Vitamin D and Primary Hyperparathyroidism – 2011, 2016
Vitamin D and primary hyperparathyroidism: more insights into a complex relationship - Nov 2016
📄 Download the PDF from VitaminDWiki
Clinical Significance of Vitamin D Deficiency in Primary Hyperparathyroidism, and Safety of Vitamin D Therapy - 2011
Nasser Mikhail, MD, MSc
From Southern Medical Journal
Posted: 01/24/2011; South Med J. 2011;104(1):29-33. © 2011 Lippincott Williams & Wilkins
from Medscape
Vitamin D deficiency occurs more frequently in patients with primary hyperparathyroidism (PHPT) compared with the general population, and is usually associated with an aggravated form of the disease. Current guidelines recommend measurement of serum levels of 25-hydroxy vitamin D (25-OHD) in all patients with PHPT, and their repletion if the levels are less than 50 mmol/L (20 ng/mL). Limited data suggest that vitamin D treatment is generally safe in subjects with mild PHPT and coexisting vitamin D deficiency. Adverse effects include hypercalcuria and, less commonly, exacerbation of hypercalcemia. Well-designed trials are needed to evaluate the safety of vitamin D replacement therapy in a wide spectrum of patients with concomitant PHPT and vitamin D deficiency. These trials should address the impact of such therapy on the complications and course of PHPT.
Key Points
Vitamin D inadequacy is more common in patients with primary hyperparathyroidism than in the general population.
Hypovitaminosis D is generally associated with an aggravated biochemical profile of primary hyperparathyroidism.
Initiation of vitamin D therapy is recommended in patients with serum levels of 25-hydroxy vitamin D <20 ng/mL and concomitant hyperparathyroidism.
Well-designed trials are needed to better define the safety and efficacy of vitamin D therapy in patients with concomitant vitamin D deficiency and primary hyperparathyroidism.
Introduction
The panel of the Third International Workshop on Asymptomatic Primary Hyperparathyroidism recently recommended measurement of serum concentrations of 25-hydroxy vitamin D (25-OHD)—the best available test of vitamin D nutritional status—in all patients with suspected primary hyperparathyroidism (PHPT), and initiation of vitamin D supplementation in patients with levels below 20 ng/mL before making any medical or surgical decisions.[1] The panel guidelines were based on the following observations: 1) The high prevalence of vitamin D deficiency in patients with PHPT; 2) vitamin D inadequacy may worsen the clinical picture of PHPT; 3) in some patients with PHPT, vitamin D deficiency can mask hypercalcemia, and therefore obscure the diagnosis; 4) available evidence, although limited, suggests that vitamin D therapy in vitamin D-deficient patients with PHPT does not generally increase serum calcium levels. While the definition of vitamin D deficiency in the general population is still a matter of debate, the panel selected serum levels of 25-OHD <20 ng/mL as the cutoff for initiation of vitamin D therapy. This threshold was based on a study that showed circulating parathyroid hormone (PTH) levels could not be suppressed further when serum levels of 25-OHD are maintained above 20 ng/mL in healthy subjects who received vitamin D2 and calcium supplements for eight weeks.[2] Meanwhile, many physicians are reluctant to prescribe vitamin D to patients with PHPT, due to concern of worsening existing hypercalcemia and lack of sufficient data to support the safety and benefit of such intervention. Indeed, early case reports suggested that patients with PHPT and clinical evidence of vitamin D deficiency (manifested as osteomalacia) had their serum calcium levels increased from 1 to 2 mg/dL after vitamin D administration.[3,4]
Prevalence of Vitamin D Deficiency in Primary Hyperparathyroidism
The majority of prevalence studies of vitamin D deficiency in patients with PHPT are limited by lack of adequate control subjects, and by failure to report serum 25-OHD levels as a function of the season of the year (a factor that can significantly alter serum concentrations of 25-OHD).[5,6] Despite these limitations, available data worldwide suggest that the prevalence of vitamin D deficiency among patients with PHPT is higher than in the general population. In one of the few controlled studies, Moosgaard et al[5] from Denmark reported that 81% of 289 patients with PHPT had vitamin D deficiency (defined as 25-OHD serum levels <25 ng/mL) compared with 60% of age-, sex-, and season-matched subjects referred for osteoporosis evaluation, and 35% of healthy blood donors. In France, Boudou et al[7] found that 93% of 72 patients with PHPT had serum 25-OHD values <20 ng/dL, compared with the 38% prevalence previously reported among French postmenopausal women.[6] In the United States (US), the frequencies of patients with PHPT having serum 25-OHD levels <20 ng/mL and <25 ng/mL were 53% and 61%, respectively.[8,9] The previous proportions are higher than the most recent statistics released by the National Health and Nutrition Examination Survey (NHANES, 2001–2004) suggesting that 36% of the US population have serum 25-OHD levels <20 ng/mL.[10] The causes of low circulating levels of 25-OHD in patients with PHPT are not totally understood. Proposed mechanisms include accelerated conversion of 25-OHD to 1,25-dihydroxy vitamin D (1,25-(OH)2D) by PTH, and the increased catabolism of 25-OHD by the high levels of 1,25-(OH)2D that prevail in PHPT.[11,12] In addition, the increased adiposity described in patients with PHPT might be a contributing factor, due in part to sequestration of vitamin D in fatty tissue.[13,14]
Characteristics of Primary Hyperparathyroidism Complicated by Vitamin D Deficiency
Overall, hypovitaminosis D is associated with a more severe form of PHPT in terms of biochemical and possibly bony features. This observation is not unexpected, because vitamin D deficiency per se is known to stimulate PTH secretion, causing secondary hyperparathyroidism which may be additive to the pre-existing PHPT.[15] Retrospective studies from the US showed that patients with PHPT having vitamin D deficiency, defined as serum 25-OHD levels <15 ng/mL, <20 ng/mL, or <25 ng/mL had higher circulating levels of PTH, calcium, and alkaline phosphatase, and greater parathyroid adenoma weight compared with their vitamin D-sufficient counterparts.[9,16–18] Moreover, low serum levels of 25-OHD were shown to be related to persistent elevation of PTH after parathyroidectomy in some, but not all studies.[17–19]
Data regarding the relationship between serum 25-OHD values and bony manifestations of PHPT were inconsistent. In a series of 243 consecutive patients with PHPT, Moosgard et al[20] found that low serum levels of 25-OHD were associated with decreased bone mineral density (BMD) in the forearms, femoral neck, and whole body, independently of PTH levels. Conversely, in a smaller cross-sectional study, Carnevale et al [21] did not find a significant correlation between 25-OHD plasma levels and BMD at the lumbar spine and femoral neck in 62 women with PHPT. While no significant association was found between serum levels of 25-OHD and the prevalence of fractures in the cross-sectional study of Moosgard et al,[20] one retrospective study from Sweden showed that low serum levels of 25-OHD3 in the 10-year period preceding parathyroidectomy were an independent risk factor for the occurrence of fractures.[22] With respect to the possible link between vitamin D deficiency and renal complications of PHPT, available data suggest no significant difference in urinary calcium excretion or in the occurrence of kidney stones between patients with and without vitamin D deficiency.[5,17]
Diagnostic Importance of Normalization of Vitamin D Status
A laboratory abnormality frequently encountered during the work up for osteoporosis is the presence of high serum PTH levels coupled with repeatedly normal serum calcium (both total and ionized) concentrations. In this setting, measurement of circulating 25-OHD levels (and their repletion if low) will aid in distinction between the following three conditions:
Secondary hyperparathyroidism (HPT). In this case, PTH values will return to normal range with replenishment of vitamin D stores, while calcium levels remain within normal limits.[15]
Concomitant PHPT and vitamin D deficiency. In some cases of PHPT, hypercalcemia may be masked by concomitant vitamin D deficiency. Thus, adequate supplementation of vitamin D will lead to emergence of frank hypercalcemia with persistence of elevated PTH levels; ie, vitamin D therapy uncovers the diagnosis of PHPT.[1]
Normocalcemic HPT. This variant of PHPT is characterized by high serum PTH, but consistently normal plasma calcium concentrations after the exclusion of all causes of secondary HPT, including vitamin D deficiency.[23] If normocalcemic HPT coexists with vitamin D deficiency, PTH levels remain elevated, and calcium values remain within normal limits even after the correction of 25-OHD levels.[15]
Effects of Vitamin D Supplementation in Concomitant Primary Hyperparathyroidism and Vitamin D Deficiency
Review of the literature up to July 2010 revealed five studies that evaluated vitamin D therapy in patients with concomitant hypovitaminosis D and PHPT (Table).[1[5,24–27] Patients included in these trials had mild, mostly asymptomatic PHPT with serum calcium levels <12 mg/dL. Other important limitations include lack of randomization and, except in one study, absence of control subjects.[27] The overall results suggested that vitamin D supplementation in the setting of mild PHPT was safe. Meanwhile, the most common adverse effect of vitamin D therapy appeared to be an increase in urinary calcium, which reached statistical significance in two studies.[15,26] A fairly consistent finding after vitamin D therapy in the five studies was a mild decrease in serum PTH levels with mean reductions ranging from 3% to 25%, implying the coexistence of mild secondary hyperparathyroidism on top of PHPT.[15]
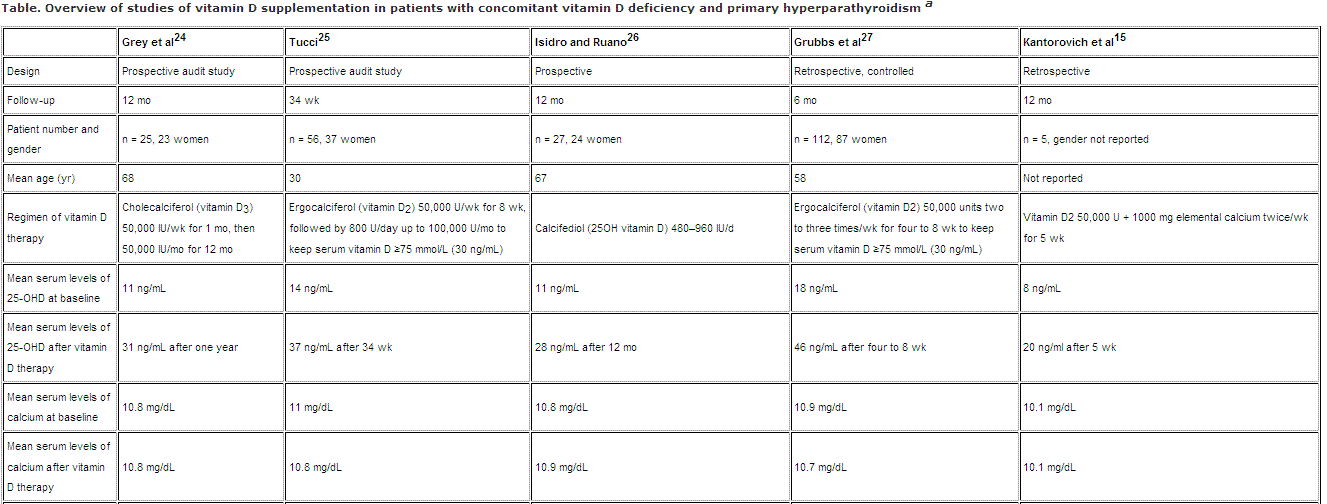
Two of the five studies evaluated the impact of vitamin D therapy on BMD. The first did not record significant effect on BMD in the lumbar spine and femoral neck in a subgroup of 12 patients after 12 months of vitamin D administration.[24] In the second study there was a significant increase in BMD at the lumbar spine and femoral neck in five patients who received calcium and vitamin D for five weeks.[1[5] The retrospective data of Grubbs et al[27] suggested that vitamin D replacement prior to parathyroidectomy had no significant effect on cure rates, frequency of recurrent disease, or occurrence of eucalcemic PTH elevation after parathyroidectomy.
Current Needs
There is an urgent need for well-designed trials to evaluate the safety and efficacy of vitamin D therapy in patients with coexistent vitamin D deficiency and PHPT. Subjects with serum calcium concentrations greater than 12 mg/dL have not been studied before, and should be included in new trials. In addition to the effects of vitamin D on the biochemical and hormonal profiles of PHPT, study outcomes should assess the impact of vitamin D on symptoms and complications of PHPT. While some differences still exist between various methods of measurement of 25-OHD in serum, recent data reported by the International Vitamin D External Quality Assessment Scheme (DEQAS) showed improvement in the precision of various assays of serum 25-OHD in the last 15 years.[28] This encouraging step should enhance the research field of vitamin D, and will help determine optimum serum levels of 25-OHD in patients with PHPT.
Conclusion
In PHPT, vitamin D inadequacy is a common feature that is usually associated with advanced disease phenotype, at least at the biochemical level. In addition, vitamin D deficiency can lead to diagnostic confusion in patients presenting with high serum PTH, but normal calcium concentrations. Vitamin D therapy is indicated in patients with PHPT having serum levels of 25-OHD <20 ng/mL. Close monitoring of serum and urine calcium levels is essential, as limited data suggest that those levels may increase in some patients in response to vitamin D replacement therapy.
References
Eastell R, Arnold A, Brandi ML, et al. Diagnosis of asymptomatic primary hyperparathyroidism: proceedings of the third international workshop. J Clin Endocrinol Metab 2009;94:340–350.
Malabanan A, Veronikis IE, Holick MF. Redefining vitamin D insuffiency. Lancet 1998;351:805–806.
Woodhouse NJ, Doyle FH, Joplin GF. Vitamin-D deficiency and primary hyperparathyroidism. Lancet 1971;2:283–286.
Lumb GA, Stanbury SW. Parathyroid function in human vitamin D deficiency and vitamin D deficiency in primary hyperparathyroidism. Am J Med 1974;56:833–839.
Moosgaard B, Vestergaard P, Heickendorff L, et al. Vitamin D status, seasonal variations, parathyroid adenoma weight and bone mineral density in primary hyperparathyroidism. Clin Endocrinol (Oxf) 2005;63:506–513.
Lips P, Duong T, Oleksik A, et al. A global study of vitamin D status and parathyroid function in postmenopausal women with osteoporosis: baseline data from the Multiple Outcomes of Raloxifene Evaluation Clinical Trial. J Clin Endocrinol Metab 2001;86:1212–1221.
Boudou P, Ibrahim F, Cormier C, et al. A very high incidence of low 25 hydroxyvitamin D serum concentration in a French population of patients with primary hyperparathyroidism. J Endocrinol Invest 2006;29:511–515.
Silverberg SJ, Shane E, Dempster DW, et al. The effects of vitamin D insufficiency in patients with primary hyperparathyroidism. Am J Med 1999;107:561–567.
Kandil E, Tufaro AP, Carson KA, et al. Correlation of plasma 25-hydroxyvitamin D levels with severity of primary hyperparathyroidism and likelihood of parathyroid adenoma localization on sistamibi scan. Arch Otolaryngol Head Neck Surg 2008;134:1071–1075.
Ginde AA, Liu MC, Camargo CA Jr. Demographic differences and trends of vitamin D insufficiency in the US population, 1988–2004. Arch Intern Med 2009;169:626–632.
Clements MR, Davies M, Hayes ME, et al. The role of 1,25-dihydroxyvitamin D in the mechanism of acquired vitamin D deficiency. Clin Endocrinol (Oxf) 1992;37:17–27.
Davies M, Heys SE, Selby PL, et al. Increased catabolism of 25-hydroxyvitamin D in patients with partial gastrectomy and elevated 1,25-dihydroxy vitamin D levels. Implications for metabolic bone disease. J Clin Endocrinol Metab 1997;82:209–212.
Bolland MJ, Grey AB, Gamble GD, et al. Association between primary hyperparathyroidism and increased body weight: a meta-analysis. J Clin Endocrinol Metab 2005;90:1525–1530.
Arunabh S, Pollack S, Yeh J, et al. Body fat content and 25-hydroxyvitamin D levels in healthy men. J Clin Endocrinol Metab 2003;88:157–161.
Kantorovich V, Gacad MA, Seeger LL, et al. Bone mineral density increases with vitamin D repletion in patients with coexistent vitamin D insufficiency and primary hyperparathyroidism. J Clin Endocrinol Metab 2000;85:3541–3543.
Rao DS, Honasoge M, Divine GW, et al. Effect of vitamin D nutrition on parathyroid adenoma weight: Pathogenetic and clinical implications. J Clin Endocrinol Metab 2000;85:1054–1058.
Beyer TD, Chen EL, Nilubol N, et al. Short-term outcomes of parathyroidectomy in patients with or without 25-hydroxy vitamin D insufficiency. J Surg Res 2007;143:145–150.
Untch BR, Barfield ME, Dar M, et al. Impact of 25-hydroxyvitamin D deficiency on perioperative parathyroid hormone kinetics and results in patients with primary hyperparathyroidism. Surgery 2007;142:1022–1026.
Stewart ZA, Blackford A, Somervell H, et al. 25-hydroxyvitamin D deficiency is a risk factor for symptoms for symptoms of postoperative hypocalcemia and secondary hyperparathyroidism after minimally invasive parathyroidectomy. Surgery 2005;138:1018–1025.
Moosgaard B, Christensen SE, Vestergaard P, et al. Vitamin D metabolites and skeletal consequences in primary hyperparathyroidism. Clin Endocrinol (Oxf) 2008;68:707–715.
Carnevale V, Manfredi G, Romagnoli E, et al. Vitamin D status in female patients with primary hyperparathyroidism: does it play a role in skeletal change? Clin Endocrinol (Oxf) 2004;60:81–86.
Nordenström E, Westerdahl J, Lindergård B, et al. Multifactorial risk for bone fractures in primary hyperparathyroidism. World J Surg 2002;26:1463–1467.
Lowe H, McMahon DJ, Rubin MR, et al. Normocalcemic primary hyperparathyroidism: further characterization of a new clinical phenotype. J Clin Endocrinol Metab 2007;92:3001–3005.
Grey A, Lucas J, Horne A, et al. Vitamin D repletion in patients with primary hyperparathyroidism and coexistent vitamin D insufficiency. J Clin Endocrinol Metab 2005;90:2122–2126.
Tucci JR. Vitamin D therapy in patients with primary hyperparathyroidism and hypovitaminosis D. Eur J Endocrinol 2009;161:189–193.
Isidro ML, Ruano B. Biochemical effects of calcifediol supplementation in mild, asymptomatic, hyperparathyroidism with concomitant vitamin D deficiency. Endocrine 2009;36:305–310.
Grubbs EG, Rafeeq S, Jimenez C, et al. Preoperative vitamin D replacement therapy in primary hyperparathyroidism: safe and beneficial? Surgery 2008;144:852–858.
Carter GD, Berry JL, Gunter E, et al. Proficiency testing of 25-hydroxyvitamin D (25-OHD) assays. J Steroid Biochem Mol Biol 2010;121:176–179.
