Not as much active vitamin D if poor kidney function and low vitamin D
Calcitriol/Calcifediol ratio: an indicator of vitamin D hydroxylation efficiency?
BBA Clinical. Available online 14 March 2015, doi:10.1016/j.bbacli.2015.03.004
Marzia Pasqualia, Lida Tartaglionea, Silverio Rotondia, Maria Luisa Mucia, Giusi Mandanicia, Alessio Farcomenib, Martino Marangellac, Sandro Mazzaferroa, ,
a Department of Cardiovascular, Respiratory, Nephrologic, Anesthesiologic and Geriatric Sciences, Sapienza University of Rome, Italy
b Dept. of Public Health and Infectious Diseases, Sapienza University of Rome
c A.O. Ospedale Mauriziano, Torino
Highlights
1,25D/25D ratio could represent an index of vitamin D hydroxylation efficiency.
1,25D/25D ratio progressively increases from HD to CKD, TX, no-CKD and PHP.
Each of these clinical conditions affected the value of the ratio.
In selected populations 1,25D/25D ratio could guide substitutive therapeutic choices.
 See also VitaminDWik
* Overview Kidney and vitamin D
* Calcitriol (active Vitamin D) recommended after kidney transplant – March 2014
* Chronic Kidney Disease study not aware of appropriate forms of vitamin D – March 2014
* Time-release form of active vitamin D granted a patent for chronic kidney disease – July 2014
* Vitamin D level can be high, but little benefit: due to kidney, genes, low Magnesium etc.
* not considered by study on this page
* Omega 3 increases vitamin D in the blood – many studies
* not considered by study on this page
* Calcitriol **category listing has items along with related searches**
* Vitamin D3 becomes Calcidiol which becomes Calcitriol has the following graphics
See also VitaminDWik
* Overview Kidney and vitamin D
* Calcitriol (active Vitamin D) recommended after kidney transplant – March 2014
* Chronic Kidney Disease study not aware of appropriate forms of vitamin D – March 2014
* Time-release form of active vitamin D granted a patent for chronic kidney disease – July 2014
* Vitamin D level can be high, but little benefit: due to kidney, genes, low Magnesium etc.
* not considered by study on this page
* Omega 3 increases vitamin D in the blood – many studies
* not considered by study on this page
* Calcitriol **category listing has items along with related searches**
* Vitamin D3 becomes Calcidiol which becomes Calcitriol has the following graphics
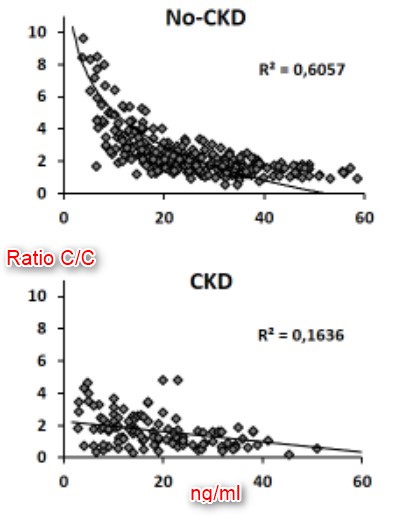
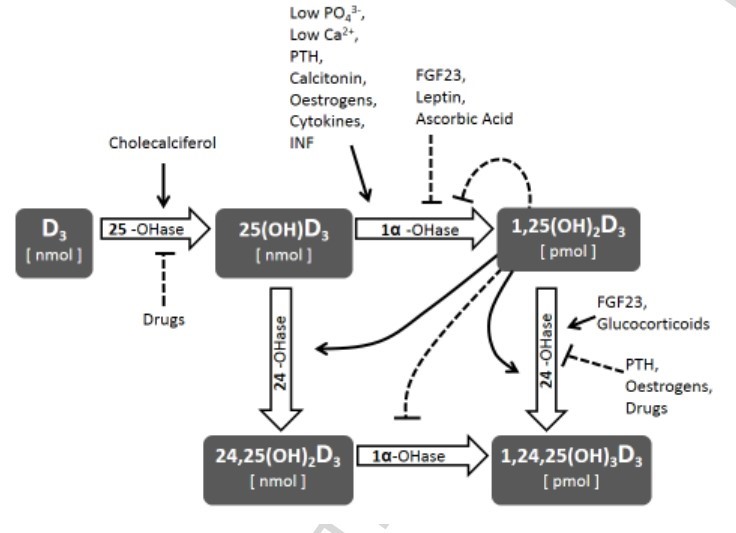
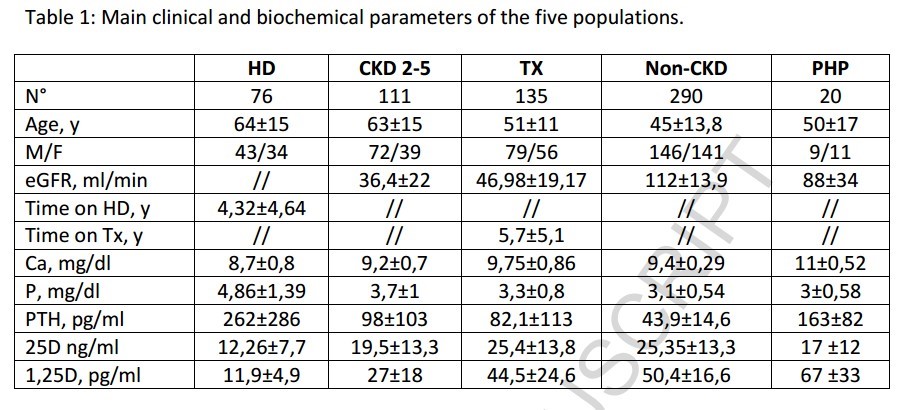
Calcifediol (25D) availability is crucial for calcitriol (1,25D) synthesis, but regulation of vitaminD hydroxylases is the major responsible for 1,25D synthesis. The net efficiency of vitaminD hydroxylases might be informative. We assume that the ratio between calcitriol and calcifediol (25D/1,25D) serum concentrations could suggest the vitaminD hydroxylation efficiency. We evaluated 25D/1,25D in different patient populations: hemodialysis (HD, n = 76), CKD stage 2–5 (n = 111), renal transplant (TX, n = 135), patients with no renal disease (No-CKD, n = 290), and primary hyperparathyroidism (PHP, n = 20). The geometric mean of 1,25D/25D (pg/ng) averaged 1.11 (HD), 1.36 (CKD), 1.77 (TX), 2.22 (No-CKD), and 4.11 PHP, with a progressive increment from HD to PHP (p-value for the trend < 0.001). Each clinical conditions elicited a significant effect on 25D/1,25D (p < 0.0001) and adjusted multivariate analysis indicated levels of Cas, Ps, PTH, and 25D as predictors of 25D/1,25D. Both in vitaminD deficient and replete subjects (25D < or ≥ 20 ng/ml) 25D/1,25D associated with each clinical condition (p < 0.0001) and mean values increased progressively from HD to PHP (p-values for the trend < 0.0001). Regression analysis between 25D (substrate) and 25D/1,25D (efficiency) revealed an exponential negative correlation in No-CKD (r2Exp = 0.53, p < 0.001) with sharp increments of 25D/1,25D when 25D values are < 20 ng/ml. At variance, in CKD (r2lin = 0.19) and in TX (r2lin = 0.32) the regression was linear as if, in case of deficit, some inhibition of the system were operating. In conclusion 1,25D/25D can reflect the efficiency of vitaminD hydroxylases more than separate evaluation of 25D and 1,25D and could facilitate the therapeutic choices in different patient populations.
| Abrev | Description | 1. of patients |
| HD | haemodialysis | 76 |
| CKD 2-5 | (proteinuria >150 mg/dl and eGFR<90 ml/min/1.73 m2 |
111 |
| TX | Transplant at least 6 months prior to enrollment | 135 |
| Non CKD | 290 | |
| PHP | corrected serum Ca >10.5 mg/dl PTH levels >70 pg/ml and history of nephrolithiasis |
20 |
Clipped from PDF
Interestingly, in separate populations, the correlation between PTH and 1,25D/25D ratio was positive in No-CKD patients and negative in those with any type of CRF (HD, TX, CRF).
. . replete CKD patients exhibited a hydroxylation efficiency that, on average, did not significantly differ from HD patients, suggesting that the system is blunted in spite of some residual renal function.
Moving from quantitative data to qualitative behaviour, regression analysis between 25D levels and the 1,25D/25D ratio can theoretically describe the response of hydroxylation to progressive vitamin D depletion; this analysis indicated a negative linear trend in CKD and TX patients and a negative but exponential regression in No-CKD and, unexpectedly, in HD patients. If we consider the behaviour in the No-CKD population as a reference, it is evident from our data that the exponential increment begins when 25D levels decline below 20 ng/ml, consistent with the most commonly adopted threshold for deficiency. In CKD and TX patients, the qualitative response is hampered, consistent with ensuing or persistent SH conditions, respectively. As per the unexpected results suggesting that HD patients behave as No-CKD patients, it is evident from the data that the ratio invariably averages significantly lower values in HD patients compared to any other group and that the exponential increment is observed only when 25D values are approximately undetectable (<5 ng/ml). Assuming that the contribution of renal hydroxylation is trivial in HD patients, we can hypothesise that in this case, the ratio is reflective of the efficiency of extra-renal calcitriol synthesis. It would be interesting to confirm this last observation in anephric patients in whom the circulating calcitriol levels are, by definition, of exclusive extra-renal origin.
