Draft of UK policy on Vitamin D, keeping 10 ng definition
PDF is attached at the bottom of this page
Document is from http://guidance.nice.org.uk/PHG/71 they are requesting comments
Note: current UK definition of Vitamin D deficiency as <25 nmol/L ( <10ng/mL )
Comments by VitaminDWiki
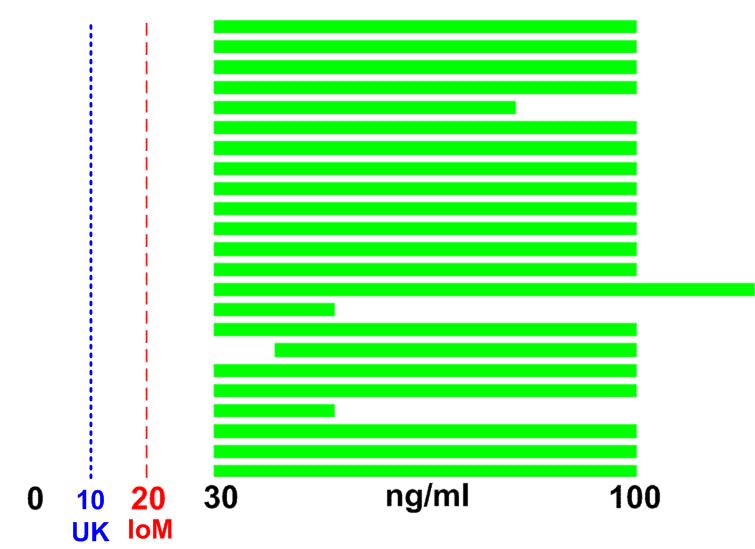
Why bother commenting on this document if it keeps the 10ng definition, which is the lowest in the world
10 ng/ml often does not even prevent Rickets, or any of the diseases which have been proven to be prevented or treated by vitamin D
Healthy Start vitamin D supplementation in UK is a farce - Nov 2010
It took over 50 years for the UK to agree that vitamin C could prevent scurvy - why should they be any faster now?
The myth that >2,000 IU of vitamin D was toxic was started in the UK only 50 years ago
Perhaps better to try a different way to increase vitamin D, such as using social media (Brazil)
Reminder: Doctors have at least 10 reasons to be reluctant to increase vitamin D: which has the following quote
- It is difficult to get a man to understand something when his salary is dependent upon his not understanding it Upton Sinclair
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
PUBLIC HEALTH GUIDANCE: DRAFT SCOPE
1 Guidance title
Vitamin D: implementation of existing guidance to prevent deficiency
2 Background
a) The National Institute for Health and Clinical Excellence (NICE) has been asked by the Department of Health (DH) to develop guidance to help safely implement existing evidence-based recommendations on the prevention of vitamin D deficiency. It will focus on at-risk groups including infants, children, older people, pregnant and breastfeeding women and those who have limited exposure to the sun.
b) This guidance will support a number of related policy and guidance documents including:
'Delivering a healthy start for pregnant women, new mums, babies and young children' (DH 2011a)
'Healthy lives, healthy people: our strategy for public health in England' (DH 2010)
'Update on vitamin D' (Scientific Advisory Committee on Nutrition 2007)
'Vitamin D - advice on supplements for at risk groups' (Chief Medical Officers 2012).
c) This guidance will provide recommendations for good practice, based on the best available evidence of effectiveness, including cost effectiveness. It is aimed at commissioners, managers and other professionals with public health as part of their remit working Vitamin D: implementation of existing guidance to prevent deficiency draft scope for consultation within the NHS, local authorities and the wider public, private, voluntary and community sectors. It is also aimed at the suppliers and providers of vitamin D supplements. In addition, it may be of interest to people at risk of vitamin D deficiency, their families and carers and other members of the public.
d) The guidance will support NICE guidance on antenatal care, maternal and child nutrition and skin cancer. For further details, see section 6.
This guidance will be developed using the NICE public health guidance process and methods guides.
3 The need for guidance
a) Vitamin D is essential for skeletal growth and bone health. Dietary sources are limited (they include oily fish, egg yolk and fortified margarines, breakfast cereals and infant formula). The major natural source of vitamin D is from skin synthesis following exposure to sunlight. Existing evidence suggests that from October to April in the UK, there is no ambient ultraviolet sunlight of the appropriate wavelength. During this time, the population relies on body stores (gathered during exposure in the summer) and dietary sources to maintain vitamin D levels (Scientific Advisory Committee on Nutrition 2007).
b) Severe vitamin D deficiency results in rickets (among children) and osteomalacia (among children and adults). It has also been associated with some diseases and long-term conditions, such as osteoporosis, diabetes and some cancers (Scientific Advisory Committee on Nutrition 2007). There have been reports that rickets is re-emerging among children in the UK (Pearce and Cheetham 2010). The national diet and nutrition survey of British adults (Ruston et al. 2004) suggests that about a quarter of British women aged 19-24, and a sixth of those aged 25-34, are at risk of vitamin D deficiency.
c) Vitamin D deficiency can occur during periods of rapid growth, during pregnancy and while breastfeeding. A newborn baby's vitamin D status is largely determined by the mother's level of vitamin D. Women and children of South Asian, African, Caribbean and Middle Eastern descent, and those who remain covered when outside, are at greatest risk. Infants who are exclusively breastfed, particularly for more than 6 months, are at increased risk because scope for consultation the amount of vitamin D in breast milk will not meet their requirements. (See NICE guidance on antenatal care and maternal and child nutrition.) Older people are at increased risk because their skin cannot synthesise vitamin D efficiently, they are likely to spend more time indoors and may not have enough dietary vitamin D. Limited sun exposure - for example, among those who are housebound or prisoners - may also increase people's risk (Scientific Advisory Committee on Nutrition 2007).
d) In 1991, the Committee on Medical Aspects of food policy (COMA) set reference nutrient intakes (RNI) for different population groups (DH 1991). In most instances, an intake above the RNI can only be guaranteed by taking a vitamin D supplement. In 2007, the Scientific Advisory Committee on Nutrition confirmed that these recommendations should remain unchanged. All UK health departments (Chief Medical Officers 2012) and NICE (see guidance on antenatal care and maternal and child nutrition) have issued evidence-based guidance on vitamin D supplements for various at-risk groups. They have also provided guidance on how to distribute free Healthy Start supplements (which contain vitamin D) to eligible families.
e) Health professionals could make 'a significant difference' if they ensure those at risk of vitamin D deficiency understand how important the vitamin is, and how to get a daily supplement. That's the view of the 4 UK Chief Medical Officers. They also stress the need to ensure people who may be eligible for the Healthy Start scheme know how they can apply. All at-risk groups should be made aware of how they can obtain the vitamins locally (Chief Medical Officers 2012). (Note: The Healthy Start scheme includes free formula milk and fruit and vegetables, as well as vitamin supplements.) Evidence suggests implementation of these recommendations has been limited - and uptake of Healthy Start vitamins among families who qualify for the scheme varies markedly across England (Chief Medical Officers 2012; Feeding for Life Foundation 2012; DH: personal communication 2012). The cost effectiveness of implementing existing guidance on vitamin D remains unclear.
f) Healthy Start vitamins are usually available from health clinics, children's centres, Sure Start centres, outreach programmes or GP surgeries, although there have been supply problems in the past. Manufacturers have not made them directly available to pharmacies, although this can be arranged locally. (Pharmacies may sell other commercially available supplements containing vitamin D at a higher price.) It is possible for those not eligible for Healthy Start to buy Healthy Start supplements and this has been encouraged in some areas (DH 2011b). They cost 91p for women and £1.80 for children for an 8-week supply. Primary care spending on treatments for vitamin D deficiency rose from £28 million in 2004 to £76 million in 2011 (Robinson 2012; Health and Social Care Information Centre 2012).
4 The guidance
Public health guidance will be developed according to NICE processes and methods. For details see section 5.
This document defines exactly what this guidance will (and will not) examine, and what the guidance developers will consider. The scope is based on a referral from the DH (see appendix A).
4.1 Who is the focus?
4.1.1 Groups that will be covered
Population groups at increased risk of vitamin D deficiency:
All pregnant and breastfeeding women.
Infants and young children aged under 5 years.
Older people aged 65 and over.
People who have low (or no exposure) to the sun.
- For example, those who cover their skin for cultural reasons and those who are housebound or confined indoors for long periods (such as people in prison).
- People with dark skin,
- for example, people of African, African-Caribbean and South Asian origin (because their bodies cannot make as much vitamin D as those with paler skins).
{Ignores obesity: Comment by VitaminDWiki}
4.1.2 Groups that will not be covered
People being treated for vitamin D deficiency.
People with diseases or conditions which may be associated with an increased risk of vitamin D deficiency.
{Thus wait until after the persons have MS, Diabetes, Cancer, Hypertension, Comment by VitaminDWiki}
4.2 Activities
4.2.1 Activities that will be covered
a) Activities to increase awareness or uptake of vitamin D supplements among at-risk groups in a range of settings, in line with existing evidence-based guidance for England. This includes guidance issued by the Committee on Medical Aspects of food policy, the Scientific Advisory Committee on Nutrition, the DH and NICE.
b) Activities to increase provision of vitamin D supplements in a range of settings, in line with existing guidance for England (see above).
c) Activities to increase uptake of Healthy Start vitamins in a range of settings among eligible groups in England.
4.2.2 Activities/measures that will not be covered
Activities that will not be covered include:
Management of vitamin D deficiency.
Management of conditions that may increase the risk of vitamin D deficiency.
Fortification of food and drinks with vitamin D.
Recommendations on the intake of vitamin D for different population groups.
Measures that will not be covered include:
Indicators of vitamin D status.
The relative contribution of dietary and cutaneous vitamin D synthesis to the vitamin D status of the UK population.
The association between vitamin D status and health outcomes.
Length and intensity of sun exposure for different population groups.
4.3 Key questions and outcomes
Below are the overarching questions that will be addressed, along with some of the outcomes that would be considered as evidence of effectiveness:
Question 1: How effective and cost effective are interventions to increase awareness and implementation of existing guidance on vitamin D among health professionals? What are the implications for professional training and practice?
Question 2: How effective and cost effective are interventions to increase awareness and uptake of existing guidance on vitamin D among at-risk groups (with special consideration given to those eligible for the Healthy Start scheme)?
Question 3: What helps or hinders the implementation of existing guidance on vitamin D among commissioners, providers, practitioners and at-risk groups?
Question 4: What provision is made for vitamin D supplements for different at-risk groups through Healthy Start, prescriptions and over-the-counter sales?
Expected outcomes
• Awareness of vitamin D guidance among at-risk groups (including any differences between socioeconomic groups).
• Awareness of vitamin D guidance among health professionals, commissioners and providers.
• Uptake of Healthy Start vitamins among eligible groups.
• Adherence to vitamin D guidance among at-risk groups (including any differences between socioeconomic groups).
• Adherence to vitamin D guidance among health professionals, commissioners and providers.
• Reported barriers and facilitators to implementing vitamin D guidance among at-risk groups, health professionals, commissioners and providers.
• Indicators of product availability.
Economic outcomes
For economic modelling, estimates of length and quality of life, with and without an intervention, will be needed to estimate quality-adjusted life years (QALYs) gained.
An economic analysis will be conducted from several perspectives - the NHS, local government, public sector and societal (as appropriate).
Disaggregated information on health and non-health related costs and benefits may be captured in cost-consequences analyses. Return on investment (ROI) may also be considered.
The main time horizon for cost effectiveness will be lifetime, but shorter timescales will be required for ROI analyses.
4.4 Status of this document
This is the draft scope, released for consultation on 7 January 2013 to 1 February 2013. Following consultation, the final version of the scope will be available at the NICE website in March 2013.
5 Further information
The public health guidance development process and methods are described in Methods for development of NICE public health guidance (third edition) (2012) and The NICE public health guidance development process (third edition) (2012).
6 Related NICE guidance Published
Skin cancer prevention: information, resources and environmental changes NICE public health guidance 32 (2010)
Weight management before, during and after pregnancy. NICE public health guidance 27 (2010)
Maternal and child nutrition. NICE public health guidance 11 (2008) Antenatal care. NICE clinical guideline 62 (2008)
Postnatal care. NICE clinical guideline 37 (2006) Under development
Sunlight exposure: benefits and safety. NICE public health guidance (publication date to be confirmed)
Prisons: physical conditions and diseases. NICE public health guidance (publication date to be confirmed)
Appendix A Referral from the Department of Health
The Department of Health asked NICE to develop public health guidance on: 'The safe implementation of existing evidence-based guidance on prevention of vitamin D deficiency, to include recommendations relating to infants, children, older people, pregnant and lactating women and those with limited sun exposure'.
Appendix B Potential considerations
It is anticipated that the Public Health Advisory Committee (PHAC) will consider the following issues:
The target audience, actions taken and by whom, context, frequency and duration.
Critical elements. For example, whether effectiveness and cost effectiveness varies according to:
- the diversity of the population (for example, in terms of the user's age, gender or ethnicity)
- the status of the person delivering it and the way it is delivered
- its frequency, length and duration, where it takes place and whether it is transferable to other settings
- its intensity.
• Any trade-offs between equity and efficiency.
• Any factors that prevent - or support - effective implementation.
• Any adverse or unintended effects.
• Current practice.
• Availability and accessibility for different groups.
Appendix C References
Chief Medical Officers (2012) Vitamin D - advice on supplements for at risk groups [online].
Department of Health (1991) Dietary reference values for food energy and nutrients for the United Kingdom. Report of the panel on dietary reference values of the Committee on Medical Aspects of food policy. London: HMSO
Department of Health (2010) Healthy lives, healthy people: our strategy for public health in England. London: Department of Health
Department of Health (2011 a) Delivering a healthy start for pregnant women, new mums, babies and young children. London: Department of Health
Department of Health (2011 b) Help pregnant women, new mothers and children get their free healthy start vitamins. London: Department of Health
Feeding for Life Foundation (2012) Mind the gap - are the current vitamin recommendations meeting the needs of the under 5s in the UK? London: Feeding for Life Foundation
Health and Social Care Information Centre, Prescribing and Primary Care Services (2012) *Prescription cost analysis England 2011. London: Health and Social Care Information Centre
Pearce SHS, Cheetham TD (2010) Diagnosis and management of vitamin D deficiency [online]
Robinson S (2012) Treating vitamin D deficiency [online]
Ruston D, Hoare J, Henderson L et al. (2004) The national diet and nutrition survey: adults aged 19 to 64 years. Nutritional status (anthropometry and blood analytes), blood pressure and physical activity. London: The Stationery Office
Scientific Advisory Committee on Nutrition (2007) Update on vitamin D. Position statement by the Scientific Advisory Committee on Nutrition. London: The Stationery Office
See also VitaminDWiki
Low vitamin D associated with poor regions in the UK, median was 10 ng – Nov 2012
4 out of 10 Irish have vitamin D levels inadequate even for bone health – Aug 2012
UK pediatricians have a lot to learn about vitamin D – May 2012
Scotts having extremely low vitamin D got no, or only minimal treatment – Feb 2012
11X more non-immigrant children allergic to peanuts if vitamin D less than 20 ng – Feb 2013
The women who most need vitamin D are the least to get it – thesis March 2013
Healthy Start vitamin D supplementation in UK is a farce - Nov 2010 just 180 IU
Let 1000 die from vitamin D deficiency rather than incur 1 lawsuit
A look at inner workings of committee looking at vitamin D for UK - Dec 2012
Obese need more Vitamin D: Volume dilution, IU per pound, or BMI – RCT Dec 2012
UK-NICE draft on vitamin D – extremely low doses and levels– May 2014
43 reasons for Vitamin D deficiency which this document appears to ignore
- obesity
- Cloudy areas (Scotland)
- High latitudes (Scotland)
- Children more than 5 years old. Expect a huge drop in vitamin D levels as children pass their 6th birthday
- Recent trauma, Chemotherapy - all of which use vitamin D
- Planned or recent surgery - which uses a lot of vitamin D
- work shifts which have the person sleeping at noon
- 25 experts recommend vitamin D level of min 30 ng – Nov 2009
- The MINIMUM recommendation = 30 ng was higher than the IoM = 20, or UK = 10