Is High Dose Vitamin D Harmful
Is High Dose Vitamin D Harmful?
Calcif Tissue Int Dec 2012 DOI 10.1007/s00223-012-9679-1
Kerrie M. Sanders • Geoffrey C. Nicholson • Peter R. Ebeling
K. M. Sanders (&) • G. C. Nicholson • P. R. Ebeling Department of Medicine, Western Health, NorthWest Academic Centre, University of Melbourne, PO Box 294, St Albans, VIC 3021, Australia e-mail: ksanders@unimelb.edu.au
G. C. Nicholson
Rural Clinical School, School of Medicine, University of Queensland, Toowoomba, QLD, Australia
Comment by VitaminDWiki
- This paper seems to consider that it is OK to have an annual amount of something in a single day
- Imagine the problem of taking 300 liters of water in a single day, or 300 grams of Vitamin C, etc. etc.
This paper ignores the documented problems of Vitamin D2, and treats D3 equal to D2
This paper ignores the need for cofactors - a huge imbalance in the body is a big problem
This paper ignores the need of Vitamin K2 to make sure that any excess Calcium does not cause problems
This paper ignores the problem of too much Calcium and not enough Magnesium when increasing vitamin D
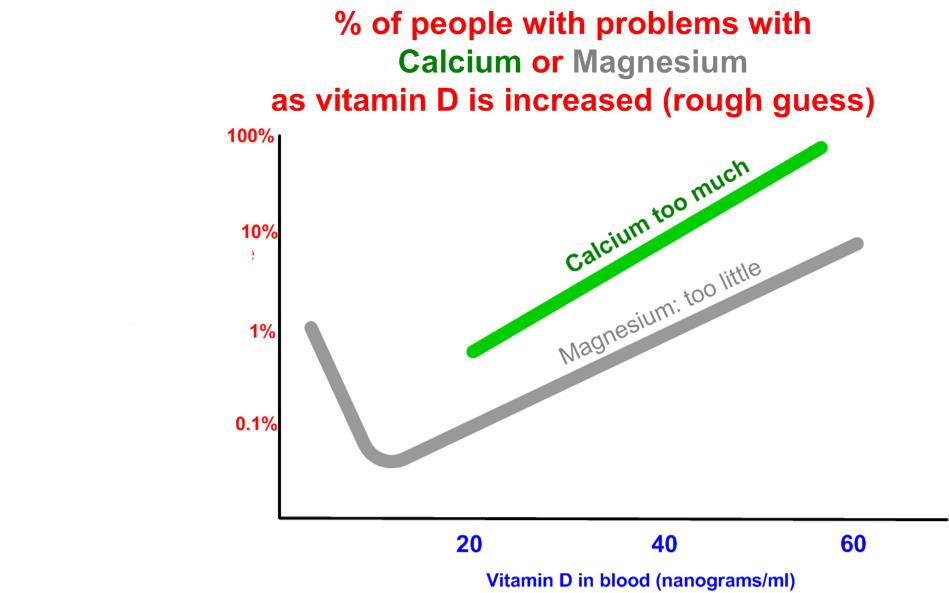
See also VitaminDWiki
Must balance co-factors when increasing vitamin D
- Calcium bioavailability and how much to take probably reduce to 500mg of Calcium
Overview Magnesium and vitamin D probably increase to 500 mg of Magnesium
Overview Vitamin K and Vitamin D 100 ug and perhaps 100 ug per 1000 IU of vitamin D
Overview Rare Allergic reaction to vitamin D about 1 in 300 people
Is 50 ng of vitamin D too high, just right, or not enough includes section on is > 40 ng too much?
Take vitamin D3 daily or weekly which has the following notional chart
PDF attached to the bottom of this page
Just a portion of each of the tables are shown below
This study was cited 97 times as of May 2021
Abstract
With the potential to minimize the risk of many chronic diseases and the apparent biochemical safety of ingesting doses of oral vitamin D several-fold higher than the current recommended intakes, recent research has focussed on supplementing individuals with intermittent, high-dose vitamin D. However, two recent randomized controlled trials (RCTs) both using annual high-dose vitamin D reported an increase, rather than a decrease, in the primary outcome of fractures. This review summarises the results from studies that have used intermittent, high doses of vitamin D, with particular attention to those finding evidence of adverse effects. Results from observational, population-based studies with evidence of a U- or J-shaped curve are also presented as these findings suggest an increased risk in those with the highest serum 25D levels. Speculative mechanisms are discussed and biochemical results from studies using high-dose vitamin D are also presented. Emerging evidence from both observational studies and RCTs suggests there should be a degree of caution about recommending high serum 25D concentrations for the entire population. Furthermore, benefit of the higher doses commonly used in clinical practice on falls risk reduction needs to be demonstrated. The safety of loading doses of vitamin D should be demonstrated before these regimens become recommended as routine clinical practice. The current dilemma of defining vitamin D insufficiency and identifying safe and efficacious repletion regimens needs to be resolved.
The need for vitamin D supplementation has evolved because it is widely recognized that a significant proportion of many populations has inadequate vitamin D status [1]. Serum 25-hydroxyvitamin D (25[OH]D) levels of 25 nmol/L or less are considered "deficient," while the definition of "insufficiency" varies, with some experts regarding less than 50 nmol/L as the cut-off [2] and others considering less than 75 nmol/L as "insufficient" [3-5]. "Insufficient" in this review refers to 25(OH)D levels in the range 25-50 nmol/L, "intermittent" dosing refers to at least 1-week dosing intervals, and high dose refers to an intermittent bolus dose of at least 20,000 IU or a daily dose of 4,000 IU. Although the risk of many chronic disorders may be reduced by an upward shift in the community's vitamin D status, daily dosing has proven to be problematic, particularly for older people, the group most likely to directly benefit from an improvement in vitamin D status [6]. Many randomized trials have reported poor compliance with daily regimens [7]. Furthermore, some people require substantial doses of vitamin D to achieve serum 25(OH)D levels within the target range [8, 9]. An intermittent, larger dose of vitamin D reduces this compliance issue in a simple and cost-effective manner and reduces the likelihood that the target group will remain below the threshold of 25(OH)D regarded as insufficient, although significant controversy exists regarding what level of serum 25(OH)D is sufficient [3, 10-12]. Both the public and practitioners want to make informed decisions regarding both the target level of 25(OH)D to optimize health and the appropriate dosing regimen to achieve this target level.
According to current evidence from biochemical, observational, and randomized controlled trials (RCTs), serum 25(OH)D levels of at least 50 nmol/L are required for normalization of PTH levels, to minimize the risk of osteomalacia, and for optimal bone and muscle function [2, 13, 14], with many experts regarding 75 nmol/L as the threshold for optimal bone health [12, 15-17]. The skeletal consequences of 25(OH)D insufficiency include secondary hyperparathyroidism, increased bone turnover and bone loss, and increased risk of low-trauma fractures. From a skeletal perspective, evidence from RCTs suggests that vitamin D may be considered a threshold nutrient with little bone benefit observed at levels of 25(OH)D above that at which parathyroid hormone (PTH) is normalized [2]. However, molecular studies have demonstrated that vitamin D plays a role in cell differentiation, function, and survival [18, 19]. Adequate calcium intake is imperative to gain optimal benefit from improving vitamin D status in those with insufficient 25(OH)D levels. The relative contributions of vitamin D and calcium to reducing fracture risk remain unclear [20], and improving calcium intake is also associated with suppression of PTH levels [21, 22]. Observational studies have shown a decreased risk of many disorders, including certain types of cancer, mental disorders, cardiovascular disease, and skin and autoimmune disorders, associated with serum 25(OH)D levels greater than 70-80 nmol/L [9, 12, 16]. It has therefore been argued that 25(OH)D levels should be in the range of 70-100 nmol/L to maximize these nonskeletal benefits.
With the potential to minimize the risk of many chronic diseases and the apparent safety of ingesting doses of oral vitamin D severalfold higher than the current recommended intakes, recent research has focused on supplementing individuals with intermittent, high-dose vitamin D. There is an urgent need to determine the efficacy and safety of these regimens. Using biochemical parameters of safety, particularly plasma and urine calcium, there are numerous studies reporting that a single oral dose of 300,000-600,000 IU of D2/D3 rapidly enhances serum 25(OH)D and reduces PTH in people with deficiency [23-25]. Although dosing intervals of greater than 2-3 months and/or intermittent bolus doses ([200,000 IU) are not regarded as physiological [26], such an approach offers a realistic and pragmatic public health measure to target at-risk populations and addresses the emerging public health issue of widespread vitamin D insufficiency [6, 27].
However, two recent RCTs, both using annual high-dose vitamin D, reported an increase, rather than a decrease, in the primary outcome of falls [27] and fractures [27, 28].
These findings highlight the need for a better understanding of different dosing regimens before pragmatic, population-based interventions are implemented. This review summarizes results from studies that have used intermittent, high doses of vitamin D, with particular attention to those finding evidence of adverse effects. Results from observational, population-based studies with evidence of a U-or /-shaped curve are also presented as these findings suggest an increased risk in those with the highest serum 25(OH)D levels. We do not attempt to present a comprehensive, systematic review of the extensive number of observational and intervention studies reporting the health benefits of improving vitamin D status in adults with insufficient status [7, 29-32] or to present a balanced view of the potential risk/benefit ratio of vitamin D supplementation.
Biochemical Outcomes of Single, Large Doses of Vitamin D
The immediate concern of hypervitaminosis D is hyper-calciuria and hypercalcemia [2]. However, a large therapeutic window exists for vitamin D-related hypercalcemia, which has not been reported at serum 25(OH)D levels below 220 nmol/L and generally not reported below 500 nmol/L [2]. Based on these biochemical parameters, Vieth and colleagues [17] conducted a 6-month safety and efficacy study and concluded that consumption of more than 4,000 IU/day causes no harm and effectively raises 25(OH)D levels to high-normal concentrations (<140 nmol/L) in practically all adults. The 2011 Institute of Medicine report on dietary intake of vitamin D recommended an upper limit of 4,000 IU/day, although it also stated that up to 10,000 IU/day is safe [33].
Of the studies included in this review, the cases of hypercalcemia and/or hypercalciuria are few and their incidence in the randomized trials is rarely different from that observed in the placebo group (Table 1). The study by Grimnes and colleagues [34], where one group was given 6,500 IU/day and another was given 400 IU/day, reported a significant difference between groups in serum ionized calcium at 12 months. In another trial, two of 33 patients receiving a single bolus dose of 300,000 IU vitamin D3 had mild hypercalcemia [35]. Participants in this study were older, recruited from a rheumatology clinic, and likely to have reduced kidney function compared with other trial participants who were, on average, younger (Table 1). However, some of the larger trials did not specifically investigate for hypercalcemia or hypercalciuria (Table 1).
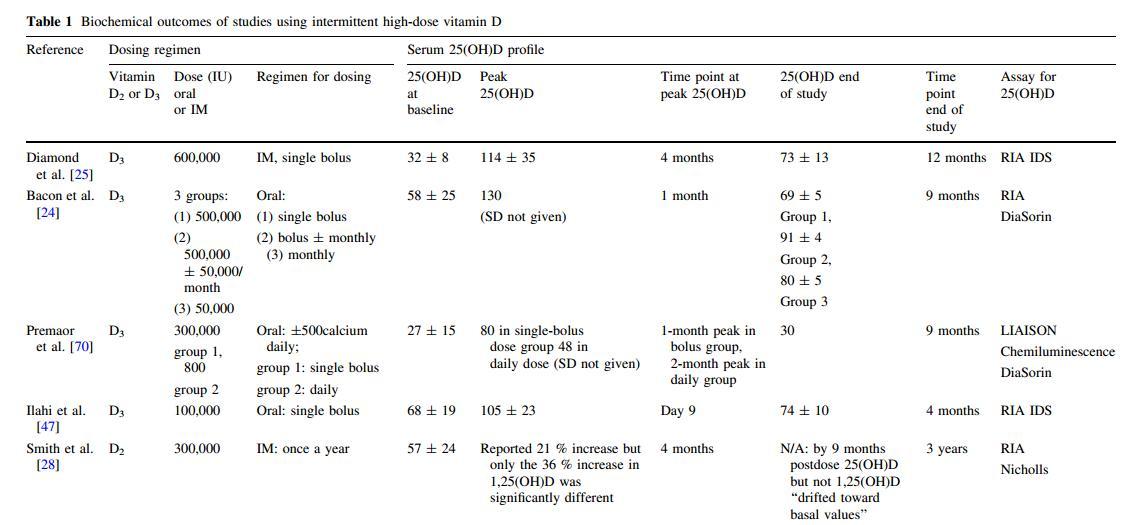
{This may be only a portion of the table which is in the PDF}
A 1995 review of the safety and effectiveness of different regimes of vitamin D supplementation in the elderly suggested that daily low-dose supplementation is the regimen of choice for prevention of hypovitaminosis D but that intermittent high-dose regimens would be a safe and be significant variation in the level of serum 25(OH)D effective alternative in patients with poor compliance [36]. reached when individuals are given the same dose and form As reported in individual trials [8, 9, 37], there appears to of the vitamin.
Table 2 Observational studies with evidence of harm at higher end of vitamin D status
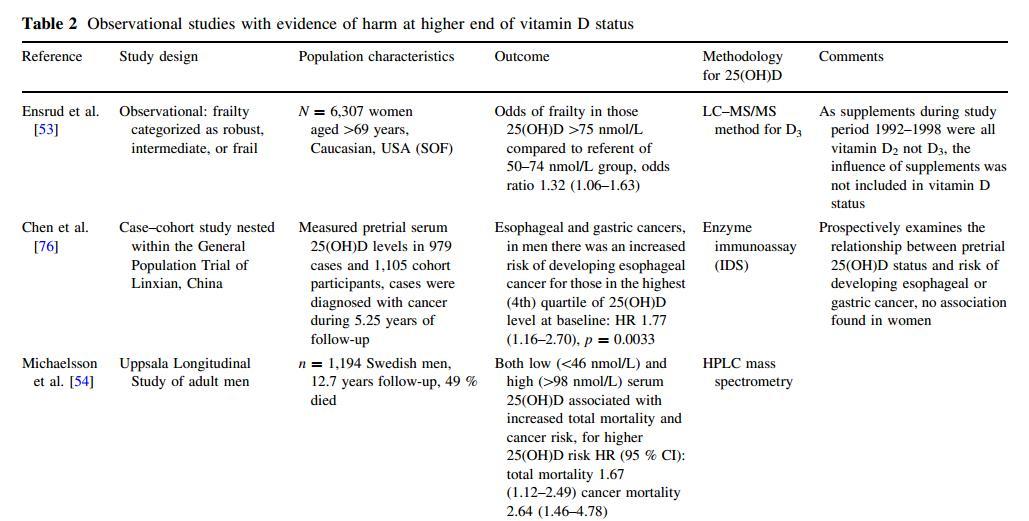
{This may be only a portion of the table which is in the PDF}
Vitamin D doses expressed in international units (IU). D2 vitamin D2 (ergocalciferol), D3 vitamin D3 (cholecalciferol), IM intramuscular injection, 25(OH)D serum 25-hydroxyvitamin D (nmol/L), 1,25(OH)D serum 1,25-dihydroxyvitamin D (levels quoted as mean and standard deviation unless stated otherwise), IQR interquartile range, N/A not available, RIA radioimmunoassay, HPLC high-pressure liquid chromatography, LC-MS/MS liquid chromatography coupled with tandem mass spectrometry, HR hazard ratio (95 % confidence interval), RR relative risk (95 % confidence interval), BTM bone turnover markers, CTX C-terminal-telopeptide (bone resorption marker), BAP bone-specific alkaline phosphatase, P1NP amino-terminal propeptide of type 1 procollagen (bone formation markers), F females, M males
There is a caveat that earlier studies were reliant on 25(OH)D assays that have shown considerable intra- and intersample variation in the assessment of serum 25(OH)D concentrations and were unreliable in measuring serum D2 levels [38]. Although the performance of radioimmunoassay and enzyme-linked assays is acceptable, the bias and imprecision of many automated methods may be problematic at the lower, clinically and analytically important range (<50 nmol/L) of the assay [2].
Evidence from Observational Studies
A majority of observational studies have reported that vitamin D is associated with a beneficial effect on risk of colon, breast, prostate, and ovarian cancers [39]. Since vitamin D synthesis and serum 25(OH)D levels are inversely correlated with latitude and positively correlated with sunlight, some studies have mapped disease incidence rates with latitude to investigate a possible protective effect of vitamin D status and risk of disease. Encouragingly, there is a consistency of findings between geographic studies and serum studies where samples of the population have had biochemistry assessments, ideally with the blood collection point several years prior to any diagnosis of cancer or other disease of interest [39]. Vitamin D and its metabolites are thought to reduce the incidence of many types of cancer by inhibiting tumor angiogenesis and hyperproliferation as well as stimulating cellular apoptosis [40]. Since vitamin D regulates a gamut of physiological processes, including immune modulation, resistance to oxidative stress, and modulation of other hormones, it is not surprising that low vitamin D has been associated with increased risk of several cancers and chronic diseases [41] as well as cancer mortality [42].
Nevertheless, there are now several observational studies reporting a U-orJ-shaped association between disease and serum 25(OH)D and latitude and/or ultraviolet B radiation levels, where those in the highest percentiles have an inverse risk compared with those in the lowest (Table 2)[43]. While cross-sectional data have many limitations, the findings are hypothesis-generating [44] and can be used to develop protocols for RCTs. The findings from prospective case-control cohort studies where blood collection occurred many years prior to diagnosis add another dimension to the evidence. The results from these studies generally support vitamin D supplementation in those with low vitamin D status. However, the findings argue for caution before increasing 25(OH)D levels and associated dosing regimens beyond evidence clearly supported by RCTs and meta-analyses
[45].
RCTs Demonstrating Harm
The evidence of harm relating to high-dose vitamin D centers on the findings of two RCTs that used annual high-dose vitamin D (Table 3), although results from RCTs using lower, more frequent dosing regimens have not been consistently clear. The different forms of the vitamin used in the studies and the different delivery modes demonstrate that the adverse outcomes are not restricted to one form of the vitamin. either study included calcium supplementation as part of the protocol. In the British Wessex study, 9,440 community-dwelling participants (4,354 men and 5,086 women) aged 75-100 years were randomly allocated to receive an annual injection of 300,000 IU vitamin D2 or matching placebo every autumn over 3 years [28]. In the entire cohort the risk of any first fracture was not different in the two treatment groups. However, the vitamin D group showed an increased risk of hip/femur fracture (hazard ratio [HR] = 1.49, 95 % confidence interval [CI] 1.02-2.18) and hip/femur/wrist fracture (HR = 1.40, 95 % CI 1.07-1.82). Analysis of the female subjects showed that vitamin D treatment was associated with a borderline increased risk of any nonvertebral fracture (HR = 1.21, 95 % CI 1.00-1.47) and increased risk of hip/femur (HR = 1.80, 95 % CI 1.12-2.90) and hip/femur/wrist fracture (HR = 1.59, 95 % CI 1.17-2.16). However, vitamin D treatment was not associated with increased risk of any fracture in males. o effect on falls was observed, although this was not a primary outcome and falls were ascertained by 6-monthly recall. The other study, of 2,256 community-dwelling Australian women aged 70-92 years randomly allocated to receive an annual oral dose of 500,000 IU vitamin D3, demonstrated a 15 % (95 % CI 1.02-1.30) increased rate of falls and a 26 % (95 % CI 1.00-1.59) increased rate of fractures [27]. A temporal pattern was observed, with the greatest increase occurring in the first 3 months after dosing (falls: p for homogeneity = 0.02). A temporal pattern of risk was not demonstrated in the Wessex study, although the 6-monthly ascertainment of fractures did not optimize this post hoc analysis (unpublished).
Serial biochemistry was performed only in a very small proportion of participants in both these RCTs (0.04 % and 6.1 % participants; Smith et al. [28] and Sanders et al. [27], respectively). either study recruited participants based on low 25(OH)D levels at screening. We are unable to infer that the adverse effects are confined to participants whose 25(OH)D levels were either deficient/insufficient or replete at baseline. It is well documented that the incremental increase in serum 25(OH)D is likely to be lower in those already replete prior to supplementation [24], and there is substantial variation in dose-response curves between individuals [8, 9]. There is therefore no evidence base to justify large annual loading doses of vitamin D to specific groups based on their baseline 25(OH)D level. Based on the reduction in fractures using 4-monthly dosing regimens in the Trivedi et al. [46] RCT and the biochemical results by Bacon et al. [24] and Ilahi et al. [47], it seems prudent to restrict intermittent higher doses to intervals not greater than 2-4 months. However, the reasoning is speculative, and RCT evidence with physical outcomes using a variety of dosing regimens is urgently needed. The fall characteristics from the Australian study do not suggest that the increased falls were attributable to one subgroup of participants experiencing the most falls. The proportion of participants falling multiple times did not vary between the vitamin D and placebo groups (unpublished data), and Kaplan-Meier plots of time to first fall show significant differences between the groups (p = 0.003). In another recent publication, a small group (n = 12) of older (mean age 73 years) subjects was treated with a single oral dose of 600,000 IU vitamin D3 [48]. Serum 25(OH)D increased from 54 ± 14 nmol/L at baseline to 168 ± 43 nmol/L at day 3 when the bone turnover markers C-terminal telopeptide (CTX) and N-terminal telopeptide (NTX) peaked at over 50 % above baseline. PTH decreased and 1,25-dihydroxyvitamin D (1,25[OH]D) increased by 25-50 % [48]. Rossini and colleagues [48] suggest that this transient increase in bone turnover markers may explain the negative clinical results obtained in studies using intermittent high-dose vitamin D. Sanders and colleagues [49] have also reported increased bone turnover among a sample of participants who underwent biochemistry assessments and had a very high incremental rise in serum 25(OH)D levels. While increased bone turnover may contribute to the demonstrated increase in fracture risk, this does not explain the clear evidence of increased falls in the Australian study
(Table 3).
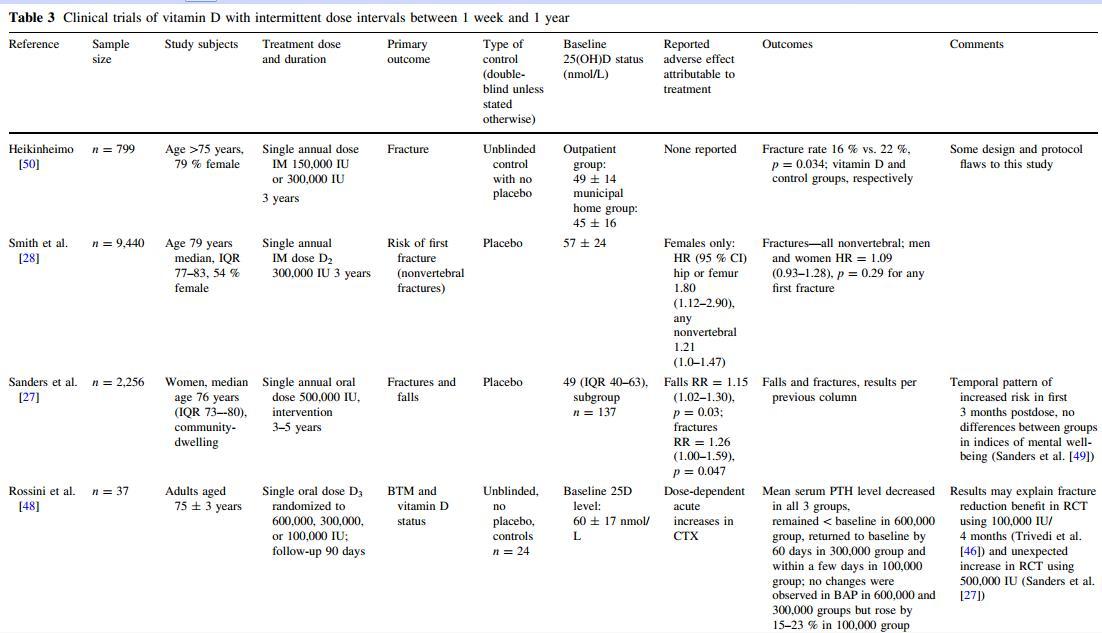
{This may be only a portion of the table which is in the PDF}
Speculative Mechanisms
The mechanism by which high-dose vitamin D might increase falls and fracture is uncertain. The opposing outcomes of two studies [28, 46] that used the same total annual dose (300,000 IU intramuscularly) suggest that the dosing regimen (i.e., 4-monthly vs. annually) rather than the total dose might determine the outcome. While a dosing interval of 12 months is equivalent to four biological half-lives of vitamin D, at the time these two studies were conducted there was biochemical evidence of safety and preliminary evidence that these intermittent bolus doses may be efficacious at decreasing fracture risk in older women [36, 46, 50]. In addition, the Australian study team was specifically addressing the drop in vitamin D and increased fractures that occurs during winter [51]. The line of reasoning regarding the dosing interval is supported by the temporal risk pattern observed in the study of Sanders et al. [27] and the fact that harm has not been reported in the numerous studies that have used more frequent dosing [52]. However, the lower-level evidence of a U-shaped dose-response curve reported in some observational studies [53-55] is not consistent with a temporal pattern since it is unlikely that those in the highest quintile of vitamin D status in the community use high intermittent doses of supplemental vitamin D. However, it is possible that seasonal fluctuations in 25(OH)D levels may contribute to this apparent phenomenon. Vieth [26] contends that a U-or J-shaped curve of risk is observed only in populations residing farther away from the equator and who, therefore, have greater seasonal fluctuations. It is argued that the annual downward phase in seasonal cycles almost definitely creates a non-steady-state situation for the paracrine production of 1,25(OH)D responsible for the noncalcemic effects of vitamin D. It is also possible the adverse mechanism may be associated with gender since Smith and colleagues [28] did not demonstrate an increased fracture risk among men and the majority of participants in the Trivedi et al. [46] study demonstrating a reduced fracture risk using 100,000 IU every 4 months were men (76 %). A Welsh RCT using the same dosing regimen as Trivedi et al. [46] but with 76 % women reported no difference in fracture outcome [56] (Tables 1, 3). In addition, the Australian RCT recruited only women [27]. There is also weak evidence from RCTs using more frequent dosing regimens that the mechanism is not a single aspect but may be more complex (Table 4).
Table 4
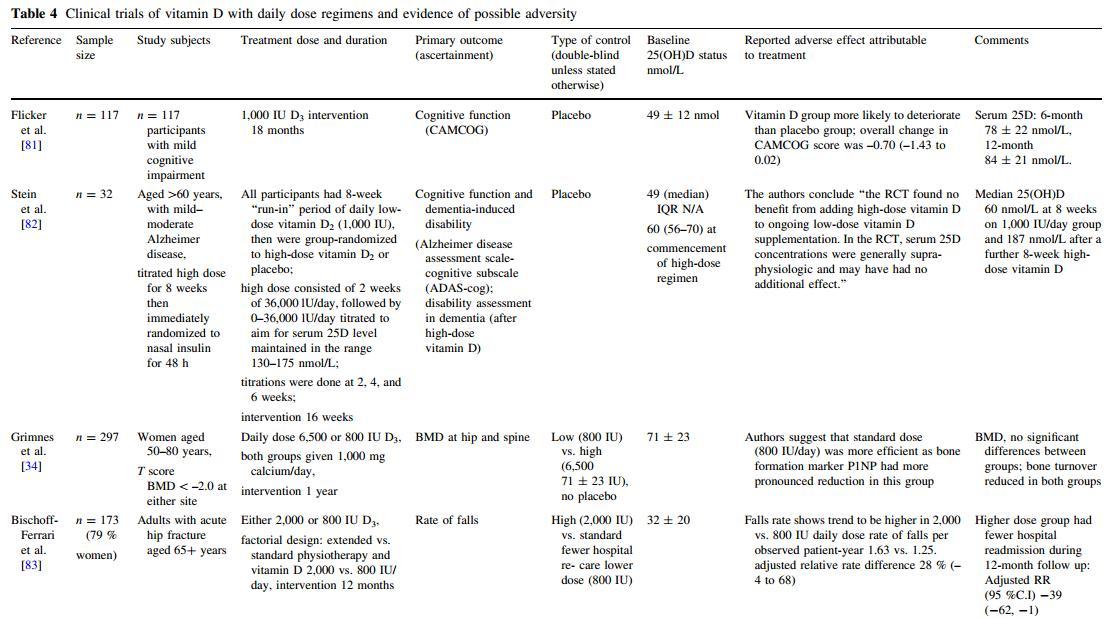
It has been hypothesized that the increased numbers of falls and fractures may have, ironically, resulted from the benefits of vitamin D in that the older women randomized to vitamin D felt better and consequentially engaged in more at-risk falls behavior [52]. However, the Australian authors subsequently published mental well-being outcomes of this study. No significant differences were detected in any of the measured outcomes of mental health [57], making this explanation less likely. No differences between the groups relating to the circumstances or activity of the fall events has been identified (unpublished).
In their editorial, Dawson-Hughes and Harris [52] also hypothesized that the 500,000 IU dose may have triggered a short-term protective reaction in which CYP24 (25-hydroxyvitamin D-24-hydroxylase), the enzyme that catabolizes 1,25(OH)D, was upregulated, resulting in decreased blood and tissue levels of 1,25(OH)D. Although this hypothesis is consistent with results from an animal study [58], both the Wessex [28] and Rossini et al. [48]
studies demonstrated increases (25-50 %) in serum 1,25(OH)D in those who had serial biochemistry assessments. From an evolutionary approach, Vieth [59] presents theargumentthatoralsupplementationofvitaminD is needed to improve health outcomes by lessening the destabilizing effect of annual fluctuations in serum 25(OH)D. He argues that the paracrine regulation of 1,25(OH)D in many tissues is disrupted by unstable 25(OH)D levels and that this adversely affects bone mineral density, mental well-being, infection, and cancer risk. The profile of 25(OH)D levels from the two studies showing harm does not appear distinctly different from a range of high-dose biochemical studies (Table 1). Although there is no uniformity in the time points of 25(OH)D assessment, peak 25(OH)D levels from these studies tend to be around 120-140 nmol/L. Ilahi and colleagues [47] suggest that the dosing interval of intermittent dosing regimens be not greater than 70 days to ensure that 25(OH)D levels do not decline below a target of 70 nmol/L.
The increased risk of falls in the Australian study demonstrates that the adverse mechanism is not confined to the skeleton. Post hoc analysis of changes in muscle strength in a nested substudy of these older women who underwent annual physical functioning assessments suggests a decline in muscle strength in those whose 25(OH)D level showed the greatest fluctuation from baseline [60]. Since there are vitamin D receptors in muscle, a sudden increase in vitamin D receptor occupancy could have an adverse effect on muscle function [61]. Vitamin D receptors are also present in the central nervous system [62], so an adverse effect on balance or coordination is also possible. Another recent Australian RCT of 686 ambulant women aged at least 70 years reported neither a beneficial nor an adverse effect on falls or physical function using a 3-monthly dosing of 150,000 IU cholecalciferol compared to placebo [63]. The study intervention period was 9 months, and the baseline 25(OH)D level measured in a subgroup of 40 participants was 66 ± 23 nmol/L. A review by Stockton and colleagues [64] concluded that vitamin D supplementation does not have a significant effect on muscle strength in vitamin D-replete adults.
Concluding Summary
While epidemiological studies provide evidence that vitamin deficiencies are associated with an increased risk of chronic disorders and/or cancer, the consequent philosophy that higher doses of the vitamin are protective and confer a reduced risk of these diseases is flawed [65, 66]. Two recent editorials on high-dose vitamin D have drawn analogies from the hard lessons learned from RCTs on high-dose vitamins A, B, C, and E [65, 66]. Supraphysio-logical levels of the vitamin taken as supplements do not emulate the apparent benefits of diets high in food that contain those vitamins and other lifestyle factors [67]. The findings from two recent high-dose RCTs [27, 28] identify a potential harm associated with high-dose vitamin D and support the notion that vitamin D could be now added to this list. Thus, in addition to evidence from enzyme kinetics relating to vitamin D metabolism [44], there is now high-level RCT evidence that vitamin D supplementation has potential toxicities other than simply hypercal-cemia/-uria. As our understanding of the pharmacokinetics of vitamin D metabolism becomes more sophisticated, clinical trials with novel dosing regimens should apply the principles of conventional pharmacology and vitamin D metabolism to the study design.
Interpretation of findings from many large RCTs has been limited by the lack of assessment of 25(OH)D status in the majority of participants. Future studies of supplementation should be adequately funded to allow comprehensive or universal measurement of serum 25(OH)D and related biochemical parameters [65, 68], with particular attention to large and rapid fluctuations in vitamin D status. Future studies should not base toxicity solely on the risk of hypercalcemia/-uria. There is an urgent need for dose-ranging studies with physical function outcomes [61, 64]. Emerging evidence from both observational studies and RCTs suggests that there should be a degree of caution about recommending high serum 25(OH)D concentrations for the entire population. Furthermore, a benefit of the higher doses commonly used in clinical practice on falls risk reduction needs to be demonstrated [69]. While it is recognized that intramuscular high-dose vitamin D preparations may be the only way of ensuring adequate vitamin D status in specific at-risk groups of patients, such as those suffering fat malabsorption, the safety of loading doses of vitamin D administered to the general population should be demonstrated before these regimens become recommended as routine clinical practice [65]. The current dilemma of defining vitamin D insufficiency and identifying safe and efficacious repletion regimens needs to be resolved.
References
1. Mithal A, Wahl D, Bonjour J, Burckhardt P, Dawson-Hughes B, Eisman J et al (2009) Global vitamin D status and determinants of hypovitaminosis D. Osteoporos Int 20(11):1807-1820
2. Nowson C, McGrath J, Ebeling P, Haikerwal A, Daly R, Sanders K et al (2012) Vitamin D and health in adults in Australia and New Zealand: a position statement. Med J Aust 196(11):686-687
3. Dawson-Hughes B, Mithal A, Bonjour JP, Boonen S, Burckhardt P, Fuleihan GE et al (2010) IOF position statement: vitamin D recommendations for older adults. Osteoporos Int 21(7):1151-1154
4. Henry HL, Bouillon R, Norman AW, Gallagher JC, Lips P, Heaney RP et al (2010) 14th Vitamin D Workshop consensus on vitamin D nutritional guidelines. J Steroid Biochem Mol Biol 121(1-2):4-6
5. Heaney RP, Holick MF (2011) Why the IOM recommendations for vitamin D are deficient. J Bone Miner Res 26(3):455-457
6. Ebeling PR (2005) Megadose therapy for vitamin D deficiency. Med J Aust 183(1):4-5
7. Tang B, Eslick G, Nowson C, Smith C, Bensoussan A (2007) Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet 370(9588):657-1656
8. Giusti A, Barone A, Pioli G, Girasole G, Razzano M, Pizzonia M et al (2010) Heterogeneity in serum 25-hydroxyvitamin D response to cholecalciferol in elderly women with secondary hyperparathyroidism and vitamin D deficiency. J Am Geriatr Soc 58:1489-1495
9. Leidig-Bruckner G, Roth H, Bruckner T, Lorenz A, Raue F, Frank-Raue K (2011) Are commonly recommended dosages for vitamin D supplementation too low? Vitamin D status and effects of supplementation on serum 25-hydroxyvitamin D levels—an observational study during clinical practice conditions. Osteo-poros Int 22:231-240
10. Chung M, Balk EM, Brendel M, Ip S, Lau J, Lee J et al (2009) Vitamin D and calcium: a systematic review of health outcomes. Evid Rep Technol Assess (Full Rep) 183:1-420
11. Hansen KE (2011) High-dose vitamin D: helpful or harmful? Curr Rheumatol Rep 13(3):257-264
12. Holick M (2007) Medical progress: vitamin D deficiency. N Engl J Med 357:266-281
13. Committee to Review Dietary Reference Intakes for Calcium and Vitamin D, Food and Nutrition Board (2010) Dietary reference intakes for calcium and vitamin D. Institute of Medicine, Washington, DC
14. Lips P, Bouillon R, van Schoor NM, Vanderschueren D, Ver-schueren S, Kuchuk N et al (2010) Reducing fracture risk with calcium and vitamin D. Clin Endocrinol (Oxf) 73(3):277-285
15. Dawson-Hughes B, Heaney RP, Holick MF, Lips P, Meunier PJ, Vieth R (2005) Estimates of optimal vitamin D status. Osteoporos Int 16(7):713-716
16. Heaney R (2008) Vitamin D: criteria for safety and efficacy. Nutr Rev 66(Suppl 2):s178-s181
17. Vieth R, Chan P, MacFarlane G (2001) Efficacy and safety of vitamin D3 input exceeding the lowest observed adverse effect concentration. Am J Clin Nutr 73:288-294
18. Montero-Odasso M, Duque G (2005) Vitamin D in the aging musculoskeletal system: an authentic strength preserving hormone. Mol Aspects Med 26:203-219
19. Anderson P, May B, Morris H (2003) Vitamin D metabolism: new concepts and clinical implications. Clin Biochem Rev 24: 13-26
20. Group DIPART (2010) Patient level pooled analysis of 68,500 patients from seven major vitamin D fracture trials in US and Europe. BMJ 340:b5463
21. Elders P, Lips P, Netelenbos J, Van Grinkel F, Khoe E, Van der Vijgh W et al (1994) Long-term effect of calcium supplementation on bone loss in perimenopausal women. J Bone Miner Res 9:963-970
22. Reid I, Mason B, Horne A, Ames R, Reid H, Bava U et al (2006) Randomized controlled trial of calcium in healthy older women. Am J Med 119(9):777-785
23. Cipriani C, Romagnoli E, Scillitani A, Chiodini I, Clerio R, Carnevale V et al (2010) Effect of a single oral dose of 600,000 IU of cholecalciferol on serum calciotropic hormones in young people with vitamin D deficiency: a prospective intervention study. J Clin Endocrinol Metab 95:4771-4777
24. Bacon C, Gamble G, Horne A, Scott M, Reid I (2009) High-dose vitamin D3 supplementation in the elderly. Osteoporos Int 20(8): 1407-1415
25. Diamond T, Ho K, Rohl P, Meerkin M (2005) Annual intramuscular injection of a megadose of cholecalciferol for treatment of vitamin D deficiency: efficacy and safety data. Med J Aust 183(1):10-12
26. Vieth R (2011) The pharmacology of vitamin D. In: Feldman D, Pike J, Adams J (eds) vitamin D. Academic Press, London, pp 1041-1066
27. Sanders K, Stuart A, Williamson E, Simpson J, Kotowicz M, Young D et al (2010) Annual high-dose oral vitamin D for falls and fractures in elderly women: a randomised, double-blind, placebo-controlled study. JAMA 303(18):1815-1822
28. Smith H, Anderson F, Raphael H, Maslin P, Crozier S, Cooper C (2007) Effect of annual intramuscular vitamin D on fracture risk in elderly men and women—a population-based, randomized, double-blind, placebo-controlled trial. Rheumatology 46(12): 1852-1857
29. Bischoff-Ferrari H, Willett W, Wong J, Giovannucci E, Dietrich T, Dawson-Hughes B (2005) Fracture prevention with vitamin D supplementation: a meta-analysis of randomized controlled trials. JAMA 293(18):2257-2264
30. Bischoff-Ferrari H, Willett W, Wong J, Stuck A, Staehelin H, Orav J et al (2009) Prevention of nonvertebral fractures with oral vitamin D and dose dependency: a meta-analysis of randomized controlled trials. Arch Intern Med 169(6):551-561
31. Reid I, Bolland M, Grey A (2008) Effect of calcium supplementation on hip fractures. Osteoporos Int 19(8):1119-1123
32. Cranney A, Weiler H, O'Donnell S, Puil L (2008) Summary of evidence-based review on vitamin D efficacy and safety in relation to bone health. Am J Clin Nutr 88(2):513S-519S
33. Ross AC, Manson JE, Abrams SA, Aloia JF, Brannon PM, Clinton SK et al (2011) The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab 96(1): 53-58
34. Grimnes G, Joakimsen R, Figenschau Y, Torjesen PA, Almas B, Jorde R (2012) The effect of high-dose vitamin D on bone mineral density and bone turnover markers in postmenopausal women with low bone mass—a randomized controlled 1-year trial. Osteoporos Int 23(1):201-211
35. von Restorff C, Bischoff-Ferrari H, Theiler R (2009) High-dose oral vitamin D3 supplementation in rheumatology patients with severe vitamin D3 deficiency. Bone 45:747-749
36. Byrne P, Freaney R, McKenna M (1995) Vitamin D supplementation in the elderly: review of safety and effectiveness of different regimes. Calcif Tissue Int 56:518-520
37. Heaney R, Davies K, Chen T, Holick M, Barger-Lux M (2003) Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am J Clin Nutr 77:204-210
38. Binkley N, Gemar D, Engelke J, Gangnon R, Ramamurthy R, Krueger D et al (2011) Evaluation of ergocalciferol or chole-calciferol dosing, 1,600 IU daily or 50,000 IU monthly in older adults. J Clin Endocrinol Metab 96(4):981-988
39. Garland C, Garland F, Gorham E, Lipkin M, Newmark H, MohrS et al (2006) The role of vitamin D in cancer prevention. Am J Public Health 96(2):252-261
40. Lin R, White J (2004) The pleiotropic actions of vitamin D. BioEssays 26(1):21-28
41. Evatt M (2010) Beyond vitamin status: is there a role for vitamin D in Parkinson disease? Arch Neurol 67(7):795-796
42. Grant W (2002) An estimate of premature cancer mortality in the U.S. due to inadequate doses of solar ultraviolet-B radiation. Cancer 94(6):1867-1875
43. Freedman DM, Looker AC, Abnet CC, Linet MS, Graubard BI (2010) Serum 25-hydroxyvitamin D and cancer mortality in the NHANES III study (1988-2006). Cancer Res 70(21):8587-8597
44. Vieth R (2004) Enzyme kinetics hypothesis to explain the U-shaped risk curve for prostate cancer vs. 25-hydroxyvitamin D in Nordic countries. Int J Cancer 111(3):468
45. Grant WB (2004) Geographic variation of prostate cancer mortality rates in the United States: implications for prostate cancer risk related to vitamin D. Int J Cancer 111(3):470-472
46. Trivedi D, Doll R, Khaw K (2003) Effect of four monthly oral vitamin D supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. BMJ 326(7387):469-475
47. Ilahi M, Armas L, Heaney R (2008) Pharmokinetics of a single, large dose of cholecalciferol. Am J Clin Nutr 87:688-691
48. Rossini M, Adami S, Viapiana O, Fracassi E, Idolazzi L, Povino M et al (2012) Dose-dependent short-term effects of single high doses of oral vitamin D3 on bone turnover markers. Calcif Tissue Int 91:365-369
49. Sanders K, Ebeling P, Stuart A, Williamson E, Kotowicz M, Nicholson G (2011) Annual high-dose oral vitamin D3:is increased risk of fractures attributable to changes in bone turnover markers? In: ANZBMS/IOF JSBR annual scientific meeting, Gold Coast, Australia
50. Heikinheimo R (1992) Annual injection of vitamin D and fractures of aged bones. Calcif Tissue Int 51(2):105-110
51. Pasco J, Henry M, Kotowicz M, Sanders K, Seeman E, Pasco J et al (2004) Seasonal periodicity of serum vitamin D and parathyroid hormone, bone resorption, and fractures: the Geelong Osteoporosis Study. J Bone Miner Res 19(5):752-758
52. Dawson-Hughes B, Harris S (2010) High-dose vitamin D supplementation: too much of a good thing? JAMA 303(18):1861-1862
53. Ensrud K, Ewing S, Fredman L, Hochberg M, Cauley J, Hillier T et al (2010) Circulating 25-hydroxyvitamin D levels and frailty status in older women. J Clin Endocrinol Metab 95:5266-5273
54. Michaelsson K, Baron J, Snellman G, Gedeborg R, Bysberg L, Sundstrom J et al (2010) Plasma vitamin D and mortality in older men: a community-based prospective cohort study. Am J Clin Nutr 92(4):841-848
55. McGrath J, Eyles D, Pedersen C, Ko P, Burne T et al (2010) Neonatal vitamin D status and risk of schizophrenia. Arch Gen Psychiatry 67(9):889-894
56. Lyons R, Johansen A, Brophy S, Newcombe R, Phillips C, Lervy B et al (2007) Preventing fractures among older people living in institutional care: a pragmatic randomised double blind placebo controlled trial of vitamin D supplementation. Osteoporos Int 18(6):811-818
57. Sanders K, Stuart A, Williamson E, Jacka F, Dodd S, Nicholson G et al (2011) A randomised controlled trial investigating an annual high dose vitamin D3 and mental well-being: vital D study. Br J Psychiatry 198:357-364
58. Beckman MJ, Johnson JA, Goff JP, Reinhardt TA, Beitz DC, Horst RL (1995) The role of dietary calcium in the physiology of vitamin D toxicity: excess dietary vitamin D3 blunts parathyroid hormone induction of kidney 1-hydroxylase. Arch Biochem Biophys 319(2):535-539
59. Vieth R (2009) Proposal A: optimal health requires high 25OHD levels >40 ng/ml (>100 nmol/L). Fourteenth Workshop on Vitamin D. Brugge, Belgium, p 72
60. Sanders K, Ebeling P, Stuart A, Williamson E, Simpson J, Kotowicz M, et al. (2011) The efficacy of high-dose vitamin D3 administered once a year: is the increased risk of falls attributable to changes in muscle strength? American Society of Bone and Mineral Research scientific meeting. San Diego, CA, September 16-20
61. Reid IR (2010) Vitamin D—let's get back to the evidence base. IBMS Bonekey 7(7):249-253
62. Buell JS, Dawson-Hughes B (2008) Vitamin D and neurocogni-tive dysfunction: preventing "D"ecline? Mol Aspects Med 29(6):415-422
63. Glendenning P, Zhu K, Inderjeeth C, Howat P, Lewis JR, Prince RL (2012) Effects of three-monthly oral 150,000 IU cholecalciferol supplementation on falls, mobility, and muscle strength in older postmenopausal women: a randomized controlled trial. J Bone Miner Res 27(1):170-176
64. Stockton KA, Mengersen K, Paratz JD, Kandiah D, Bennell KL (2011) Effect of vitamin D supplementation on muscle strength: a systematic review and meta-analysis. Osteoporos Int 22(3):859-871
65. Cummings S (2010) A single, annual, high dose of oral vitamin D increased falls and fractures in older women. ACP J Club 153(3): JC3-3
66. Byers T (2010) Anticancer vitamins du jour—the ABCED's so far. Am J Epidemiol 172(1):1-3
67. World Cancer Research Fund, American Institute for Cancer Research (2007) Food, nutrition, physical activity, and the prevention of cancer: a global perspective. AICR, Washington, DC
68. Francis R (2007) The vitamin D paradox. Rheumatology 46: 1749-1750
69. Bouillon R, Norman AW, Lips P (2007) Vitamin D deficiency. N Engl J Med 357(19):1980-1981
70. Premaor MO, Scalco R, da Silva MJ, Froehlich PE, Furlanetto TW (2008) The effect of a single dose versus a daily dose of cholecalciferol on the serum 25-hydroxycholecalciferol and parathyroid hormone levels in the elderly with secondary hyperparathyroidism living in a low-income housing unit. J Bone Miner Metab 26(6):603-608
71. Pekkarinen T, Valimaki VV, Aarum S, Turpeinen U, Hamalainen E, Loyttyniemi E et al (2010) The same annual dose of 292000 IU of vitamin D (cholecalciferol) on either daily or four monthly basis for elderly women: 1-year comparative study of the effects on serum 25(OH)D concentrations and renal function. Clin Endocrinol (Oxf) 72(4):455-461
72. Jorde R, Sneve M, Figenschau Y, Svartberg J, Waterloo K (2008) Effects of vitamin D supplementation on symptoms of depression in overweight and obese subjects: randomized double blind trial. J Intern Med 264:599-609
73. Vieth R, Kimball S, Hu A, Walfish P (2004) Randomized comparison of the effects of the vitamin D3 adequate intake versus 100 mcg (4000 IU) per day on biochemical responses and the wellbeing of patients. Nutr J 3:8
74. Law M, Withers H, Morris J, Anderson F (2006) Vitamin D supplementation and the prevention of fractures and falls: results of a randomised trial in elderly people in residential accommodation. Age Ageing 35:482-486
75. Aloia J, Bojadzievski T, Yusupov E, Shadzad G, Pollack S, Mikhail M et al (2010) The relative influence of calcium intake and vitamin D status on serum parathyroid hormone and bone turnover biomarkers in a double-blind, placebo-controlled parallel group, longitudinal factorial desigin. J Clin Endocrinol Metab 95(7):3216-3224
76. Chen W, Dawsey SM, Qiao YL, Mark SD, Dong ZW, Taylor PR et al (2007) Prospective study of serum 25(OH)-vitamin D concentration and risk of oesophageal and gastric cancers. Br J Cancer 97(1):123-128
77. Ahn J, Peters U, Albanes D, Purdue MP, Abnet CC, Chatterjee N et al (2008) Serum vitamin D concentration and prostate cancer risk: a nested case-control study. J Natl Cancer Inst 100(11): 796-804
78. Tuohimaa P, Tenkanen L, Ahonen M, Lumme S, Jellum E, Hallmans G et al (2004) Both high and low levels of blood vitamin D are associated with a higher prostate cancer risk: a longitudinal, nested case-control study in the Nordic countries. Int J Cancer 108(1):104-108
79. Stolzenberg-Solomon R, Vieth R, Azad A, Pietinen P, Taylor P, Virtamo J et al (2006) A prospective nested case-control study of vitamin D status and pancreatic cancer risk in male smokers. Cancer Res 66(20):10213-10219
80. Stolzenberg-Solomon RZ, Hayes RB, Horst RL, Anderson KE, Hollis BW, Silverman DT (2009) Serum vitamin D and risk of pancreatic cancer in the prostate, lung, colorectal, and ovarian screening trial. Cancer Res 69(4):1439-1447
81. Flicker L, Mead K, MacInnis R, Nowson C, Scherer S, Stein M et al (2003) Serum vitamin D and falls in older women in residential care in Australia. J Am Geriatr Soc 51(11):1533-1538
82. Stein MS, Scherer SC, Ladd KS, Harrison LC (2011) A randomized controlled trial of high-dose vitamin D2 followed by intranasal insulin in Alzheimer's disease. J Alzheimers Dis 26(3): 477-484
83. Bischoff-Ferrari HA, Dawson-Hughes B, Platz A, Orav EJ, Stahelin HB, Willett WC et al (2010) Effect of high-dosage cholecalciferol and extended physiotherapy on complications after hip fracture: a randomized controlled trial. Arch Intern Med 170(9):813-820
short url = http://is.gd/highdose
