COVID-19 cytokine storms perhaps better stopped by the CYP11A1 Vitamin D pathway
COVID-19 and Vitamin D: A lesson from the skin
Exp Dermatol. 2020 Aug 11. doi: 10.1111/exd.14170
Radomir M Slominski 1, Joanna Stefan 2, Mohamad Athar 2, Michael F Holick 3, Anton M Jetten 4, Chander Raman 1 2, Andrzej T Slominski 2 5
e-mail: aslominski@uabmc.edu; chanderraman@uabmc.edu;
 * Getting Vitamin D into your blood and cells has the following
* Getting Vitamin D into your blood and cells has the following
📄 Download the PDF from VitaminDWiki

The negative outcomes of COVID-19 diseases respiratory distress (ARDS) and the damage to other organs are secondary to a "cytokine storm" and to the attendant oxidative stress. Active hydroxyl-forms of vitamin D are anti-inflammatory, induce anti-oxidative responses, and stimulate innate immunity against infectious agents. These properties are shared by calcitriol and the CYP11A1 -generated non-calcemic hydroxyderivatives. They inhibit the production of pro-inflammatory cytokines, downregulate NF-κΒ, show inverse agonism on RORγ and counteract oxidative stress through the activation of NRF-2. Therefore, a direct delivery of hydroxyderivatives of vitamin D deserves consideration in the treatment of COVID-19 or ARDS of different etiology.
We also recommend treatment of COVID-19 patients with high dose vitamin D since populations most vulnerable to this disease are likely vitamin D deficient and patients are already under supervision in the clinics. We hypothesize that different routes of delivery (oral and parenteral) will have different impact on the final outcome.
Background
The COVID-19 is currently the foremost health issue in the world. SARS-CoV-2 (severe acute respiratory syndrome coronavirus) is an enveloped positive strain RNA virus in the family Coronaviridae, which also includes the virus SARS-CoV-1 (which was another outbreak in 2002- 2003)1. COVID-19 has a fatality rate up to ~5%, which is several times higher than influenza2,3. The leading cause of death in the patients is due to acute respiratory distress syndrome (ARDS)2 induced by proinflammatory responses and oxidative stress (Fig. 1A).
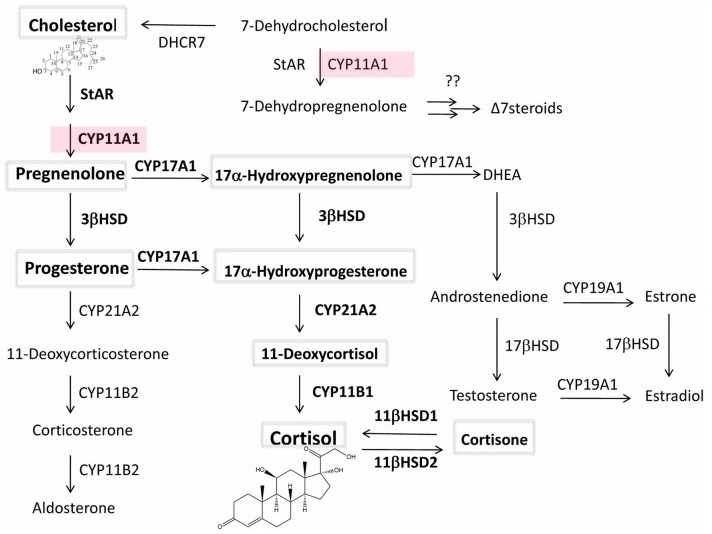
Vitamin D is a fat-soluble prohormone, which after production in the skin or oral delivery affects important physiological functions in the body including regulation of the innate and adaptive immunity4-6. Vitamin D can be activated through canonical and non-canonical pathways (Fig. 1A). In the former, it is metabolized to 25-hydroxyvitamin D3 (25(OH)D3) by CYP2R1 and CYP27A1 in the liver with further metabolism in the kidney to the biologically active 1,25-dihydroxyvitamin D3 (1,25(OH)2D3 ) by CYP27B17-9. This metabolism also occurs in a variety of organs, including skin and the immune system7,9.
An alternative pathway of vitamin D activation by CYP11A1 leads to production of more than 10 metabolites some of which are non-calcemic even at high doses8,10,11. These hydroxyderivatives, including 20(OH)D3 and 20,23(OH)2D3 , are produced in humans 12-15. In addition, 20(OH)D3 has been detected in the honey , which defines it as a natural product16. CYP11A1 is expressed not only in adrenals, placenta and gonads but also in immune cells and other peripheral organs 17.
Both 1,25(OH)2D3 and non-calcemic CYP11A1 derived metabolites use various, although partially overlapping, mechanisms in enacting their anti-inflammatory and anti-oxidative effects (Figure 1B). 1,25(OH)2D3 mediates many of its anti-inflammatory and anti-microbial effects through the vitamin D receptor (VDR) 6,9. 1,25(OH)2D3 can also inhibit the mitogen-activated protein kinase (MAPK) and NF-kB signaling4,9.
The non-calcemic CYP11A1 -derived vitamin D compounds also have their own methods to fight inflammation (Fig. 1B). 20(OH)D3 and their downstream hydroxyderivatives act on VDR as biased agonists11,18,19. They also act as inverse agonists on the retinoic acid-related orphan receptors, RORa and RORy, transcription factors with critical roles in several immune cells and immune responses20-23 (Fig. 1B). In addition, CYP11A1 -derived derived vitamin D3 derivatives and classical 1,25(OH)2D3 can act as agonists on aryl hydrocarbon receptor (AhR)24. Although binding pocket of this receptor can accommodate many different molecules, we believe that secosteroidal signal transduction can be linked to detoxification and anti-oxidative action11 or down-regulation of pro-inflammatory
Premises
ARDS and other adverse effects of COVID-19 are induced by cytokine storm
A leading cause of ARDS is “cytokine storm”,a hyperactive immune response triggered by the viral infection (Fig. 1A)2,26. It is initiated when the pattern recognition receptor of the innate immune cells recognize the pathogen-associated molecular pattern from a pathogen such as bacteria or virus 26,27. The immune cells then release all types of cytokines (interferons, interleukins 1, 6 and 17, chemokines, colony stimulating factors, and tumor necrosis factor (TNF)) leading to hyperinflammation and organ damage 27-29. In the lungs, alveolar cells are targeted leading to acute lung injury and subsequently ARDS 27,30. In severe cases of CoVID-19 other organs and systems are also damaged2,3. Thus, it is crucial to find ways to prevent the “cytokine storm” from going out of control. Although different drugs have been suggested to fight the cytokine storm26,27, they have mixed results and in certain cases can even worsen the disease27. Thus, there is a great need for alternative therapies.
Oxidative stress is also involved in the development of ARDS through action of reactive oxygen ies (ROS) and nitrogen species (NRS)31-33. The production of ROS and RNS can be triggered by ogens promoting the secretion of cytokines, which stimulate ROS production thereby producing sitive feedback loop (Fig. 1A)31,33-35. Nuclear factor erythroid 2p45-related factor 2 (NRF-2) is a transcription factor that plays a role in the detection of excessive ROS and RNS and induction of mechanisms counteracting the oxidative damage36. NRF-2 loss due to ROS can lead to elevation in proinflammatory cytokine levels and stronger inflammatory responses to stimuli31,36.
Anti-inflammatory and antioxidative activities of active forms of vitamin D There is a strong experimental evidence that active forms of vitamin D including the classical 1,25(OH)2D3, and novel CYP11A1 -derived hydroxyderivatives8,11 exert potent anti-inflammatory activities including inhibition of IL-1, IL-6, IL-17, TNFa and INFy production or other proinflammatory pathways (Supplemental table 1) 11,18,20,37,38. The mechanism of action includes downregulation of NF-kB involving action on VDR and inverse agonism on RORy leading to attenuation of Th17 responses (Fig 1B)11,18,20,37-39. These compounds also induce antioxidative and reparative responses with mechanism of action involving activation of NRF-2 and p5311,39-41.
Antiviral effects of active forms of vitamin D
Low vitamin D status in winter permits viral epidemics and vitamin D supplementation could reduce the incidence, severity, and risk of viral diseases42-45. In addition, several reports have found a strong association between vitamin D deficiency/insufficiency and enhanced COVID-19 severity and mortality45-53 with the most recent study defining low plasma 25(OH)D3 as an independent risk factor for COVID-19 infection and hospitalization54. Therefore, we retrospectively analyzed microarray data of human epithelial cells treated with 20,23(OH)2D3 and 1,25(OH)2D324. We found the downregulation of pathways connected with influenza infection and viral RNA transcription, translation, replication, life cycle and of host interactions with influenza factors with 20,23(OH)2D3 expressing higher anti-viral potency (Table 1).
lWhile 1,25(OH)2D3 has the limitation imposed by the toxicity that includes hypercalcemia7,9, CPY11A1-derived 20(OH)D3, 20(OH)D2 and 20,23(OH)2D3 are not toxic and non-calcemic at very high doses (3-60 pg/ kg) at which 1,25(OH)2D3 and 25(OH)D3 are calcemic55-59.
Hypothesis
The hyperinduction of proinflammatory cytokines production (cytokine storm), further magnified by oxidative stress induced by the viral infection or cytokines themselves, acting reciprocally in self- amplifying circuitry, gradually damage/destroy the affected organs leading to death in the severe cases of COVID-19 infection (Fig. 1A). A solution to the problem fulfilling above premises, are active forms of vitamin D including the classical 1,25(OH)2D3 and 25(OH)D3 (precursors to 1,25(OH)2D3) 5,7,9,45,60 and novel CYP11A1 -derived hydroxyderivatives including 20(OH)D3 and 20,23(OH)2D3 8,11,61. The former are FDA approved and can immediately be used in the clinic, while the latter are still not approved yet although they fulfill the definition of natural products. They would both terminate “cytokine storm” and oxidative stress with possible anti-viral activity to rescue the patient from the death path (Fig. 1). Their preferable routes of delivery are listed in Fig. 1C to reach immediately the most affected organs. In this context, active hydroxyforms of vitamin D2 should also be considered59,62-64.
As relates to the vitamin D precursor it is reasonable to propose that patients being admitted with COVID-19 infection to receive as soon as possible 200,000 IU of vitamin D2 or vitamin D3 followed by 4,000-10,000 lU/day, if justifiable45,65. Vitamin D3 at 200,000IU orally has been used to attenuate inflammatory responses induced by the sunburn66. It must be noted that application of 250,000- )0 IU of vitamin D was reported be safe in critically ill patients and was associated with increased hospital length of stay and improved ability of the blood to carry oxygen (reviewed in 67,68)
Relevance and perspective
Different routes of delivery of vitamin D precursor can have a profound effect on the final panel of circulating in the body vitamin D derivatives (Fig. 1C). Vitamin D delivered orally during the passage through the liver is hydroxylated to 25(OH)D7, which is not recognized by CYP11A that only acts on its precursor, vitamin D itself 69. This likely results in 30 times lower concentration of 20(OH)D3 in serum in comparison to 25(OH)D314. However, its levels are higher than that of 25(OH)D3 in the epidermis, a peripheral site of vitamin D3 activation14. Therefore, adequate systemic (adrenal gland) or local (immune system) production of CYP11A1 -derived vitamin D hydroxyderivatives would require parenteral delivery of vitamin D. These routes of vitamin D precursor delivery could include sublingual tablets, intra-muscular, subcutaneous or intravenous injections as well as its aerosolized form of delivery to the lung (Fig. 1C). As relates CYP11A1 -derived products these would be predominantly generated in the adrenal gland for systemic purposes. However, they can also be generated in peripheral organs expressing CYP11A1 including skin and immune system17,70.
Since vitamin D is readily available, easy to use and relatively nontoxic, it can represent an immediate solution to the problems at relatively high doses, since populations most vulnerable to negative outcome of COVID-19 disease are likely vitamin D deficient and the patients are already under supervision in the hospital environment and are monitored for adverse effects. Vitamin D toxicity is typically not observed until extremely high doses of vitamin D in the range of 50,000 100,000 IUs daily for several months or years71. Doses up to 500,000 IUs have been routinely given to nursing home patients twice a year in Scandinavian countries to reduce risk for fracture without any evidence of vitamin D intoxication including hypercalcemia, hyperphosphatemia and soft tissue calcification71.
In addition, we believe that routes of delivery are likely to impact the final outcome, because bypassing liver vitamin D3 will also be accessible to CYP11A1 for metabolism in organs expressing this enzyme.
References
Coronaviridae Study Group of the International Committee on Taxonomy of V. The species Severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol. 2020;5(4):536-544.
Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033-1034.
Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020.
Abhimanyu, Coussens AK. The role of UV radiation and vitamin D in the seasonality and outcomes of infectious disease. Photochem PhotobiolSci. 2017;16(3):314-338.
Wacker M, Holick MF. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinol. 2013;5(1):51-108.
Dankers W, Colin EM, van Hamburg JP, Lubberts E. Vitamin D in Autoimmunity: Molecular Mechanisms and Therapeutic Potential. Front Immunol. 2016;7:697.
Holick MF. Vitamin D deficiency. N EnglJ Med. 2007;357(3):266-281.
Tuckey RC, Cheng CYS, Slominski AT. The serum vitamin D metabolome: What we know and what is still to discover. J Steroid Biochem Mol Biol. 2019;186:4-21.
Bikle DD. Vitamin D: Newer Concepts of Its Metabolism and Function at the Basic and Clinical Level. J Endocr Soc. 2020;4(2):bvz038.
Slominski AT, Kim TK, Li W, Yi AK, Postlethwaite A, Tuckey RC. The role of CYP11A1 in the production of vitamin D metabolites and their role in the regulation of epidermal functions. J Steroid Biochem Mol Biol. 2014;144 Pt A:28-39.
Slominski AT, Chaiprasongsuk A, Janjetovic Z, et al. Photoprotective Properties of Vitamin D and Lumisterol Hydroxyderivatives. Cell Biochem Biophys. 2020;78(2):165-180.
Slominski AT, Kim TK, Shehabi HZ, et al. In vivo evidence for a novel pathway of vitamin D(3) metabolism initiated by P450scc and modified by CYP27B1. FASEB J. 2012;26(9):3901-3915.
Slominski AT, Kim TK, Shehabi HZ, et al. In vivo production of novel vitamin D2 hydroxy- derivatives by human placentas, epidermal keratinocytes, Caco-2 colon cells and the adrenal gland. Mol Cell Endocrinol. 2014;383(1-2):181-192.
Slominski AT, Kim TK, Li W, et al. Detection of novel CYP11A1 -derived secosteroids in the human epidermis and serum and pig adrenal gland. Sci Rep. 2015;5:14875.
Slominski AT, Kim TK, Li W, Tuckey RC. Classical and non-classical metabolic transformation of vitamin D in dermal fibroblasts. Exp Dermatol. 2016;25(3):231-232.
Kim TK, Atigadda V, Brzeminski P, et al. Detection of 7-dehydrocholesterol and vitamin D3 derivatives in honey . Molecules. 2020 📄 PDF is in VitaminDWiki
Slominski RM, Tuckey RC, Manna PR, et al. Extra-adrenal glucocorticoid biosynthesis: implications for autoimmune and inflammatory disorders. Genes Immun. 2020;21(3):150- 168. 📄 Download the PDF from sci-hub via VitaminDWiki
Lin Z, Marepally SR, Goh ESY, et al. Investigation of 20S-hydroxyvitamin D3 analogs and their 1alpha-OH derivatives as potent vitamin D receptor agonists with anti-inflammatory activities. Sci Rep. 2018;8(1):1478.
Kim TK, Wang J, Janjetovic Z, et al. Correlation between secosteroid-induced vitamin D receptor activity in melanoma cells and computer-modeled receptor binding strength. Mol Cell Endocrinol. 2012;361(1-2):143-152.
Slominski AT, Kim TK, Takeda Y, et al. RORalpha and ROR gamma are expressed in human skin and serve as receptors for endogenously produced noncalcemic 20-hydroxy- and 20,23- dihydroxyvitamin D. FASEB J. 2014;28(7):2775-2789.
Jetten AM, Cook DN. (Inverse) Agonists of Retinoic Acid-Related Orphan Receptor gamma: Regulation of Immune Responses, Inflammation, and Autoimmune Disease. Annu Rev Pharmacol Toxicol. 2020;60:371-390.
Slominski AT, Kim TK, Hobrath JV, et al. Endogenously produced nonclassical vitamin D hydroxy-metabolites act as "biased" agonists on VDR and inverse agonists on RORalpha and RORgamma. J Steroid Biochem Mol Biol. 2017;173:42-56.
Jetten AM, Takeda Y, Slominski A, Kang HS. Retinoic acid-related Orphan Receptor gamma (RORgamma): connecting sterol metabolism to regulation of the immune system and autoimmune disease. Curr Opin Toxicol. 2018;8:66-80.
Slominski AT, Kim TK, Janjetovic Z, et al. Differential and Overlapping Effects of 20,23(OH)(2)D3 and 1,25(OH)(2)D3 on Gene Expression in Human Epidermal Keratinocytes: Identification of AhR as an Alternative Receptor for 20,23(OH)(2)D3. Int J MolSci. 2018;19(10).
Gutierrez-Vazquez C, Quintana FJ. Regulation of the Immune Response by the Aryl Hydrocarbon Receptor. Immunity. 2018;48(1):19-33.
Liu Q, Zhou YH, Yang ZQ. The cytokine storm of severe influenza and development of immunomodulatory therapy. Cell Mol Immunol. 2016;13(1):3-10.
Tisoncik JR, Korth MJ, Simmons CP, Farrar J, Martin TR, Katze MG. Into the eye of the cytokine storm. Microbiol Mol Biol Rev. 2012;76(1):16-32.
Pugin J, Ricou B, Steinberg KP, Suter PM, Martin TR. Proinflammatory activity in bronchoalveolar lavage fluids from patients with ARDS, a prominent role for interleukin-1. Am J Respir Crit Care Med. 1996;153(6 Pt 1):1850-1856.
Chen G, Wu D, Guo W, et al. Clinical and immunologic features in severe and moderate Coronavirus Disease 2019. J Clin Invest. 2020.
Kuiken T, Fouchier RA, Schutten M, et al. Newly discovered coronavirus as the primary cause of severe acute respiratory syndrome. Lancet. 2003;362(9380):263-270.
Schieber M, Chandel NS. ROS function in redox signaling and oxidative stress. Curr Biol. 2014;24(10):R453-462.
Ye S, Lowther S, Stambas J. Inhibition of reactive oxygen species production ameliorates inflammation induced by influenza A viruses via upregulation of SOCS1 and SOCS3. J Virol. 2015;89(5):2672-2683.
Vlahos R, Stambas J, Bozinovski S, Broughton BR, Drummond GR, Selemidis S. Inhibition of Nox2 oxidase activity ameliorates influenza A virus-induced lung inflammation. PLoS Pathog. 2011;7(2):e1001271.
To EE, Vlahos R, Luong R, et al. Endosomal NOX2 oxidase exacerbates virus pathogenicity and is a target for antiviral therapy. Nat Commun. 2017;8(1):69.
Imai Y, Kuba K, Neely GG, et al. Identification of oxidative stress and Toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell. 2008;133(2):235-249.
Lee C. Therapeutic Modulation of Virus-Induced Oxidative Stress via the Nrf2-Dependent Antioxidative Pathway. Oxid Med Cell Longev. 2018;2018:6208067.
Janjetovic Z, Tuckey RC, Nguyen MN, Thorpe EM, Jr., Slominski AT. 20,23-dihydroxyvitamin D3, novel P450scc product, stimulates differentiation and inhibits proliferation and NF- kappaB activity in human keratinocytes. J Cell Physiol. 2010;223(1):36-48.
Janjetovic Z, Zmijewski MA, Tuckey RC, et al. 20-Hydroxycholecalciferol, product of vitamin D3 hydroxylation by P450scc, decreases NF-kappaB activity by increasing IkappaB alpha levels in human keratinocytes. PLoS One. 2009;4(6):e5988.
Chaiprasongsuk A, Janjetovic Z, Kim TK, et al. CYP11A1 -derived vitamin D3 products protect against UVB-induced inflammation and promote keratinocytes differentiation. Free Radic Biol Med. 2020;155:87-98.
Chaiprasongsuk A, Janjetovic Z, Kim TK, et al. Protective effects of novel derivatives of vitamin D3 and lumisterol against UVB-induced damage in human keratinocytes involve activation of Nrf2 and p53 defense mechanisms. Redox Biol. 2019;24:101206.
Slominski AT, Janjetovic Z, Kim TK, et al. Novel non-calcemic secosteroids that are produced by human epidermal keratinocytes protect against solar radiation. J Steroid Biochem Mol Biol. 2015;148:52-63.
Teymoori-Rad M, Shokri F, Salimi V, Marashi SM. The interplay between vitamin D and viral infections. Reviews in Medical Virology. 2019;29(2):e2032.
Huang F, Zhang C, Liu Q, et al. Identification of amitriptyline HCl, flavin adenine dinucleotide, azacitidine and calcitriol as repurposing drugs for influenza A H5N1 virus-induced lung injury. PLoS Pathog. 2020;16(3):e1008341.
Jakovac H. COVID-19 and vitamin D—Is there a link and an opportunity for intervention? American Journal of Physiology-Endocrinology and Metabolism. 2020;318(5):E589-E589.
Grant WB, Lahore H, McDonnell SL, et al. Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients. 2020;12(4).
Cheng RZ. Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)? Med Drug Discov. 2020;5:100028.
D'Avolio A, Avataneo V, Manca A, et al. 25-Hydroxyvitamin D Concentrations Are Lower in Patients with Positive PCR for SARS-CoV-2. Nutrients. 2020;12(5).
Hastie CE, Mackay DF, Ho F, et al. Vitamin D concentrations and COVID-19 infection in UK Biobank. Diabetes Metab Syndr. 2020;14(4):561-565.
Ilie PC, Stefanescu S, Smith L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin Exp Res. 2020.
Jakovac H. COVID-19 and vitamin D-Is there a link and an opportunity for intervention? Am J Physiol Endocrinol Metab. 2020;318(5):E589.
Marik PE, Kory P, Varon J. Does vitamin D status impact mortality from SARS-CoV-2 infection? Med Drug Discov. 2020:100041.
McCartney DM, Byrne DG. Optimisation of Vitamin D Status for Enhanced Immuno- protection Against Covid-19. Ir Med J. 2020;113(4):58.
Panarese A, Shahini E. Letter: Covid-19, and vitamin D. Aliment Pharmacol Ther. 2020;51(10):993-995.
Merzon E, Tworowski D, Gorohovski A, et al. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: an Israeli population-based study. The FEBS Journal.n/a(n/a).
Slominski AT, Janjetovic Z, Fuller BE, et al. Products of vitamin D3 or 7-dehydrocholesterol metabolism by cytochrome P450scc show anti-leukemia effects, having low or absent calcemic activity. PLoS One. 2010;5(3):e9907.
Wang J, Slominski A, Tuckey RC, et al. 20-hydroxyvitamin D(3) inhibits proliferation of cancer cells with high efficacy while being non-toxic. Anticancer Res. 2012;32(3):739-746.
Slominski A, Janjetovic Z, Tuckey RC, et al. 20S-hydroxyvitamin D3, noncalcemic product of CYP11A1 action on vitamin D3, exhibits potent antifibrogenic activity in vivo. J Clin Endocrinol Metab. 2013;98(2):E298-303.
Chen J, Wang J, Kim TK, et al. Novel vitamin D analogs as potential therapeutics: metabolism, toxicity profiling, and antiproliferative activity. Anticancer Res. 2014;34(5):2153-2163.
Slominski AT, Kim TK, Janjetovic Z, et al. 20-Hydroxyvitamin D2 is a noncalcemic analog of vitamin D with potent antiproliferative and prodifferentiation activities in normal and malignant cells. Am J Physiol Cell Physiol. 2011;300(3):C526-541.
Shirvani A, Kalajian TA, Song A, Holick MF. Disassociation of Vitamin D's Calcemic Activity and Non-calcemic Genomic Activity and Individual Responsiveness: A Randomized Controlled Double-Blind Clinical Trial. Sci Rep. 2019;9(1):17685.
Slominski AT, Li W, Kim TK, et al. Novel activities of CYP11A1 and their potential physiological significance. J Steroid Biochem Mol Biol. 2015;151:25-37.
Holick MF. Vitamin D: A millenium perspective. J Cell Biochem. 2003;88(2):296-307.
Bikle DD. Vitamin D: an ancient hormone. Exp Dermatol. 2011;20(1):7-13.
Slominski A, Semak I, Wortsman J, et al. An alternative pathway of vitamin D metabolism. Cytochrome P450scc ( CYP11A1 )-mediated conversion to 20-hydroxyvitamin D2 and 17,20- dihydroxyvitamin D2. FEBS J. 2006;273(13):2891-2901.
Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911-1930.
Scott JF, Das LM, Ahsanuddin S, et al. Oral Vitamin D Rapidly Attenuates Inflammation from Sunburn: An Interventional Study. J Invest Dermatol. 2017;137(10):2078-2086.
Ebadi M, Montano-Loza AJ. Perspective: improving vitamin D status in the management of COVID-19. Eur J Clin Nutr. 2020:1-4.
Grant WB, Baggerly CA, Lahore H. Reply: "Vitamin D Supplementation in Influenza and COVID-19 Infections. Comment on: Evidence That Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths Nutrients 2020, 12(4), 988". Nutrients. 2020;12(6).
Slominski A, Semak I, Zjawiony J, et al. The cytochrome P450scc system opens an alternate pathway of vitamin D3 metabolism. FEBS J. 2005;272(16):4080-4090.
Slominski A, Zjawiony J, Wortsman J, et al. A novel pathway for sequential transformation of 7-dehydrocholesterol and expression of the P450scc system in mammalian skin. Eur J Biochem. 2004;271(21):4178-4188.
Holick MF. Vitamin D Is Not as Toxic as Was Once Thought: A Historical and an Up-to-Date Perspective. Mayo Clinic Proceedings. 2015;90(5):561-564.
Newer publication by Slominski etc. - Sept 2021
Vitamin D and lumisterol novel metabolites can inhibit SARS-CoV-2 replication machinery enzymes
📄 Download the PDF from VitaminDWiki
Update - Aug 2020
Reply to Jakovac and to Rocha et al.: Can vitamin D prevent or manage COVID-19 illness - Aug 13, 2020
X Andrzej T. Slominski ,1,7 Radomir M. Slominski,2,3 Paul A. Goepfert,2,4 Tae-Kang Kim,1 Michael F. Holick ,5 Anton M. Jetten,6 and Chander Raman2,3
Effectiveness of honey for symptomatic relief in upper respiratory tract infections: a systematic review and meta-analysis - Aug 2020
Vitamin D is not mentioned
📄 Download the PDF from VitaminDWiki
Perhaps 100 IU of vitamin D in 10 grams of honey - June 2020
Detection of 7-Dehydrocholesterol and Vitamin D3 Derivatives in Honey - June 2020 doi: 10.3390/molecules25112583
256 ng/gram of honey = 2560 ng. 10 grams = 2.56 micrograms
ASSUMING (could be way off here) that there are 40 IUs/microgram for 20(OH)D3
Then 10 grams of honey would have 100 IU