Complex relationship between UVB and vitamin D
The following study is a quick extract from the PDF at the bottom of this page
Bolding was added by VitaminDWiki
Probably presented at the Bermeyer Coonference in Garmishpartenkirchen (Germany) in March 2012
See also VitaminDWiki
- No – 10 minutes per day of sun-UVB is NOT enough
- Which may be to be significantly revised as a result of this paper
Dark skin does not generate as much vitamin D from the same amount of UV – Nov 2011
Patent application for a skin cream which enhances production of vitamin D – Holick July 2013
Vitamin D production after UVB: Aspects of UV-related and personal factors
Scandinavian Journal of Clinical & Laboratory Investigation, 2012; 72(Suppl 243): 24-31
MORTEN K. B. BOGH Bogh@dadlnet.dk
Department of Dermatology, Malmo Hospital, Lund University, Sweden
Correspondence: Morten K.B. Bogh, Department of Dermatology, Malmo Hospital, @skane.se
ISSN 0036-5513 print/ISSN 1502-7686 online © 2012 Informa Healthcare DOI: 10.3109/00365513.2012.681929
Lund University, Sweden
Abstract
During the last decade vitamin D has become a hot topic and our knowledge of its vital role in health and disease is constantly expanding. Solar ultraviolet-B (UVB, 280-320 nm) is both the initiator of vitamin D production in the skin and a § risk factor for sunburn and skin carcinogenesis. At present, this dilemma is debated worldwide. In Northern Europe, it is £; possible to reach a sufficient vitamin D status through sun exposure in the summer months. However, in the winter, the 2 ambient UVB radiation is too low to initiate any production of vitamin D and this has led to a widespread concern and focus on vitamin D status. This review focuses on aspects of UV-related and personal factors of importance for the cutaneous vitamin D production after UVB exposure.
Introduction
Ultraviolet-B radiation (UVB, 280-320 nm) is the only part of the solar UV radiation (UVR, 290-400 nm) spectrum that causes formation of vitamin D in the skin, and is believed to provide more than 90 % of the vitamin D required [1,2]. However, UVB is also known to be an important etiological factor in the development of melanoma as well as non-melanoma skin cancer [3]. In spite of this, sun exposure has been recommended to avoid vitamin D insufficiency [4]. This dilemma has led to a scientific debate [5,6]. A better understanding of the UVB-induced vitamin D production is necessary, as only few objective, prospective studies have addressed the photobiology of vitamin D in vivo [7]. This review focuses on aspects of UV-related and personal factors of importance for the vitamin D production in the skin after UVB exposure, and previous studies are discussed in relation to own research [8-11].
Background
The synthesis of vitamin D3 starts in the bowel epithelium with an oxidation of cholesterol to 7dehydrocholesterol (7-DHC). 7-DHC is then transported to the skin wherein it is photolyzed by UVB radiation to pre-vitamin D3 which is isomerized by heat to vitamin D3 and transported to the liver by a vitamin D binding protein (DBP). Here it is hydroxylated by 25-hydroxylase to 25-hydroxyvitamin D3 (25(OH)D3, calcidiol), which is then transported to the kidneys where it is transformed to the active 1,25-dihydroxyvitamin D3 (1,25(OH)2D3, calcitriol) by 1-alpha-hydroxylase [12,13]. Both pre-vitamin D3 and vitamin D can absorb UV radiation and this can lead to isomerization of these molecules to form inert photoproducts, such as lumisterol, tachysterol and 5,6-transvitamin D3 as well as suprasterol I and II [14,15]. Pre-vitamin D3 is thermodynamically unstable and spontaneously forms vitamin D3 in response to heat. This heat transformation occurs within few hours [16].
As it is not possible to reach toxic vitamin D levels by sun exposure alone, a therapeutic window has been claimed to exist for the conversion of pre-vitamin D3 to vitamin D3 and that excessive amounts of UV radiation will convert the already produced pre-vitamin D3 and vitamin D3 to inert photoproducts [15,17]. The action spectrum for the UV-induced cutaneous conversion of 7-DHC to pre-vitamin D3 was originally published by MacLaughlin in 1982 [14], and was officially defined by the Commission Internationale de l'Eclairage (CIE) in 2006 [2].
The action spectrum shows that 7-DHC absorbs UV radiation at wavelengths starting at about 260 nm with essentially no production above 315 nm and with an optimal synthesis occurring between 295-300 nm. 7-DHC is contained in the cell membranes of keratinocytes and fibroblasts located in the epidermis of the skin.
Solar UVR comprises 90-99 % UVA (320-400 nm) and 1-10 % UVB (280-320 nm). Exposure of human skin to solar UVB radiation is the major source of vitamin D [1]. The ozone layer in the stratosphere almost completely absorbs light of a wavelength below 290 nm whereas that above is only partially absorbed and the UVA not at all. The absorption depends on the zenith angle which varies with the latitude, season and time of day. Thus near the equator at noon the UVB exposure is at its strongest [18].
Anything that decreases or prevents UVB photons from reaching the skin decreases the photosynthesis of vitamin D. Sunscreens, glass and clothes effectively absorb UVB radiation [19]. However, sunscreens have not been shown to block cutaneous vitamin D production in practice, mainly because of inadequacies in application to the skin and because people using sunscreens may have a more extensive sun exposure [20]. Furthermore, there is significant debate regarding whether melanin in the skin diminishes the cutaneous vitamin D production [7].
UV measurement unit
When measuring UV doses, the time component is taken into account, and thus the unit is the irradiance to a given area during a given time. In physical terms exposure is denoted in units J/m2 (J = watt x second). However, when measuring the UV action on the skin the physical dose is weighted by the erythema action spectrum. The units representing the amount of erythema weighted doses are minimal erythemal dose (MED) and standard erythema dose (SED). The literature often uses the unit MED, which is the smallest dose to cause erythema with more or less well-defined borders at 24 h after irradiation of the skin. The MED unit has the advantage that it is directly related to the biological consequences of exposure in a given individual. However, the MED unit is subjective, since the erythemal response varies considerably among individuals. To overcome this problem the standard erythema dose (SED) was established [21].
One standard erythema dose (SED) is defined as 100 J/m2 (10 mJ/cm2 ) at 298 nm using the CIE erythema action spectrum. It is equivalent to the UVR dose needed to provoke a just perceptible erythema in the most sensitive of a group of sensitive people 24 hours after exposure [21]. In Denmark (56 °N), one SED is equivalent to the dose received by approximately 10 minutes of sun exposure at noon on a clear and sunny day in July, with an UV index of 6 . The UV index refers to the UV intensity at noon, and a UV index of 6 corresponds to an erythema-weighted dose of 6 SED in the hour with maximum intensity [22].
Skin pigmentation
The differences in skin pigmentation are due to differences in the amount of melanin in the skin. Melanin is synthesized in the melanocytes by an oxidation of tyrosine and is induced by ultraviolet irradiation. Melanin is stored in melanosomes, which are transferred to keratinocytes. Melanocytes are located in the basal layer of epidermis. To maintain cutaneous pigmentation there is a constant need for synthesis of melanosomes as keratinocytes are desquamated from the skin. The cutaneous pigmentation depends on the activity of melanocytes as the numbers of mel-anocytes are the same in individuals of different ethnic backgrounds [23]. A commonly held view is that when humans migrated north at the end of the latest ice age, pale skin evolved in order to compensate for the less intense solar UVR [24,25]. This vitamin D hypothesis for skin lightening is based on the assumption that dark skin needs more UVB than fair skin to generate a given amount of vitamin D. However, several studies question whether this is the case [26-28], and there is significant debate on whether melanin in the skin affects the UVB-induced vitamin D production [7]. The reason for loss of skin colour as humans moved north is not known and several other reasons for the change in pigmentation has also been hypothesized such as genetic drift, adaptation to colder climates and sexual selection purposes [25].
Skin pigmentation can be determined subjectively or objectively. The Fitzpatrick skin type classification is a subjective self-reported skin type classification based on what a person recalls as a typical reaction to 2 hours of unprotected sun exposure first time in May (in Scandinavia). The system has 4 grades for fair-skinned and 2 grades for dark-skinned individuals [29]. However, the Fitzpatrick skin type classification has been criticized scientifically because of recall bias [30]. An objective measurement of skin pigmentation has been sought to replace the Fitzpatrick classification. Pigment Protection Factor (PPF, measuring interval 1.0-24.0) measured by a skin reflectance measurement system (UV-Optimize 558 system, Chromolight, Matic, Espergaerde, Denmark) is a validated objective alternative to the Fitzpatrick skin type classification [31]. PPF is an objective measure of the photoprotection afforded by skin pigment and stratum corneum. PPF corresponds to the number of SED (standard erythema doses) expected to induce just perceptible erythema by a MED photo-test. PPF indicates tendency to sunburn and predicts the number of SED to 1 MED in an individual. Constitutive skin pigmentation is the skin pigmentation on UVR unexposed skin e.g. the buttock. Facultative skin pigmentation is acquired skin pigmentation obtained by UVR exposure. A typical person with skin type III has a mean PPF value of 3-5 on the buttocks and up to 8-10 on other body locations, depending on previous sun exposure.
Aspects that influences the vitamin D production after UVB
The importance of baseline vitamin D
Previous studies. Previous studies have suggested that individuals with low vitamin D concentrations have a faster vitamin D production after UVB than individuals with already normal or high vitamin D concentrations [32-38]. A faster increase in the vitamin D concentration for individuals with low vitamin D concentration has also been reported after intake of oral vitamin D supplementation [39]. However, no previous studies on UVB and vitamin D have been published to specifically confirm this relation.
Own investigation [8] and discussion.
In order to define the importance of baseline vitamin D, 50 participants with a wide interval in baseline 25(OH)D3 concentrations (5-116 nmol/L) were selected out of 182 participants who initially were screened for 25(OH)D3 concentrations. When exposed to a fixed broadband (BB) UVB dose of 3 SED four times with two or three days' interval to 24 % body area, a significant inverse correlation between the increase in vitamin D concentration after UVB (A 25(OH)D3) and baseline vitamin D concentration was confirmed (P < 0.0001) [8]. The physiological mechanism behind this relation is unknown, but 25(OH)D3 might inhibit 25-hydroxylase in the liver, which prompts the hydroxylation of vitamin D3 to 25(OH) D3. The inverse relation may also be connected to the fact that toxic vitamin D concentrations cannot be reached through sun exposure alone. One could speculate, that when reaching a certain (sufficient) vitamin D concentration, further exposure to the sun may prompt the production of inert photoproducts and thereby turning off the vitamin D production, a mechanism that protects the organism from toxicity of vitamin D. This inverse correlation of baseline vitamin D has later been confirmed in a series of other studies [9-11].
The importance of total cholesterol
Previous studies.
As the production of vitamin D starts with cholesterol (oxidation of cholesterol from food or endogenous to 7-DHC), one would expect that the total cholesterol concentration would influence the vitamin D production after UVB. Accordingly, a recent study has reported a significant positive relation between total cholesterol concentration and baseline vitamin D [40]. However, no previous studies have documented a correlation between total cholesterol and the increase in vitamin D after UVB.
Own investigation [8] and discussion.
To investigate the importance of total cholesterol on the vitamin D production after UVB exposure, a subgroup of 28 non-sun worshippers with limited past sun exposure were selected in order to achieve a more homogeneous population. The 28 participants were exposed to a fixed BB-UVB dose of 3 SED four times with two or three days' interval to 24 % body area [8]. A positive correlation between A 25(OH) D3 and the total cholesterol concentration (measured at baseline) was found (P = 0.005). One might speculate that cholesterol-lowering medication would be problematic in regard to the vitamin D concentration. However, a fall in vitamin D status after statin therapy has never been reported, but several studies have reported increased concentrations of 25(OH)D3 after treatment with statins [40,41]. The mechanism behind this is unknown. To eliminate any confounding factors regarding the relation between statins and vitamin D, none of the participants in the study [8] received cholesterol-lowering medication.
The relation between total cholesterol and A 25(OH)D3 was not confirmed in a series of other studies [9-11], however, a more narrow population regarding baseline vitamin D was used and the population was not divided into subgroups, which may explain the lack of relation. Only a limited number of studies exist exploring the relation between vitamin D and cholesterol and more studies are needed.
The importance of skin pigmentation
Previous studies.
Epidemiologic studies have shown that individuals with dark skin generally have a lower vitamin D status than individuals with fair skin [42,43]. A commonly held view is that increased melanin requires increased doses of UVB exposure to produce equivalent vitamin D, thereby providing an explanation for the low vitamin D status in individuals with dark skin [17,44-48]. However, a number of studies have found a similar vitamin D increase regardless of differences in skin pigmentation [26-28,49,50]. Accordingly, there is a significant debate on whether skin pigmentation affects the UVB induced vitamin D production. It has been proposed, that low vitamin D status among certain ethnic groups could be due to other factors such as sun behaviour, genes or diet [51].
Own investigation [8] and discussion.
To investigate the importance of skin pigmentation on the vitamin D production after UVB exposure, a homogenous group of 28 non-sun worshippers with a wide variation in skin pigmentation (Fitzpatrick skin type I-VI [29]) were used, and no significant relation between A 25(OH)D3 and constitutive (P = 0.5) or facultative (P = 0.4) skin pigmentation were found, when using a fixed BB-UVB dose of 3 SED four times with two or three days ' interval to 24 % body area [8]. As described previously, the importance of baseline vitamin D must be taken into account. Pairs of participants with fair skin and dark skin with matched baseline 25(OH)D3 concentrations were therefore created, and no significant difference in the increase in 25(OH)D3 after identical UVB exposure were found (P = 0.7), despite significant difference in skin pigmentation (P = 0.008) and no significant difference in baseline 25(OH)D3 concentration (P = 0.07) [8].
(No difference in vitamin D produced with skin color)
An ex vivo study from 1981 by Holick et al. [17] on isolated human skin samples, was one of the first studies reporting that melanin is of importance for limiting the cutaneous production of vitamin D. This observation was later reproduced in a small in vivo study from 1982 by Clemens et al. [45], who found a vitamin D increase in the fair-skinned (n= 2) but not in the dark-skinned (n = 3). Similar observations were reported by Matsouka et al. in 1991 [46], who exposed different ethnic groups (n = 7-8) categorized as "white", "oriental", "Indian" and "black". Two recent in vivo studies by Armas et al. [47] and Chen et al. [48], have also reported that vitamin D production is more efficient in fair-skinned individuals. However, the importance of skin pigmentation has not been confirmed in a number of other studies. Stamp [26], Lo et al. [27], Brazerol et al. [28], and Marks et al. [49], found a similar vitamin D increase in spite of differences in skin pigmentation. Furthermore, Rockell et al. [50] found that only facultative skin colour is a determinant of vitamin D production and claim that constitutive skin type is of no importance. Conclusively, the role of skin pigmentation on vitamin D status is unclear . Additionally, the baseline vitamin D should be taken into account [7,8]. However, our own research discussed above also contains limitations [8]. The fact that the study was done in the winter, when the melanin is located in the basal layer of epidermis, should be taken into account. An effect of skin pigmentation may have been found in the summer, when sun exposure stimulates the production of melanin, which then moves higher up in the epidermis. Another important factor, hypothesized by Bjorn [52], is the use of the TL 12 UV source, which contains shortwave components < 290 nm. However, Solar UVR contains no radiation below 290 nm. As the action spectrum for pre-vitamin D3 starts at about 260 nm, the vitamin D production may therefore be independent of skin pigmentation only with UV of short wavelengths, but not by solar UVR emitting wavelengths above 290 nm. Furthermore, the use of a small sample size and the variance of 8.5 % found in the vitamin D analysis, despite including two serum samples from each individual and performing the analysis twice, may have confounded the results. Finally, each participant was exposed to four UVB doses of 3 SED. This dose may have been sufficient to reach a state of saturation in fair-skinned, and using a smaller fixed UVB dose may have been a more logical approach.
The importance of UVB dose and dose rate
Previous studies. Although there are several ex vivo studies about the dose-response relationship between vitamin D and UVB radiation [17,44,53], there exits only few human in vivo studies [36,47,54]. The majority of these studies used MED and thereby treated the lighter skin tones with lower UV-doses than those for darker skin tones, assuming that skin pigmentation is of importance for the UV-induced vitamin D production. Consequently, it is difficult to draw firm conclusions from these studies because of prejudice.
Despite the basic photobiological fact that skin carcinogenesis and erythema are known not to depend on dose rate over a wide interval of irradiances [55-57], no study has investigated the relationship between the UVB induced vitamin D production and dose rate.
Own investigation [9] and discussion.
To investigate the importance of UVB dose, 55 fair-skinned participants (Fitzpatrick skin type I-IV [29]) with a relatively narrow interval of 25(OH)D3 concentrations (16-50 nmol/L) were randomized to four BB-UVB exposures with two or three days' interval to 24 % body area with either 0.375, 0.75, 1.5 or 3.0 SED. Notably, a fixed UVB dose (SED) regardless of skin pigmentation was used, instead of using MED which depends on skin pigmentation. A significant positive relation was found between A 25(OH)D3 (adjusted for baseline 25(OH)D3) and the UVB dose (P = 0.001). The 25(OH)D3 concentration increased by 24.8 nmol/L (mean) after a total UVB dose of 12 SED (4 X 3 SED) and by 14.2 nmol/L after a total UVB dose of just 1.5 SED (4 X 0.375 SED) [9]. The data shows that a very small UVB dose results in significant 25(OH)D3 production.
In addition, the importance of dose rate was investigated. To define the relationship between A 25(OH)D3 and dose rate, the 55 participants were randomized to receive a given UVB dose during 1, 5, 10 or 20 minutes. The dose rate was found to be of no importance to A 25(OH)D3 (P = 0.8) [9]. The dose rates (1-20 minutes) were chosen as this time frame was considered the most relevant duration of sun exposure with regard to normal everyday life. This is presumably the first study exploring this relationship.
The importance of body area
Previous studies. Only few human studies deal with the importance of body area with regard to the UVB induced vitamin D production [19,58,59]. Barth et al. [58] irradiated different body areas and reported increasing vitamin D concentrations with increasing body areas, whereas Matsuoka et al. [19] suggested that the vitamin D response after UVB reaches a plateau when more than 33 % of the body area is irradiated. However, both studies used MED. As previously discussed it can be considered problematic to use MED, an individual biological dose based on the assumption that skin pigmentation reduces the vitamin D production after UVB. A recent study by Vahavihu et al. [59] with narrow-band (NB) UVB (TL 01 lamps, with an emission peak at 311 ± 2 nm) used SED and irradiated three different body areas and found a similar increase in vitamin D after whole body exposure and after exposure of only head and arms
Own investigation [10] and discussion. To investigate the relation between the UVB induced vitamin D production and body area, 92 fair-skinned participants (Fitzpatrick skin type I-IV [29]) were randomized to having 6 %, 12 % or 24 % of their skin exposed four times with two or three days' interval to a fixed BB-UVB dose of 0.75, 1.5 or 3.0 SED. When all 92 participants were included in the statistical analyses, a significant positive relation was found between A 25(OH)D3 and the UVB dose (P < 0.0001) and between A 25(OH)D3 and the exposed body area (P = 0.006) [10]. However, partitioning the UVB dose and exposed body area disclosed a state of saturation as detailed in Table I.
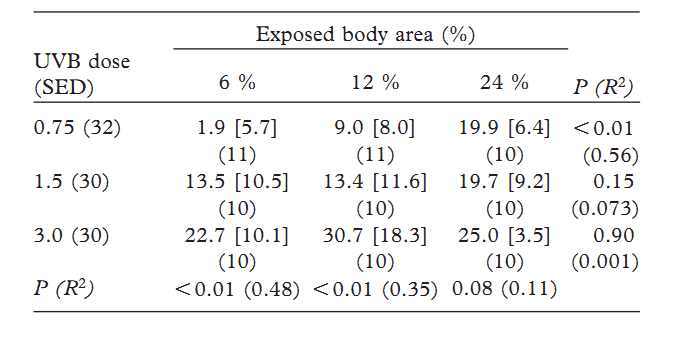
Table I. Increase (mean) in 25(OH)D3 (nmol/L) after exposure to UVB (0.75 - 3.0 SED) of different body areas (6 % - 24 %).
A total of 92 participants were included.
The number of participants is given in brackets and standard deviations in square brackets. Modified from [10].
There is a considerable inter-individual variation in the vitamin D production after UVB exposure, there are many possible explanations but the mechanisms are not presently fully understood and require further investigation [8-11,36].
Notably, exposure to higher UVB doses and to larger body areas gives a less favourable UV risk-benefit ratio. Accordingly, Matsuoka et al. [19] and Vahavihu et al. [59] also found a state of saturation in the vitamin D production with increasing body areas. The benefit/risk ratio seems only favourable for small UVB doses and body areas whereas excessive sun exposure does not provide proportionally further benefit. As it is known that toxic vitamin D concentrations cannot be reached through sun exposure alone, a question arises whether there exists a certain vitamin D concentration, where further sun exposure induces the production of inert photoproducts and thereby turns off the vitamin D production. Another explanation could be, that there is a certain saturation point in the capacity to hydroxylate vitamin D3 to 25(OH)D3 in the liver, where increasing concentrations of 25(OH)D3 might inhibit the function of the 25-hydroxylase. However, the reason behind the mechanism is unknown.
The importance of UVB exposure frequency
Previous studies.
In Northern Europe, it is possible to reach a sufficient vitamin D status through sun exposure in the summer months. However, in winter (from approximately October to April) the ambient UVB radiation is negligible, the outdoor activity too low, and the clothes covering the body too extended to initiate and maintain a sufficient vitamin D concentration [18]. A recent study shows that the majority of the population in the UK becomes vitamin D insufficient during the winter [60]. However, the UVB exposure frequency for maintaining the summer vitamin D status during winter is not previously investigated.
Own investigation [11] and discussion.
To clarify how summer vitamin D concentrations can be maintained through UVB exposure, 60 participants were included in the beginning of the autumn to a 16-week long course of UVB therapy. The participants were randomized for BB-UVB exposure of 1 SED to -88 % body area once a week, every second week or every fourth week. The controls had no intervention. Concentration of 25(OH)D3 was measured at baseline and every fourth week. The mean 25(OH)D3 concentrations after UVB exposure once a week increased significantly (71.9 to 84.5 nmol/L) (P < 0.0001) whereas UVB exposure every second week maintained 25(OH)D3 concentrations (P = 0.16). A significant decrease in mean 25(OH)D3 concentrations were found after UVB exposure once every fourth week (56.4 to 47.8 nmol/L) (P< 0.0001) and for the control group (64.8 to 40.1 nmol/L) (P < 0.0001). In conclusion, a suberythemal UVB dose of 1 SED every second week to -88 % body area is sufficient for maintaining summer 25(OH)D concentrations during winter.
Conclusions
Baseline vitamin D appears to be an important determinant of the UVB induced vitamin D synthesis. This should be taken into account in the design of future studies. A relation between the cutaneous vitamin D production and total cholesterol concentration may exist, but more studies are needed before any conclusions can be made. Vitamin D photosynthesis seems to be independent of winter skin pigmentation for a fixed UVB dose and similar baseline vitamin D concentration. However, the role of skin pigmentation is complex and requires further investigation [8].
The UVB induced vitamin D increase depends on the UVB dose but not on dose rate (1-20 min). A significant vitamin D increase can be achieved with a very low UVB dose [9].
The size of the UVB exposed body area is important only for the small UVB doses. Notably, exposure with a very low UVB dose to a very small body area is sufficient for significant vitamin D production [10].
A small suberythemal UVB dose every second week to full body area is sufficient to maintain summer vitamin D concentrations through wintertime [11].
Questions and Answers
P Brannon, USA
You did not mention adiposity. Did you measure BMI and consider if any of the individual variability could be explained by BMI and inferred adiposity?
M Bogh
We also measured weight and height and calculated BMI and found no relationship to the individual variability.
P Lips, Netherlands
I was puzzled by the relationship between cholesterol and 25(OH)D3 concentrations., because there is a huge amount of cholesterol in the circulation in relation to vitamin D. Statin users show increased concentrations of 25(OH)D3.
M Bogh
There are a limited number of studies on cholesterol and vitamin D. Our study has shown a significant positive relationship between total cholesterol and baseline vitamin D.
G Jones, USA
Regarding the possible effects of statins and cholesterol. 7-dehydrocholesterol is a precursor of vitamin D but comes off the cholesterol synthesis pathway prior to the formation of cholesterol itself. So is what you are measuring a surrogate for skin 7-dehydro-cholesterol? Has anybody tried to measure it in skin?
M Bogh
It would be interesting to measure this marker in skin and blood.
Acknowledgement
I wish to thank Professor Hans Christian Wulf for scientific guidance during my research from 2007 to 2010 at Department of Dermatology, Copenhagen University Hospital, Denmark.
Conflict of interest: None declared.
References
[1] Holick MF. Sunlight and Vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr 2004;80:1678-1688.
[2] Bouillon R, Eisman J, Garabedian M et al. Action spectrum for the production of pre-vitamin D3 in human skin. Commission Internationale de l'Eclairage (CIE). Technical Report CIE 2006;174:1-12.
[3] Armstrong BK, Kricker A. The epidemiology of UV induced skin cancer. Photochem and Photobiol 2001;63:8-18.
[4] Holick MF. Sunlight, UV-radiation, vitamin D and skin cancer: How much sunlight do we need? Adv Exp Med Biol 2008;624:1-15.
[5] Samanek AJ, Croager EJ, Gies P et al. Estimates of beneficial and harmful sun exposure times during the year for major Australien population cities. Med J Aust 2006;184:338-341.
[6] Reddy KK, Gilchrest BA. What is all this commotion about vitamin D? J Invest Dermatol 2010;130:321-326.
[7] Springbett P, Buglass S, Young AR. Photoprotection and vitamin D status. J Photochem Photobiol B. Biol 2010; 101(2): 160-168.
[8] Bogh MK, Schmedes AV, Philipsen PA, Thieden E and Wulf HC. Vitamin D production after UVB exposure depends on baseline vitamin D and total cholesterol but not on skin pigmentation. J Invest Dermatol 2010;130(2):546-553.
[9] Bogh MK, Schmedes AV, Philipsen PA, Thieden E and Wulf HC. Vitamin D production depends on ultraviolet-B dose but not on dose rate: a randomized controlled trial. Exp Dermatol 2011;20(1):14-18.
[10] Bogh MK, Schmedes AV, Philipsen PA, Thieden E and Wulf HC. Interdependence between body surface area and UVB dose in vitamin D production: a randomized controlled trial. Br J Dermatol 2011;164:163-169.
[11] Bogh MK, Schmedes AV, Philipsen PA, Thieden E and Wulf HC. A small suberythemal ultraviolet B dose every second week is sufficient to maintain summer vitamin D levels: a randomized controlled trial. Br J Dermatol 2012;166:430-33.
[12] Audran M. The physiology and patophysiology of vitamin D. Mayo Clin Proc 1985;60:851-866.
[13] Champe PC, Harvey RA, Ferrier DR (eds). Biochemistry. 3rd edn. Philadelphia:Lippincott, Williams and Wilkins, 2005, p 534.
[14] MacLaughlin JA, Anderson RR, Holick MF. Spectral character of sunlight modulates photosynthesis of pre-vitamin D3 and its photoisomers in human skin. Science 1982;216: 1001-1003.
[15] Webb AR, DeCosta BR, Holick MF. Sunlight regulates the cutaneous production of vitamin D3 by causing its photo-degradation. J Clin Endocrinol Metab 1989;68:882-887.
[16] Tian XQ, Chen TC, Matsuoka LY et al. Kinetic and ther-modynamic studies of the conversion of previtamin D3 to vitamin D3 in human skin. J Biol Chem 1993;15; 268(20):14888-14892.
[17] Holick MF, MacLaughlin JA, Doppelt SH. Regulation of cutaneous previtamin D3 photosynthesis in man: skin pigment is not an essential regulator. Science 1981;211:590-593.
[18] Webb AR, Engelsen O. Calculated ultraviolet exposure levels for a healthy vitamin D status. Photochem Photobiol 2006;82:1697-1703.
[19] Matsuoka LY, Wortsman J, Hollis BW. Use of topical sunscreen for the evaluation of regional synthesis of vitamin D3. J Am Acad Dermatol 1990;22:772-775.
[20] Norval M, Wulf HC. Does chronic sunscreen use reduce vitamin D production to insufficient levels? Br J Dermatol 2009;161(4):732-736.
[21] Diffey BL, Jansen CT, Urbach F, Wulf HC. Standard erythema dose:a review. Commission Internationale de l'Eclairage (CIE). Technical Report CIE 1997;125:1-5.
[22] Wulf HC, Eriksen P. UV index and its implications. Ugeskr Laeger 2010;172(17):1277-1279.
[23] Westerhof W. The discovery of the human melanocyte. Pigment Cell Res 2006;19:183-193.
[24] Murray FG. Pigmentation, sunlight and nutritional disease. Am Anthropol 1934;36:438-445.
[25] Juzeniene A, Setlow R, Porojnicu A et al. Development of different human skin colours. A review highlighting photo-biological and photophysical aspects. J Photochem PhotobiolB 2009;96:93-100.
[26] Stamp TCB. Factors in human vitamin D nutrition and in the production and cure of classical rickets. Proc Nutr Soc 1975;34:119-130.
[27] Lo CW, Paris PW, Holick MF. Indian and Pakistani immigrants have the same capacity as Caucasians to produce vitamin D in response to ultraviolet radiation. Am J Clin Nutr 1986;44:683-685.
[28] Brazerol WF, McPhee AJ, Mimouni F et al. Seriel ultraviolet B exposure and serum 25 hydroxyvitamin D response in young adult American blacks and white: no racial differences. J Am Coll Nutr 1988;7:111-118.
[29] Fitzpatrick TB. The validity and practicality of sun-reactive skintypes I through VI. Arch Dermatol 1988;124:869-871.
[30] Snellman E, Jansen CT, Leszczynski K et al. Ultraviolet erythema sensitivity in anamnestic (I-IV) and phototested (1-4) Caucasian skin phototypes: The need for a new classification system. Phtochem Photobiol 1995;62 (4):769-772.
[31] Wulf HC. Method and apparatus for determining an individual's ability to stand exposure to UV. US patent 1986;14: 882,598:1-32.
[32] Viljakainen HT, Palssa A, Karkkainen M et al. How much Vitamin D do the elderly need? J Am Coll Nutr 2006;25: 429-435.
[33] Binkley N, Novotny R, Krueger D et al. Low vitamin D status despite abundant sun exposure. J Clin Endocrinol Metab 2007;92(6):2130-2135.
[34] Brustad M, Edvardsen K, Wilsgaard T et al. Seasonality of UV-radiation and Vitamin D status at 69 degrees north. Photochem Photobiol Sci 2007;6:903-908.
[35] Edvardsen K, Brustad M, Engelsen O et al. The solar UV radiation level needed for cutaneous production of Vitamin D in the face. A study conducted among subjects living at a high latitude [68 °N]. Photochem Photobiol Sci 2007;6:57-62.
[36] Thieden E, Jergensen HL, Jergensen NR et al. Sunbed radiation provokes cutaneous vitamin D syntheses in humans. A randomized controlled trial. Photochemistry and Photobiology 2008;84:1487-1492.
[37] Moan J, Lagunova Z, Cicarma E et al. Sunbeds as vitamin D sources. Photochem Photobiol 2009;85:1474-1479.
[38] Rhodes LE, Webb AR, Fraser HI et al. Recommended summer sunlight exposure levels can produce sufficient (a 20 ng ml-1) but not the proposed optimal (a 32 ng ml-1) 25(OH)D levels at UK latitudes. J Invest Dermatol 2010;130(5):1411-1418.
[39] Stephenson A, Brotherwood M, Robert R et al. Cholecalcif-erol significantly increases 25-hydroxyvitamin D concentrations in adults with cystic fibrosis. Am J Clin Nutr 2007;85:1307-1311.
[40] Perez-Castrillon JL, Abad L, Vega G et al. Effects of ator-vastatin on vitamin D levels in patients with acute ischemic heart disease. Am J Cardiol 2007;99:903-905.
[41] Dobs AS, Levine, MA, Margolis S. Effects of Pravastatin, a New HMG-CoA Reductase Inhibitor, on Vitamin D Synthesis in Man. Metabolism 1991;40(5):524-528.
[42] Harris SS, Dawson-Hughes B. Seasonal changes in plasma 25-hydroxyvitamin D concentrations of young American black and white women. Am J Clin Nutr 1998;67:1232-1236.
[43] Nesby-O'Dell S, Scanlon KS, Cogswell ME et al. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: Third National Health and Nutrition Examination Survey, 19881994. Am J Clin Nutr 2002;76:187-192.
[44] Holick MF, MacLaughlin JA, Clark MB et al. Photosynthesis of pre-vitamin D3 in human skin and the physiologic consequences. Science 1980:210:203-205.
[45] Clemens TL, Adams JS, Henderson SL, Holick MF. Increased skin pigment reduces the capacity of skin to synthesis vitamin D3. Lancet 1982;319:74-76. [46] Matsuoka LY, Wortsman J, Haddad JG et al. Racial pigmentation and the cutaneous synthesis of vitamin D. Arch Dermatol 1991;127(4):536-538.
[47] Armas LAG, Dowell S, Akhter M et al. Ultraviolet-B radiation increases serum 25-hydroxyvitamin D levels:The effect of UVB dose and skin color. J Am Acad Dermatol 2007; 57:588-593.
[48] Chen TC, Chimeh F, Zhiren L et al. Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch Biochem Biophys 2007;460:213-217.
[49] Marks R, Foley PA, Jolley D et al. The effect of regular sunscreen use on vitamin D levels in an Australian population. Arch Dermatol 1995;131:415^21.
[50] Rockell JEP, Skeaff CM, Williams SM, Green TJ. Association between quantitative measures of skin colour and plasma 25-hydroxyvitamin D. Osteopor Int 2008;19(11):1639-1642.
[51] Young AR. Photobiology of Melanins. In:The pigmentary system, 2nd edn (Nordlund JJ, Boissy RE, Hearing VJ, King RA, Oetting WS, Ortonne JP, eds). Oxford:Blackwell Publishing, 2006, pp. 342-353.
[52] Bjorn LO. Vitamin D synthesis may be independent of skin pigmentation only with UV of short wavelength. J Invest Dermatol 2010;130:2848-2850.
[53] Olds WJ, McKinley AR, Moore MR et al. In vitro model of vitamin D3 (Cholecalciferol) synthesis by UV radiation:Dose-response relationships. Photochem Photobiol 2008;93:88-93.
[54] Chel VG, Ooms ME, Popp-Snijders C et al. Ultraviolet irradiation corrects vitamin D deficiency and suppresses secondary hyperparathyroidism in the elderly. J Bone Miner Res 1998;13:1238-1242.
[55] Blum HF. Action spectra and absorption spectra. In Carcino-genesis by ultraviolet light (Butler EG, Theodosius D, Sterling B et al (eds). Princeton University Press, 1959. pp. 23-30.
[56] Norins AL. Immediate Effects of Light. In The Biologic Effects of Ultraviolet Radiation - with emphasis on the skin (Urback F, eds.). Pergamon Press, Oxford;1969. pp. 31-36.
[57] Kaase H. Basic physical aspects of environmental UV radiation. In Skin cancer and UV radiation (Altmeyer P, Hoffmann K, Stucker M eds). Springer-Verlag, Berlin:1997. pp. 31-36.
[58] Barth J, Gerlach B, Knuschke P et al. Serum 25(OH)D3 and ultraviolet exposure of residents in an old people's home in Germany. Photodermatol Photoimmunol Photomed 1993;9: 229-231.
[59] Vahavihu K, Ylianttila L, Kautiainen H et al. Narrowband ultraviolet B course improves vitamin D balance in women in winter. Br J Dermatol 2010;162:848-853.
[60] Webb AR, Kift R, Durkin MT et al. The role of sunlight exposure in determining the vitamin D status of the U.K. whiteadult population. Br J Dermatol 2010;163:1050-1055.
