Vitamin D and multiple sclerosis—from epidemiology to prevention
Sundstrom P, Salzer J.
Acta Neurol Scand 2015: 132 (Suppl. 199): 56-61.
In the present review, we discuss observational and experimental data suggesting a protective effect from sun exposure and/or vitamin D in multiple sclerosis (MS). These data include geographic variations in MS occurrence, temporal trends, genetics, biobank, and questionnaire data. We look more closely at the differentiation between general effects from UV exposure, and those of vitamin D per se, including plausible mechanisms of action. Finally, primary prevention is touched upon, and we suggest actions to be taken while awaiting the results from ongoing randomized controlled trials with vitamin D in MS
The basis for the sunshine hypothesis in multiple sclerosis (MS) consists of epidemiological data on MS incidence and prevalence. These geographic variations in MS occurrence were commented upon already by Charcot and were early interpreted to show an increasing MS occurrence with increasing distance from the equator (1). Together with data from migration studies, an environmental factor in the etiology of MS was strongly suggested (reviewed in (2)).
📄 Download the PDF from VitaminDWiki
Sunshine and MS epidemiology
The most natural factor related to the distance from the equator is sunshine—exposure to UV radiation. This notion has been considerably reinforced by studies showing that latitude is a poor proxy for UV radiation and that satellite-derived data on UV intensity, which provide a more accurate estimate of UV radiation, show a stronger association with MS occurrence (3-5).
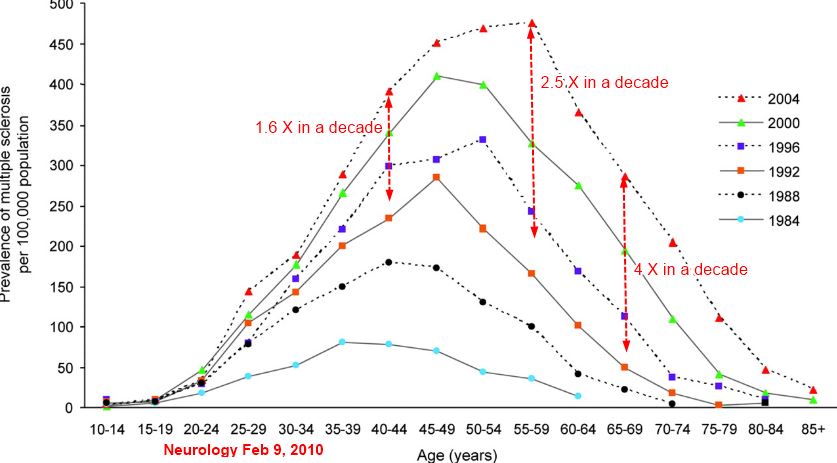
The latitude gradient in MS has been questioned recently (6), which has thrown some researchers into doubt about the sunshine hypothesis. The majority of MS epidemiological studies have studied prevalence. Prevalence data are less informative when etiological factors are considered, as the prevalence depends not only on the incidence in the last decades, but for chronic diseases like MS also on the mortality. Incidence studies are more difficult to perform and generally contain fewer cases but are superior for generating etiological hypotheses. It has been proposed that while MS prevalence data still show a latitude gradient, global MS incidence data, including Europe, do not, with the exception that the incidence gradient is still apparent in Australia and New Zealand (6).
A recent meta-analysis showed that a latitude gradient of MS incidence in Europe does indeed exist (7), and the same tendency was noted in another meta-analysis (8). Two temporal trends were observed regarding the MS incidence: (i) a general increase in MS incidence (6, 7) and (ii) a decreased difference between geographic areas earlier attributed a high and low MS risk, such as northern vs southern Europe (7). Chile is a well- suited country for studies on latitude gradients due to its elongated shape, and in one study, the incidence of hospitalizations because of MS was compared between regions (9). The authors reported no association between the incidence and latitude or UV radiation index. However, this was true after exclusion of the southernmost region, whichwas done because of different ancestry of inhabitants. If this region was included, the associations became significant.
One interpretation is that the MS incidence increase (given the likely assumption that this is true and not only ascertainment related) is higher in geographic regions previously attributed a lower incidence, thus catching up with regard to regions with higher incidence. Such an equalization of MS incidence may relate to an equalization regarding UV exposure. A series of lifestyle changes characteristic for the Western world seem to abolish latitude effects on UV radiation exposure. These changes include increased sun avoidance and use of skin protection following anti-sun exposure propaganda, increased charter tourism to warmer countries during the winter, and increased time indoors both at work and during leisure time. As discussed below, other lifestyle changes have also occurred (increased obesity, changed smoking habits, vitamin D fortification of food) that may also level out natural differences of vitamin D levels related to UV exposure. These Western world lifestyle changes offer a plausible explanation for the changes seen in the epidemiological pattern of MS.
Furthermore, deviations from the expected MS epidemiological global pattern implied by a protective effect from UV exposure may be explained by vitamin D-related mechanisms. Two examples are the unexpected low prevalence in northern Norway that could be explained by a high fish intake (10) and the unexpected high prevalence in Sardinia that may relate to a different vitamin D- related genetic background (11).
Vitamin D and MS association studies
That higher vitamin D levels are associated with less MS inflammatory activity has been shown in several publications (12-16). The strength and temporality of the association argue against reverse causation (that those with less active MS spend more time outdoors). For example, one study showed a 57% risk decrease for new active MRI lesions for each 50 nmol/l increase in 25 (OH) vitamin D levels (16). This association may represent some other anti-inflammatory effect on MS associated with sun exposure, but was seen also in an MS population on vitamin D supplementation (17).
More interesting from an etiological point of view are the two studies on presymptomatic serum samples that have shown a 61 -62% risk reduction for subsequent MS development for individuals with higher 25-hydroxy-vitamin D (25 (OH)D) levels (>75 vs <75 nmol/l and highest vs lowest quintile, respectively) (18, 19). Even more challenging are the post hoc-derived data for the younger subgroup in each study (<26.4 years and <20 years, respectively), which show an 84-91% risk reduction for MS. This protective effect was not present in samples taken at birth (20) or during pregnancy (19). Taken together, this suggests that childhood or adolescence is the crucial time period for vitamin D-dependent MS risk acquisition. There are also some data on vitamin D (21) or fish intake (10, 22) suggesting that higher intake of vitamin D per se is associated with a decreased risk of MS.
So far, the randomized controlled trials (RCT) on vitamin D supplementation in MS have been inconclusive (reviewed in (23)), but several trials are ongoing (reviewed in (24)). Vitamin D supplementation may protect against upper respiratory tract infections, however, only in populations with low baseline levels (25, 26). Interestingly, the protective effect from such supplementation appeared after more than 6 months of medication, suggesting that in addition to an adequate dosage of vitamin D, sufficient time on supplementation is needed (25).
Vitamin D and other environmental MS risk factors
Vitamin D fits well into the complex interplay between risk factors that emerges from observational studies. Epstein-Barr virus (EBV) stands out from the infectious agents because of an array of ties to MS (reviewed in (27)). An interaction between EBV and UV exposure/vitamin D in MS etiology has been suggested in several studies (28-30). These two factors may explain most (72%) of the variance of MS prevalence in England (28).
Cigarette smoking and obesity are two recognized risk factors for MS that have both been associated with lower vitamin D levels (31, 32). A recently suggested protective factor for MS is alcohol consumption (33), and in a Finnish study, alcohol consumption was associated with higher vitamin D levels (34). Shift work before the age of 20 years increased the risk of MS in one study, possibly because of the resulting lower vitamin D levels (35). Sun exposure during childhood and adolescence has a beneficial effect on MS risk (36), and obesity also seems to act as a risk factor for MS during similar ages (37). Thus, studies of other environmental risk factors give further support for the sunshine/vitamin D hypothesis in MS etiology and indicate childhood adolescence as the critical age.
Vitamin D and genetic MS risk factors
Genetic support for vitamin D in MS (reviewed in (38, 39)) may be divided into four groups of observations:
CYP27B1 encodes 1-a-hydroxylase that converts 25(OH)D to its active form, and a twin study identified two CYP27B1 variants that were associated with 25(OH)D levels (40). Also, an increased risk of MS in persons with extremely rare CYP27B1 loss-of-function variants has been shown (41), but not replicated (42). The MS GWAS published in 2011 identified as candidate genes CYP27B1 (although another polymorphism), and also
CYP24A1, which encodes 24-hydroxylase that degrades 25(OH)D as well as its active form (43). CYP24A1 (yet another polymorphism) was further identified in a GWAS searching for genetic determinants for 25(OH)D insufficiency (44).
The receptor for active vitamin D when entering cells is the vitamin D receptor (VDR). VDR polymorphisms have been associated to MS risk in an Australian population (45). The same polymorphism was confirmed in an UK/ Australian population and an interaction with HLA DRB1*1501 status was suggested (46), but these findings could not be reproduced in a Spanish population (47).
VDR binding sites (vitamin D responsive elements [VDREs]) are present on the DNA and affect transcription of the corresponding genes (38). An enrichment of VDR binding sites at genes associated with autoimmune diseases has been shown (48), and an association with MS genes including the chromatin state was shown by the same group in an updated analysis highlighting the influence of vitamin D on the immune system (49).
Vitamin D immunological effects
Vitamin D and its analogs have numerous effects on the human body. The most well-known and characterized role is in calcium and skeletal homeostasis, where vitamin D is essential for calcium absorption and reabsorption, as well as bone matrix remodeling (50). Other effects include antiproliferation, which suggests a role in malignancy prevention, and effects on muscle strength (50, 51). This section will focus on a fourth role, namely immune modulation.
Evidence for vitamin D effects on the immune system comes from several types of studies (e.g., in vitro, murine, human, etc.). The mechanism of action for vitamin D is through regulation of the transcription rate of genes. After pairing with the VDR at the cell surface, 1,25-dihydroxyvitamin D internalizes into the cell, is transported to the cell nucleus, and forms complexes with the retinoic acid X receptor before binding to VDREs on the DNA (52). This affects the transcription rate of genes, which are thus considered vitamin D regulated. These VDREs are more often exposed (‘open’ chromatin state) in immune cells compared with non-immune cells and also more often found within genomic regions associated with MS (49). The expression of the strongest associated MS- allele, HLA-DRB1*1501, is regulated by vitamin D via such a VDRE in the promotor region (53). It was recently shown that physiological vitamin D levels affect expression of genes related to immune regulation (54). On the cellular level, vitamin D stimulation influences the rate of apoptosis, energy metabolism, immune cell differentiation, signal molecule expression, and transmigration via the blood-brain barrier (39).
As for effects on the immune phenotype, vitamin D stimulation results in a vast number of alterations, most of which are projected to be beneficial in MS (52). Upon vitamin D stimulation, myeloid cells (monocytes, macrophages, and dendritic cells) produce less pro-inflammatory (e.g., IL-1, IL-12, TNF) cytokines and more IL- 10, leading to indirect effects on lymphoid cells with less TH1 and TH17 cell differentiation, and more TReg cell differentiation as the net result. CD4+ T lymphocytes are also directly influenced by vitamin D toward the same end-results, whereas B cells secrete less antibodies and become less prone to differentiate into plasma cells. The results from the ongoing RCTs may shed more light on immunological effects of vitamin D in MS (24).
Sunshine vs vitamin D
The Gordian knot in MS causation, hoped to be cut, or at least severed, by the ongoing RCTs, is the differentiation between vitamin D-independent effects of sunlight exposure and direct vitamin D effects (which potentially can be mimicked by oral supplementation). Epidemiological MS data and observational data showing an association between sun exposure or vitamin D levels and MS can be caused by either non-vitamin D sun exposure effects, by the vitamin D generated by such sun exposure, or by a combination of both.
In an experimental setting, the induction of TRegs was shown to take place both with dermally applied 1,25-dihydroxyvitamin D ointments and by UV radiation in VDR knockout mice (55).
Another study on experimental autoimmune encephalitis (EAE) showed that these UV-induced TRegs migrated into the CNS where they mitigated the inflammatory activity (56). This is an important finding as it demonstrates a link between cutaneous UV radiation and immunomodulatory effects in the CNS.
One study where vitamin D intake, as recorded by questionnaires, was associated with a ~40% lower risk of MS (21), and another study showing that high-dose (2000 IU per day) oral vitamin D during childhood was associated with a lower risk of type 1 diabetes (57), argue that orally taken vitamin D may be effective to prevent autoimmune diseases. When it comes to more solid proof of concept for prevention of disease with vitamin D supplementation, this may be coming with the much anticipated results from the VITAL study (The VITamin D and OmegA-3 TriaL) (58). In this study, >25,000 men and women >50 years of age have been randomized to either placebo, omega-3 fatty acids, 2000 IU vitamin D daily, or a combination of both. Outcomes include cancer incidence, and cardiovascular (CVD) and inflammatory diseases. However, the study design (e.g., age, size) does not allow detection of an effect on MS risk, and given the 5-year duration and recent study initiation, results are many years away.
Vitamin D and MS prevention
As of today, it is the authors’ view that there are not enough data to recommend oral vitamin D supplements for MS primary prevention. Although many observations, as discussed earlier, support a crucial role for vitamin D in MS pathogenesis, especially during adolescence/young adulthood, firm evidence for a protective effect is still lacking. Also, the differentiation between vitamin D and non-vitamin D sun exposure effects still evades us. If a primary preventive study were to be conducted, it would need to take into account not only all the other factors associated with MS risk (genes, EBV, smoking, obesity, sun exposure), but also the timing, dosing, and duration of supplementation. Although the time span where vitamin D levels probably have the most pronounced effect on MS risk has narrowed down to >0 to <25 years of age (18-20), this still means that such a study would need around 30-40 years duration to stretch from acquisition of risk over the latency period and past the average age of disease onset. Further clarifying when in life supplementation may have the greatest effect, and possible interactions with regard to EBV infection would facilitate the study design (30, 59). It is possible that other objectives (lowering the incidence of CVDs and cancers (58)) will lead to altered vitamin D recommendations in the general population, even before an MS primary preventive study has been launched. If this happens, great care has to be taken to study in parallel the incidence of diseases such as MS over time.
However, when it comes to relatives of MS cases, given the strength of indirect evidence, and the low risk of adverse events with daily doses of 2000-4000 IUs, it is probably rational to suggest supplementation with such daily doses, while awaiting more evidence . The same applies to the MS cases themselves, in whom these doses may be effective for preventing disease activity. The interest in vitamin D treatment both among patients, and in the media, has risen during the last years, and not all actors in this drama are well educated and serious. This is illustrated by a recent report of adverse effects in 21 Brazilian MS cases , 19 of whom had stopped taking their standard disease modifying treatments. They used median 100,000 IU vitamin D daily , reached serum concentrations of around 375 nmol/l, and presented with both signs of insufficient efficacy such as
MS relapses and disease progression, and
side effects such as renal failure, pseudotumor cerebri, and seizures (60).
Vitamin D treatment should by no means be used as a replacement for standard disease modifying drugs, nor should excessive doses be used. It is important that well- educated neurologists with expertise in MS also can give advice regarding vitamin D, so that the patients are not forced away from their original caregivers. As in all treatments, proper monitoring should be undertaken to detect disease activity, and follow-ups with laboratory testing (including serum 25(OH)D and calcium levels) are needed to detect adverse effects.
References
Limburg CC. The geographic distribution of multiple sclerosis and its estimated prevalence in the United States. Res Publ Assoc Res Nerv Ment Dis 1950;28: 15-24.
Gale CR, Martyn CN. Migrant studies in multiple sclerosis. Prog Neurobiol 1995;47:425-48.
Sloka S, Silva C, Pryse-Phillips W, Patten S, Metz L, Yong VW. A quantitative analysis of suspected environmental causes of MS. Can J Neurol Sci 2011;38:98-105.
Beretich BD, Beretich TM. Explaining multiple sclerosis prevalence by ultraviolet exposure: a geospatial analysis. Mult Scler 2009;15:891-8.
Orton SM, Wald L, Confavreux C et al. Association of UV radiation with multiple sclerosis prevalence and sex ratio in France. Neurology 2011;76:425-31.
Koch-Henriksen N, Sorensen PS. The changing demographic pattern of multiple sclerosis epidemiology. Lancet Neurol 2010;9:520-32.
Alcalde-Cabero E, Almazan-Isla J, Garcia-Merino A, de Sa J, De Pedro-Cuesta J. Incidence of multiple sclerosis among European Economic Area populations, 1985-2009: the framework for monitoring. BMC Neurol 2013;13:58.
Kingwell E, Marriott JJ, Jette N et al. Incidence and prevalence of multiple sclerosis in Europe: a systematic review. BMC Neurol 2013;13:128.
Diaz V, Barahona J, Antinao J et al. Incidence of multiple sclerosis in Chile. A hospital registry study. Acta Neurol Scand 2012;125:71-5.
Kampman MT, Wilsgaard T, Mellgren SI. Outdoor activities and diet in childhood and adolescence relate to MS risk above the Arctic Circle. J Neurol 2007;254: 471-7.
Spanier JA, Nashold FE, Olson JK, Hayes CE. The Ifng gene is essential for Vdr gene expression and vitamin D(3)-mediated reduction of the pathogenic T cell burden in the central nervous system in experimental autoimmune encephalomyelitis, a multiple sclerosis model. J Immunol 2012;189:3188-97.
Mowry EM, Waubant E, McCulloch CE et al. Vitamin D status predicts new brain magnetic resonance imaging activity in multiple sclerosis. Ann Neurol 2012;72:234-40.
Simpson S JR, Taylor B, Blizzard L et al. Higher 25- hydroxyvitamin D is associated with lower relapse risk in multiple sclerosis. Ann Neurol 2010;68:193-203.
Mowry EM, Krupp LB, Milazzo M et al. Vitamin D status is associated with relapse rate in pediatric-onset multiple sclerosis. Ann Neurol 2010;67:618-24.
Banwell B, Bar-Or A, Arnold DL et al. Clinical, environmental, and genetic determinants of multiple sclerosis in children with acute demyelination: a prospective national cohort study. Lancet Neurol 2011;10:436-45.
Ascherio A, Munger KL, White R et al. Vitamin D as an early predictor of multiple sclerosis activity and progression. JAMA Neurol 2014;71:306-14.
Pierrot-Deseilligny C, Rivaud-Pechoux S, Clerson P, de Paz R, Souberbielle JC. Relationship between 25- oH-D serum level and relapse rate in multiple sclerosis patients before and after vitamin D supplementation. Ther Adv Neurol Disord 2012;5:187-98.
Munger KL, Levin LI, Hollis BW, Howard NS, Ascherio A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 2006;296:2832-8.
Salzer J, Hallmans G, Nystrom M, Stenlund H, Wa- dell G, Sundstrom P. Vitamin D as a protective factor in multiple sclerosis. Neurology 2012;79:2140-5.
Ueda P, Rafatnia F, Baarnhielm M et al. Neonatal vitamin D status and risk of multiple sclerosis. Ann Neurol 2014;76:338-46.
Munger KL, Zhang SM, O’Reilly E et al. Vitamin D intake and incidence of multiple sclerosis. Neurology 2004;62:60-5.
Baarnhielm M, Olsson T, Alfredsson L. Fatty fish intake is associated with decreased occurrence of multiple sclerosis. Mult Scler 2014;20:726-32.
Pozuelo-Moyano B, Benito-Leon J, Mitchell AJ, Her- nandez-Gallego J. A systematic review of randomized, double-blind, placebo-controlled trials examining the clinical efficacy of vitamin D in multiple sclerosis. Neuroepidemiology 2012;40:147-53.
Salzer J, Bistrom M, Sundstrom P. Vitamin D and multiple sclerosis: where do we go from here? Expert Rev Neurother 2014;14:9-18.
Bergman P, Norlin AC, Hansen S et al. Vitamin D3 supplementation in patients with frequent respiratory tract infections: a randomised and double-blind intervention study. BMJ Open 2012;2:e001663.
Murdoch DR, Slow S, Chambers ST et al. Effect of vitamin D3 supplementation on upper respiratory tract infections in healthy adults: the VIDARIS randomized controlled trial. JAMA 2012;308:1333-9.
Jons D, Sundstrom P, Andersen O. Targeting Epstein- Barr virus infection as an intervention against multiple sclerosis. Acta Neurol Scand 2015;131:69-79.
Ramagopalan SV, Handel AE, Giovannoni G, Rutherford Siegel S, Ebers GC, Chaplin G. Relationship of uV exposure to prevalence of multiple sclerosis in England. Neurology 2011;76:1410-4.
Lossius A, Vartdal F, Holmoy T. Vitamin D sensitive EBNA-1 specific T cells in the cerebrospinal fluid of patients with multiple sclerosis. J Neuroimmunol 2011;240-241:87-96.
Salzer J, Nystrom M, Hallmans G, Stenlund H, Wa- dell G, Sundstrom P. Epstein-Barr virus antibodies and vitamin D in prospective multiple sclerosis biobank samples. Mult Scler 2013;19:1587-91.
Brot C, Jorgensen NR, Sorensen OH. The influence of smoking on vitamin D status and calcium metabolism. Eur J Clin Nutr 1999;53:920-6.
Vimaleswaran KS, Berry DJ, Lu C et al. Causal relationship between obesity and vitamin D status: bi-directional Mendelian randomization analysis of multiple cohorts. PLoS Med 2013;10:e1001383.
Hedstrom AK, Hillert J, Olsson T, Alfredsson L. Alcohol as a modifiable lifestyle factor affecting multiple sclerosis risk. JAMA neurology 2014;71:300-5.
Jaaskelainen T, Knekt P, Marniemi J et al. Vitamin D status is associated with sociodemographic factors, lifestyle and metabolic health. Eur J Nutr 2013;52:513- 25.
Hedstrom AK, Akerstedt T, Hillert J, Olsson T, Alfredsson L. Shift work at young age is associated with increased risk for multiple sclerosis. Ann Neurol 2011;70:733-41.
Bjornevik K, Riise T, Casetta I et al. Sun exposure and multiple sclerosis risk in Norway and Italy: the EnvIMS study. Mult Scler 2014;20:1042-9.
Munger KL, Chitnis T, Ascherio A. Body size and risk of MS in two cohorts of US women. Neurology 2009;73:1543-50.
Lin R, Charlesworth J, van der Mei I, Taylor BV. The genetics of multiple sclerosis. Pract Neurol 2012;12:279-88.
Kalman B, Toldy E. Genomic binding sites and biological effects of the vitamin D: VDR complex in multiple sclerosis. NeuroMol Med 2014;16:265-79.
Orton SM, Morris AP, Herrera BM et al. Evidence for genetic regulation of vitamin D status in twins with multiple sclerosis. Am J Clin Nutr 2008;88:441-7.
Ramagopalan SV, Dyment DA, Cader MZ et al. Rare variants in the CYP27B1 gene are associated with multiple sclerosis. Ann Neurol 2011;70:881-6.
Ban M, Caillier S, Mero IL et al. No evidence of association between mutant alleles of the CYP27B1 gene and multiple sclerosis. Ann Neurol 2013;73:430-2.
Sawcer S, Hellenthal G, Pirinen M et al. Genetic risk and a primary role for cell-mediated immune mechanisms in multiple sclerosis. Nature 2011;476:214-9.
Wang TJ, Zhang F, Richards JB et al. Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet 2010;376:180-8.
Tajouri L, Ovcaric M, Curtain R et al. Variation in the vitamin D receptor gene is associated with multiple sclerosis in an Australian population. J Neurogenet 2005;19:25-38.
Cox MB, Ban M, Bowden NA, Baker A, Scott RJ, Lechner-Scott J. Potential association of vitamin D receptor polymorphism Taq1 with multiple sclerosis. Mult Scler 2012;18:16-22.
Garcia-Martin E, Agundez JA, Martinez C et al. Vitamin D3 receptor (VDR) gene rs2228570 (Fok1) and rs731236 (Taq1) variants are not associated with the risk for multiple sclerosis: results of a new study and a metaanalysis. PLoS ONE 2013;8:e65487.
Ramagopalan SV, Heger A, Berlanga AJ et al. A ChiP-seq defined genome-wide map of vitamin D receptor binding: associations with disease and evolution. Genome Res 2010;20:1352-60.
Disanto G, Sandve GK, Berlanga-Taylor AJ et al. Vitamin D receptor binding, chromatin states and association with multiple sclerosis. Hum Mol Genet 2012;21:3575-86.
Holick MF. Vitamin D deficiency. N Engl J Med 2007;357:266-81.
Sintov AC, Yarmolinsky L, Dahan A, Ben-Shabat S. Pharmacological effects of vitamin D and its analogs: recent developments. Drug Discovery Today 2014;19:1769-74.
Hart PH, Gorman S, Finlay-Jones JJ. Modulation of the immune system by uV radiation: more than just the effects of vitamin D? Nat Rev immunol 2011;11:584-96.
Ramagopalan SV, Maugeri NJ, Handunnetthi L et al. Expression of the multiple sclerosis-associated MHC class II Allele HLA-DRB1*1501 is regulated by vitamin D. PLoS Genet 2009;5:e1000369.
Munger KL, Kochert K, Simon KC et al. Molecular mechanism underlying the impact of vitamin D on disease activity of MS. Ann Clin Transl Neurol 2014;1:605-17.
Schwarz A, Navid F, Sparwasser T, Clausen BE, Schwarz T. 1,25-dihydroxyvitamin D exerts similar immunosuppressive effects as uVR but is dispensable for local UVR-induced immunosuppression. J Invest Dermatol 2012;132:2762-9.
Breuer J, Schwab N, Schneider-Hohendorf T et al. ultraviolet B light attenuates the systemic immune response in central nervous system autoimmunity. Ann Neurol 2014;75:739-58.
Hypponen E, Laara E, Reunanen A, Jarvelin MR, Virtanen SM. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet 2001;358:1500-3.
Manson JE, Bassuk SS, Lee IM et al. The VITamin D and OmegA-3 TriaL (VITAL): rationale and design of a large randomized controlled trial of vitamin D and marine omega-3 fatty acid supplements for the primary prevention of cancer and cardiovascular disease. Contemp Clin Trials 2012;33:159-71.
Disanto G, Handel AE, Damoiseaux J et al. Vitamin D supplementation and antibodies against the Epstein-Barr virus in multiple sclerosis patients. Mult Scler 2013;19:1679-80.
Fragoso YD, Adoni T, Damasceno A et al. Unfavorable outcomes during treatment of multiple sclerosis with high doses of vitamin D. J Neurol Sci 2014;346:341-2.
- Note by VitaminDWiki Publisher wants $31.50 for the PDF
- That paper was commented on, and Publisher wants $31.50 for that as well
- Comment authors: Kimball S, Hanwell HE, Burton JM, Heaney RP, Holick MF, Hollis B, Lewanczuk R, Makhani N, Venkateswaran S.
