has a beneficial effect on controlling bacterial (13-17)(20-23) overgrowth, while the hydrogen peroxide (HPX) at a concentration of 3% demonstrated ability to reduce the viral load on surfaces by >4-log when applied for one-minute (17). HPX is produced naturally by both; the epithelial cells via superoxide dismutase enzyme, releasing ion superoxide, and by the oral bacteria itself, in effect to maintain the oral microecology(18, 19) through inducing oxidative stress similar to those triggered by viral infections; that stimulates a local innate response.(20) However, the combination with hydrogen peroxide (HPX) (1.5%-3%) proved to have a better outcome among users while maintaining the antibacterial efficiency as for CHX alone(37-39) within two weeks of use, even without specific dental hygiene instructions (40), as well as to control ventilator-associated pneumonia (VAP) measure (41, 42). Thus, this combination may have a great potential in controlling COVID19 (21), as it can induce an anti-viral response before the actual recognition of the viral antigens by the host immune cells.
📄 Download the PDF from VitaminDWiki
