Superspreaders appear to have viscous lung fluids or be obese
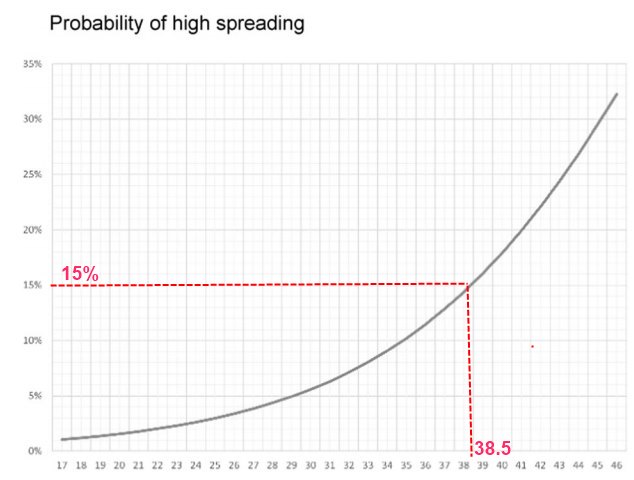
10.9 X more likely to be a superspreader if obese - Aug 2022
Correlation between body mass index and COVID-19 transmission risk
International Journal of Obesity; https://doi.org/10.1038/s41366-022-01215-y
Based on detailed contact tracing of 218 people:
"Once the risk was adjusted to confounders such as age, gender, comorbidities, and symptoms the risk was even higher ( AOR 10.89 , CI95% 2.67–44.33, p = 0.001)"
📄 Download the PDF from VitaminDWiki
1,500 hits for superspreaders obese COVID in Google Scholar as of Aug 2022
Why some people are superspreaders and how the body emits coronavirus
National Geographic Oct 2020
“Aerosols are usually less than 100 micrometers in size”
Note: Commercial masks are designed to stop much larger particles
Note: Single-layer cloth masks are even less able to stop tiny particles
Higher viscosity ==> more popped bubbles in the lungs which create aerosols
Higher viscosity ==> 10X more aerosols for the same loudness of speech
“For example, the viscosity of the respiratory lining increases during bronchial infections such as bacterial pneumonia and severe influenza due to the loss of water and increased production of cellular proteins. Chronic conditions like asthma and cystic fibrosis can also thicken up the fluids.”
Thus people with viscous lung fluids are emitting more aerosols
- The aerosols spread the virus if the person is infected
Summary: poor lung health ==> Viscous fluid ==> bubbles ==> 10X aerosols
- What does this imply for Nursing Homes (COPD, etc)?
Note: As a retired instrumentation engineer, I am aware that it would be a simple matter to design a low-cost instrument that could measure the amount of aerosols a person emits. However, what would society do with that information? Lockdown/restrict those people (1%?) emitting high aerosols and let others (99%) return to their normal lives? Henry Lahore, founder of VitaminDWiki
VitaminDWiki pages with SUPERSPREADER in title (4 as of Aug 2022)
This list is automatically updated
{LIST()}
VitaminDWiki
How to identify and limit potential superspreaders
Wonder if: increasing Vitamin D levels
==> decreases flud viscosity
==> fewer aerosols
==> reduce superspreadingBreathing category in VitaminDWiki starts with the following
{include}
Does a person in a group exhale lots of aerosols? If so, give them a super-mask
Simple, 5-second simple aerosol test of people who will be in large groups:
church, business, cruise ship, sporting event, colleges large resturant, military ...
Provide person with a "super-mask" to protect others in case the aerosol being exhaled is infectious
super-masks trap very small particles (0.02 µm) Examples: N99, FFP3, N100
Transmission of COVID-19 virus by droplets and aerosols:
A critical review on the unresolved dichotomy - Sept 2020
Environ Res. 2020 Sep; 188: 109819.
Published online 2020 Jun 13. doi: 10.1016/j.envres.2020.109819
PMCID: PMC7293495, PMID: 32569870
Mahesh Jayaweera,a,∗ Hasini Perera,b Buddhika Gunawardana,a and Jagath Manatungea
📄 Download the PDF from VitaminDWiki
The practice of social distancing and wearing masks has been popular worldwide in combating the contraction of COVID-19. Undeniably, although such practices help control the COVID-19 pandemic to a greater extent, the complete control of virus-laden droplet and aerosol transmission by such practices is poorly understood. This review paper intends to outline the literature concerning the transmission of virus-laden droplets and aerosols in different environmental settings and demonstrates the behavior of droplets and aerosols resulted from a cough-jet of an infected person in various confined spaces. The case studies that have come out in different countries have, with prima facie evidence, manifested that the airborne transmission plays a profound role in contracting susceptible hosts. The infection propensities in confined spaces (airplane, passenger car, and healthcare center) by the transmission of droplets and aerosols under varying ventilation conditions were discussed.
Interestingly, the nosocomial transmission by airborne SARS-CoV-2 virus-laden aerosols in healthcare facilities may be plausible. Hence, clearly defined, science-based administrative, clinical, and physical measures are of paramount importance to eradicate the COVID-19 pandemic from the world.
Particle sizes of infectious aerosols: implications for infection control - July 2020
Kevin P Fennelly, MD
Lancet Respiratory Medicine, DOI: https://doi.org/10.1016/S2213-2600(20)30323-4
📄 Download the PDF from VitaminDWiki
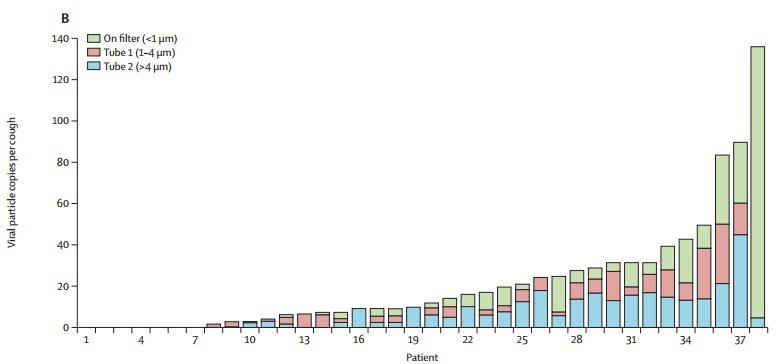
The global pandemic of COVID-19 has been associated with infections and deaths among health-care workers. This Viewpoint of infectious aerosols is intended to inform appropriate infection control measures to protect health-care workers. Studies of cough aerosols and of exhaled breath from patients with various respiratory infections have shown striking similarities in aerosol size distributions, with a predominance of pathogens in small particles (<5 μm). These are immediately respirable, suggesting the need for personal respiratory protection (respirators) for individuals in close proximity to patients with potentially virulent pathogens. There is no evidence that some pathogens are carried only in large droplets. Surgical masks might offer some respiratory protection from inhalation of infectious aerosols, but not as much as respirators. However, surgical masks worn by patients reduce exposures to infectious aerosols to health-care workers and other individuals. The variability of infectious aerosol production, with some so-called super-emitters producing much higher amounts of infectious aerosol than most, might help to explain the epidemiology of super-spreading. Airborne infection control measures are indicated for potentially lethal respiratory pathogens such as severe acute respiratory syndrome coronavirus 2.
Huge variation in the number of small particles emitted(green)
The person at the far right is a potential superspreader
Breathing Is Enough: For the Spread of Influenza Virus and SARS-CoV-2 by Breathing Only - July 2020
Journal of Aerosol Medicine and Pulmonary Drug DeliveryVol. 33, No. 4 Letters to the Editor
Gerhard Scheuch
Published Online:28 Jul 2020 https://doi.org/10.1089/jamp.2020.1616 FREE PDF
Background: The transmission of respiratory viruses such as influenza and corona viruses from one person to another is still not fully understood.
Methods: A literature search showed that there is a strong scientific rationale and evidence that viruses are very efficiently spread through aerosols by the patient's breathing only. It is not necessary for the patient to cough or sneeze.
Results: The exhaled aerosol particles are generated by normal breathing in the deep lung through reopening of collapsed small airways during inspiration. These mucus/surfactant aerosols (size range between 0.2 and 0.6 μm) can transport viruses out of the lungs of patients and be present in the room air for hours.
Conclusion: These aerosol particles are difficult to filter out of the air; because of their physical properties, new strategies must be developed to protect people from these virus aerosols.
History
In 1986 and 1987, a team of aerosol researchers from the Institute for Biophysics at the GSF (Research Center for Environment and Health) in Frankfurt investigated inhaled and exhaled aerosol particles. Their goal was to measure the growth of these aerosol particles in the airways. Using a very powerful laser directly in front of the mouth, they were able to measure both the number and the size of inhaled and exhaled particles.(1) With a two mode laser photometer, they were able to count and measure aerosol particles in a size range between 0.2 and 10 μm. These researchers discovered a strange phenomenon. The test persons' lungs seemed to function like an “aerosol generator.” Even after minutes of inhaling particle-free air, the subjects exhaled different concentrations of very small aerosol particles. Particles with an aerodynamic size range >5 μm were not found in the exhaled air during quiet breathing.
The team began to investigate this “disruptive factor” in more detail and discovered the following phenomena:
(1) The lungs produce aerosol particles with a size of ∼0.4 μm in diameter.
(2) The production rate differs considerably between individual test persons. The measured aerosol concentrations varied between a few tens of particles per liter of exhaled air to several thousand particles per liter .
(3) The particles are produced during inhalation and are released with the subsequent exhalation.a
(4) The particles do not arise in the upper but in the lower very small airways. At the beginning of an exhalation in the first ∼200 mL, there are no or very few particles, and at the end of the exhalation the concentration increases.
(5) Respiratory flow rate had no influence on aerosol concentration.
(6) On test day 1, a multiple of the exhaled particles were measured in one of the subjects. The following day, he reported sick due to a respiratory infection. When he recovered, his aerosol concentration was back to normal.
The hypothesis of how these aerosol particles are created is that small airways collapse during exhalation and are reopened during the subsequent inhalation, creating small mucus/surfactant droplets that are exhaled with the subsequent exhalation. In patients with respiratory infections, the increased production of surfactant and mucus could enhance this process. The data at that time were only published as lectures and posters at a congress
Exhaled particles and small airways - 2019
B. Bake, P. Larsson, G. Ljungkvist, E. Ljungström & A-C Olin
Respiratory Research volume 20, Article number: 8 (2019)
Background
Originally, studies on exhaled droplets explored properties of airborne transmission of infectious diseases. More recently, the interest focuses on properties of exhaled droplets as biomarkers, enabled by the development of technical equipment and methods for chemical analysis. Because exhaled droplets contain nonvolatile substances, particles is the physical designation. This review aims to outline the development in the area of exhaled particles, particularly regarding biomarkers and the connection with small airways, i e airways with an internal diameter < 2 mm.
Main body
Generation mechanisms, sites of origin, number concentrations of exhaled particles and the content of nonvolatile substances are studied. Exhaled particles range in diameter from 0.01 and 1000 μm depending on generation mechanism and site of origin. Airway reopening is one scientifically substantiated particle generation mechanism. During deep expirations, small airways close and the reopening process produces minute particles. When exhaled, these particles have a diameter of < 4 μm. A size discriminating sampling of particles < 4 μm and determination of the size distribution, allows exhaled particle mass to be estimated. The median mass is represented by particles in the size range of 0.7 to 1.0 μm. Half an hour of repeated deep expirations result in samples in the order of nanogram to microgram. The source of these samples is the respiratory tract ling fluid of small airways and consists of lipids and proteins, similarly to surfactant. Early clinical studies of e g chronic obstructive pulmonary disease and asthma, reported altered particle formation and particle composition.
Conclusion
The physical properties and content of exhaled particles generated by the airway reopening mechanism offers an exciting noninvasive way to obtain samples from the respiratory tract lining fluid of small airways. The biomarker potential is only at the beginning to be explored.
The Mechanism of Breath Aerosol Formation - 2009
Journal of Aerosol Medicine and Pulmonary Drug DeliveryVol. 22, No. 3ORIGINAL ARTICLES
Graham Richard Johnson and Lidia Morawska
Published Online:14 Sep 2009 https://doi.org/10.1089/jamp.2008.0720 FREE PDF
Background: Aerosol production during normal breathing is often attributed to turbulence in the respiratory tract. That mechanism is not consistent with a high degree of asymmetry between aerosol production during inhalation and exhalation. The objective was to investigate production symmetry during breathing.
Methods: The aerosol size distribution in exhaled breath was examined for different breathing patterns including normal breathing, varied breath-holding periods, and contrasting inhalation and exhalation rates. The aerosol droplet size distribution measured in the exhaled breath was examined in real time using an aerodynamic particle sizer.
Results and Conclusions: The dependence of the particle concentration decay rate on diameter during breath holding was consistent with gravitational settling in the alveolar spaces. Also, deep exhalation resulted in a four- to sixfold increase in concentration, and rapid inhalation produced a further two- to threefold increase in concentration. In contrast, rapid exhalation had little effect on the measured concentration. A positive correlation of the breath aerosol concentration with subject age was observed. The results were consistent with the breath aerosol being produced through fluid film rupture in the respiratory bronchioles in the early stages of inhalation and the resulting aerosol being drawn into the alveoli and held before exhalation. The observed asymmetry of production in the breathing cycle with very little aerosol being produced by exhalation is inconsistent with the widely assumed turbulence-induced aerosolization mechanism.
Title change made in 2022 caused the visitor count to reset.
There have actually been visitors to this page since it was originally made
