Boosting Immunity with Vitamin D to reduce COVID-19 risks
Boosting immunity with vitamin D for preventing complications and deaths from COVID-19. COVID 19: Impact, Mitigation, Opportunities and Building Resilience: From Adversity to Serendipity.
Perspectives of global relevance based on research, experience and successes in combating COVID-19 in Sri Lanka
National Science Foundation, Sri Lanka. pp. 171-198, Vol. I, 2021 ISBN 978-624-5896-00-4 © 2021
Wimalawansa , S.J.Professor of Medicine, Endocrinology & Nutrition, Cardiometabolic and Endocrine Institute, North Brunswick, N.J. 08902, USA suniljw@hotmail.com
Polonowita, A.: Mednet Australia, Melbourne, Australia
Apparently initially presented Jan 2021
Note: Serendib, also spelled Serendip, Arabic Sarandīb, name for the island of Sri Lanka (Ceylon) Britanica
📄 Download the PDF from VitaminDWiki
ABSTRACT
The severity and deaths from COVID-19 differed between countries. Northern and southern countries were most affected by the first and the second wave of COVID-19. In contrast, the third wave affects geographically restricted regions: certain south-east Asian countries, Brazil, etc. China, Singapore, South Korea, Vietnam, and Hong Kong, countries that previously experienced SARS, met the challenges effectively with better public cooperation and coordinated public health actions. There was a lack of firm policies in the west, ambiguous and politically motivated guidance from health organisations such as WHO and CDC and some governments that eroded trust. Due to the year-long sunlight, countries proximate to the equator reported low prevalence and death rates from COVID-19. Low reported deaths were unrelated to curfew but due to a combination of reasonable vitamin D levels secondary to passive exposure to sunlight, greater use of ivermectin and hydroxychloroquine, and conducting very low (5% of the needed) number of PCT testing. These led to detecting a few PCR positive cases and related deaths. We examined the published data on the impact of vitamin D on immune functions, differences in reported deaths, and cost-effective ways to prevent complications and deaths from COVID-19. Data suggests that sufficient vitamin D is the most crucial factor protecting against infection, reducing complications and deaths from COVID-19. Despite the rhetoric, considering current inequitable immunisations, relatively shortlived immunity, and high mutation rates, global herd immunity is unlikely to be achieved. Based on recent clinical studies, including RCTs, the combination of population-wide supplementation of vitamin D and safe sun exposure, wearing facemasks, and vaccination is the most effective way to keep COVID under control. Boosting and maintaining innate immunity with nutrients (e.g., vitamin D sufficiency) and acquire immunity through vaccination will facilitate having overall, strong immunity. This combined approach would be the most effective and economical way to control the pandemic. Data are pointing that just antibody-based adaptive immune responses primed by prior infection or vaccination are insufficient to prevent SARS.CoV-2 outbreaks. In the absence of such priming, the robust innate and adaptive immune systems ensure rapid recovery and a low risk of complications. This is most effective when the serum 25-hydroxyvitamin D [25(OH) D] concentration maintained over 50 ng/mL optimal immune systems function.
Figures and Table
F2
F3
F4
F5
F6
F7
F8
F9
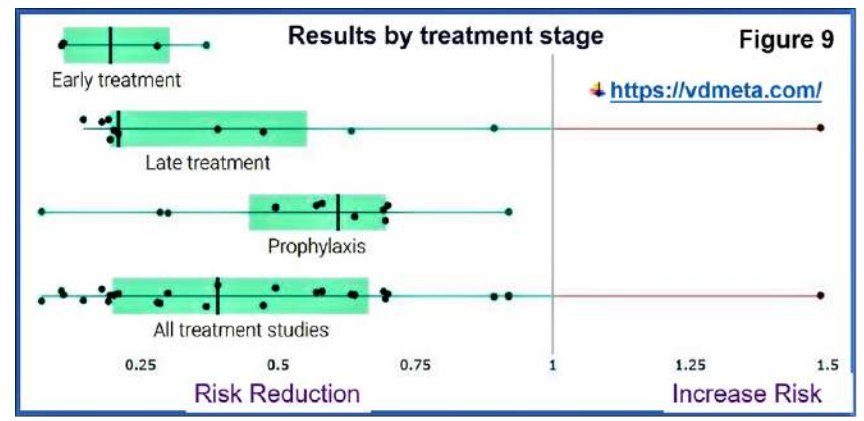
Table 1
F10
INTRODUCTION
There is a robust negative relationship between serum 25(OH)D (25-hydroxyvitamin D) concentration and clinical outcome from COVID-19 infections. In addition, clinical studies confirmed that the majority who died from COVID-19 had serum 25(OH)D concentration less than 12 ng/mL.
How much is vitamin D necessary for robust immune responses?
Scientists previously believed that the optimal immune stimulation occurs when the serum 25-hydroxyvitamin D [25(OH) D] concentration above 30 ng/mL. On the contrary, recent data confirmed that 25(OH)D concentrations higher than 50 ng/mL needed for full immune functions and protection from pathogenic microbes (Quraishi et al., 2014). This 50 ng/mL or more (at least above 40 ng/ mL) serum concentration can reliably attain daily or weekly vitamin D3 supplementation (but not more infrequently than once a month).
Most people are not getting adequate UVB rays from sunlight, either due to sun avoidance behaviour or lack of sufficient sunlight. Therefore, supplementation with the proper doses or food fortification with natural vitamin D is the way forward. These include exclusively breastfed infants, pregnant mothers, and those who are breastfeeding. However, because of too conservative doses used food fortification on its own, unable to provide adequate vitamin D supplements to achieve required levels.
Vitamin D supplementation is essential for those who live in higher and lower latitudes (42nd parallel). A few UV-B rays are reaching from sunlight for approximately six months- winter periods. Therefore, it is safer for them to consuming about 5,000 IU per day or 10,000 IU every other day without any adverse effect. However, these doses take several months to provide the required 25(OH)D blood levels. Alternatively, one can take or 50,000 IU once or twice a week, which will raise the blood levels within six weeks.
Safe sun exposure: For people with light coloured skin, one-hour daily exposure to summerlike sunlight between 10.30 AM and 1.30 PM with one-third of the skin exposed. Wearing a brimmed hat and sunglasses are strongly recommend for protecting the face and eyes. Those with melanin-rich skin require prolonged UV-B exposure daily, generally not compatible with modern lifestyles for many.
Especially during the pandemic era, the recommended daily dose is between 4,000 and 7,000 IU (average of 5,000 IU =0.125 mg) per day or 50,000 once a week for 70 kg, non-obese adult. Those who are overweight or obese require a higher intake: increase the ratio of D3 to bodyweight. It is important to note that D3 supplemented using these doses takes few months to build serum 25(OH) D levels. In clinical emergencies, due to immune dysfunctions, such as COVID-19, sepsis, Kawasaki disease, and Multisystem Inflammatory Syndrome, the use of such doses is not practical and ineffective.
In part, this can be overcome by upfront bolus doses of D3, but still, it takes one to three days to convert vitamin D to 25(OH)D in the liver (a rate-limiting step). Therefore, the administration of D3 alone is insufficient in emergencies. However, recent data confirmed that for 70 kg adults, a single oral dose of partially activated vitamin D, calcifediol (25-hydroxyvitamin D), is adequate to raise serum 25(OH)D to therapeutic level within four hours. This should be followed up with 200,000 IU vitamin D3 (single or in divided doses over four days), provides the desired blood levels within four hours and maintain it for several weeks (Castillo, 2020).
Mentioned single-dose quantity is minimal: between 0.5 to 1.0 mg calcifediol, administered at the earliest opportunity. It rapidly boosts all immune functions and allows people to maintain serum 25(OH) D concentration. This allows to overcome microbial invasions and prevent severe complications from COVID-19. Recent clinical studies using calcifediol confirmed that this protocol reduces the complications and deaths from COVID by ~75% (see clinical study section) (Castillo, 2020; Nogués, 2021).
Significant causes of vitamin D deficiency
The three most common causes of vitamin D deficiency are unavailability of UVB rays (e.g., winter months, air pollution), sun avoiding behaviour, and ageing-associated reduction of vitamin D synthesis in the skin. Typically, hypovitaminosis D occurs following insufficient exposure to sunlight. Examples include living in higher or lower latitudes or regions with significant air pollution, sun avoidance behaviour (Conticini et al., 2020; Ebadi & Montano-Loza, 2020; Grant et al., 2020; Martelletti & Martelletti, 2020; Zhu et al., 2020), persons with darker skin living in cooler environments, and gastrointestinal absorption difficulties. In addition, taking medications that up-regulate hepatic cytochrome P450 increases the catabolism of vitamin D: examples are antiepileptic and anti-retroviral therapies.
Diets contain only small amounts of vitamin D. Humans are dependent on exposure to sunlight (UV-B rays) or vitamin D supplements to obtain sufficient vitamin D. Vitamin D undergoes its first 25-hydroxylation in the liver, forming calcifediol (calcidiol):
[25-hydroxyvitamin D; 25(OH)D]. Hepatic 25-hydroxylation catalysed by enzyme CYP2R1 enzymes (2-4). The steps of activating vitamin D illustrated in Figure 1.
25(OH)D is the main form of vitamin D in circulation and storage. It has a halflife of approximately three weeks at healthy levels, such as 50 ng/mL. Vitamin D blood tests measure the concentration of 25(OH)D circulating in the bloodstream.
25(OH)D is the proper measure of vitamin D sufficiency. Blood 1,25(OH)2D concentration should not be measured to assess the vitamin D status. . There is no known function of 25(OH)D, apart from being the precursor for forming the hormonal and intracellular form of calcitriol. Circulating 25(OH)D concentration determines the amount and how this precursor enters into targets cells to produce intracellular calcitriol, which is crucial for immune functions.
Formation of active 1,25(OH)2D (calcitriol)
Figure 1.
The best-known system is the kidneys, converting 25(OH)D into feedback-controlled, low concentrations of circulating calcitriol that diffuses back into the circulation: after here called 1,25(OH)2D. This circulating 1,25(OH)2D acts as a hormone (an endocrine signalling agent). The circulatory calcitriol (the hormonal form) concentration is control by the kidneys. Its primary functions are intestinal and renal tubular calcium absorption: together with parathyroid hormone, it modulates bone metabolism and skeletal/cartilage calcification.
The second system, which is not widely known, is the distant actions generated via target cell generated intracellular calcitriol. Here, we only concentrate on the step by immune cells. Circulatory 25(OH)D enters into the cytosol of immune cells through diffusion from the circulation. In extra-renal tissues, as in immune cells, a cytochrome P450 enzyme CYP27B1 (1a-hydroxylase) converts 25(OH)D into 1,25(OH)2D (5).
Active vitamin D, 1,25(OH)2D binds to vitamin D receptor (VDR), a nuclear receptor family of ligand-regulated transcription factor (Bishop, Ismailova, Dimeloe, Hewison, & White, 2020). This interaction leads to most physiological effects of calcitriol through selectively regulating gene transcription (Christakos et al., 2016). In addition, target tissues such as immune cells also contain CYP24A1 enzyme that catabolise 25(OH)D and 1,25(OH)2D into inactive compounds; a built-in negative feedback loop to prevent the excess generation of calcitriol (Bishop et al., 2020). Important to note a significant biological difference between the hormonal calcitriol and that generated within the target tissue cells.
Autocrine and paracrine functions of calcitriol
Intracrine/autocrine synthesis of 1,25(OH)2D by macrophages and dendritic cells increase the expression of anti-microbial proteins, such as cathelicidin (LL37), and suppression of hepcidin enhances autophagy. Besides, calcitriol mediates several T-cell responses, including suppressing inflammatory T helper (Th)1 and Th17 cells and stimulating T-regulatory responses that enhance immunotolerance while preventing autoimmunity (Bishop et al., 2020).
As an autocrine signalling agent, it interacts with the intracellular VDR molecules: 1,25(OH)2D-VDR complexes enter the nucleus, binds to DNA strands, and up-or down- regulate dozens of genes, thus altering the cell’s behaviour. The type of genes modulated by calcitriol varies between the type of cells. For optimum autocrine actions, cells need a higher concentration of 25(OH)D concentration (50 ng/mL). Insufficient production of calcitriol within immune cells rapidly weakens immune functions that increase the risks for infections. As a result, some with severe vitamin D deficiency develop the disseminated infections, hyper-immune syndromes like cytochrome storm, autoimmunity and autoimmune diseases (Cao et al., 2020; Grant et al., 2020).
Epidemiological and observational studies strongly support a protective role of vitamin D in persons with COVID-19. Until the end of 2020, most published studies were retrospective or based on small samples (Castillo, 2020; Kaufman et al., 2020; Maghbooli et al., 2020; Meltzer et al., 2020; Panagiotou et al., 2020; Zemb et al., 2020). However, many valuable clinical studies have published in recent months. These include several randomised controlled clinical studies (RCTs) using vitamin D as the intervention in persons with COVID-19. Currently, there are more than 60 adequately powered, and large RCTs are underway in multiple countries.
Prolonged and weekend curfews are ineffective in controlling COVID-19
There is no scientific rationale for intermittent curfews, including weekends, to controlling COVID-19. Such is not only ineffective in preventing community spread of SARS.CoV-2 but also increase outbreaks. Moreover, lockdowns in 2020 significantly harmed people and livelihoods and the economy. Besides, such actions increased social disharmony, unemployment, loss of livelihood, alcoholism, addiction, domestic violence, child abuse, alcoholism, robberies, and the community spread of COVID-19 and deaths.
Another unintended consequence of curfew was restricting people to their homes that further prevented sun exposure. Therefore, lockdowns worsened the vitamin D deficiency and further increased the vulnerability to COVID-19 and developing complications. A similar thing happened when patients hospitalised for an extended period without providing nutrient supplements.
Types of lockdown that are acceptable to control COVID-19
During a pandemic or an epidemic, it is necessary to carry out extensive community surveillance and screening or diagnostic testing to identify and understand the extent and pattern of the spread of the disease. These real-time data are beneficial to make firm decisions to control the spread. Despite harm, there are occasions lockdowns are justifiable in pandemics. First, with a few cases or clusters in a limited area (smaller outbreaks), implementing geographically limited village or city limit-based lockdowns not exceeding 14 days.
Second, when there is a nationwide community spread (as happened in October 2020 and early April 2021), confining to houses and the country-wide strict travel restriction is necessary without exceptions, for not exceeding 21 days. In either case, the government must pre-arrange and ensure the availability of essential supplies, especially food, while giving a few day notices before imposing lockdowns.
When surveillance data indicate rapid community outbreaks spreading across districts suggests the need for a country-wide lockdown, but it must be done promptly. For example, in April 2021, a critical five-week delay before implementing the lockdown in Sri Lanka. That error led to a significant community spread across the entire country. Similar community spread occurred a few days after specific events. Examples include the USA, following the Memorial and Independence Day celebrations); April 2021 new-year celebration in Sri Lanka, and allowing large-scale religious festivals in India amid community outbreaks.
Following mentioned significant outbreaks, it may be necessary a country-wide lockdown for between 14 to 21 days. When data, however, indicate a lockdown, it must be implemented fully and promptly: doing so at the right time is essential. In these situations, shorter duration lockdowns are inappropriate and only increase the community spread of COVID-19.
Lockdowns drastically affect small businesses and self-employed and daily wage- earners, accounting for two-thirds of adults. It also markedly disrupted supply chains, tourism-related industries, and the national economy. The curfew also interrupted air travel, the hotel industry, and import and export trade, increasing food insecurity and imposed a significant financial burden on over 80% of the population. Therefore, the decision to lockdowns should not be taken lightly.
VITAMIN D DEFICIENCY, COMPLICATIONS AND PROGNOSIS OF COVID-19
Association between vitamin D intake and COVID-19 severity and deaths
Residents of nursing homes and disability centres have the worst combination ofhaving the highest prevalence of vitamin D deficiency and multiple chronic disorders (i.e., comorbidities). Besides, they have weaker immune systems, hence, vulnerable to infections. Moreover, residents in these institutions (so as prisoners), rarely get exposed to sunlight. Consequently, this group of people are highly vulnerable to contract COVID-19 and dying from it (Bäcker A, 2020): in 2020, the mortality rate exceeded 25% in some of these centres.
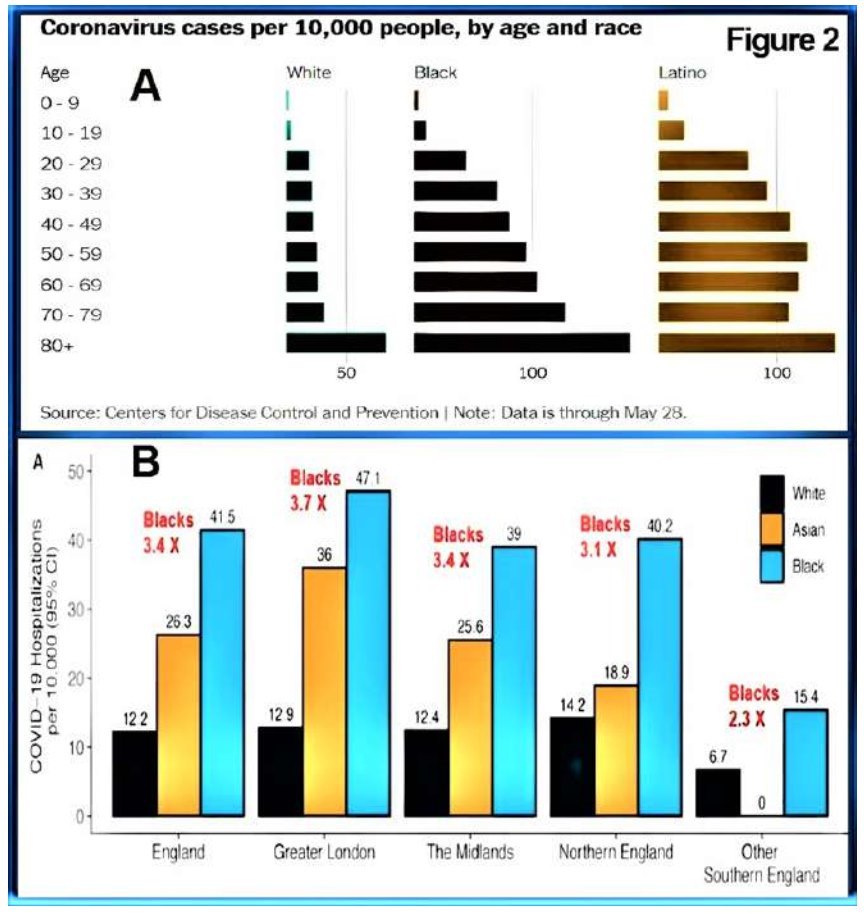
Additional evidence includes that the COVID-19 fatality rates were more than double in countries (A) with a high prevalence of vitamin D deficiency, (B) located in northern and southern latitudes, (C) have a higher percentage of elderly, and (D) among the black and ethnic minorities living in temperate regions (Bäcker A, 2020). These data strongly support hypovitaminosis D as a critical risk factor for COVID-19 infection, developing complications and death.
Ethnic minorities are at a higher risk
The intensive care (ICU) admissions and the death rates of Black, Asian, and Minority Ethnic (BAME) population living in temperate countries, such as the northern part of the USA, the UK, is over four-fold than for whites (Aldridge et al., 2020; Raisi-Estabragh et al., 2020; Trivedy et al., 2020). In addition, advanced age, chronic comorbid conditions (e.g., asthma, COPD, ischemic heart diseases, renal failure, obesity, diabetes, hypertension), and the male gender also have higher risks for developing complications and dying from COVID-19 (Ebadi & Montano-Loza, 2020; Hastie et al., 2020; Meschia et al., 2020; Temgoua et al., 2020).
Therefore, the groups mentioned above must be prioritised for treatment, including vaccination (Aldridge et al., 2020; Holmes et al., 2020). Violations of these, whether by politicians, law-enforcement officers, or administrators, must not be tolerated. In addition, the current policy and the approach by physicians not to intervene to boost patient’s immunity by D3 supplementation to achieve at least 50 ng/mL 25(OH)D is a grave mistake.
Ethnic, regional, and psychosocial differences related to hypovitaminosis D and increased mortality rates-racial disparities
Because of the routine intake of vitamin D supplementation and diets containing fatty fish, people in Scandinavian countries have a higher population average of 25(OH)D levels. Consequently, irrespective of other measures taken, the death rate is lower from COVID-19 in these countries, even though they located in northern latitudes (Bäcker, 2020). On the other hand, European countries with more significant ethnic minorities-those with melanin-rich skin (BAME groups) in the UK, Germany, and Italy, had disproportionately higher fatalities rate from COVID-19 in 2020.
There are examples of significant differences in death rates in ethnic minorities within a country. For instance, in Sweden, Somalian nationals comprise less than 1% of the total population but had 40% of deaths from COVID-19 (Brown, 2020; Osmancevic et al., 2016; Saaf et al., 2011). So, as in Muslim countries, as in Iran, Indonesia, and Middle Eastern countries (due to customs and harsh climatic conditions), women, in particular, minimise skin exposure to sunlight. This also happens in China, India, and Sri Lanka; people avoid the sun to prevent skin darkening, thus having relatively low serum 25(OH)D concentrations (Braiman, 2020).
For example, approximately 75% of suburban Indians and Chinese have low vitamin D. Hypovitaminosis D is common and is due to multiple reasons. As in India, despite plenty of sunlight, it can happen due to severe air pollution that filters out ultraviolet B (UV-B) rays. Another example is the African American population in the USA. Darker skin colour, impoverishment, and healthcare disparities have led to disproportionately high morbidity and mortality from COVID-19 (Bäcker, 2020). The underlying commonality, however, is the high prevalence of hypovitaminosis D.
Figure 2 illustrates (A) age-specific, racial disparity of infections and deaths from COVID-19 and (B) comparative hospitalisation due to COVID-19 among White, Asian and Black people in the UK and Blacks and Latinos in the USA (from, https://VitaminDWiki.com).
Plausible reasons why the reported incidences and death rates were low in tropical countries
The reported deaths from SARS.CoV- 2 in 2020 was unexpectedly low in tropical countries due to relatively higher population vitamin D due to inevitable sunlight exposure. Higher temperature and higher humidity
Figure 2.
are likely to reduce the community spread (Mecenas et al., 2020; Wang J, 2020), but some reports contradicted this (Auler et al., 2020). Low humidity enables exhaled droplets to dry into free-floating SARS-CoV-2 viruses, rather than the droplet and its viruses dropping to the ground quickly.
In 2020, a fewer number of PCR patients and deaths reported in south-east Ascian counties. Except for Singapore, other southeast Asian countries carried out less than 5% of PCR tests per million population than indicated. Because of the low number of testing, only a few patients detected. This erroneous policy continued till late October 2020, when they increased PCR testing to the current level. Because of the mentioned errors, Sri Lankan authorities continued falsely denying having the community spread of COVID-19. Even though data demonstrated that it had broad community spread since May 2020.
In addition to significantly low PCR testing, politically motivated coverups and statistical manipulations allowed countrywide dissemination of COVID-19 by the end of October 2020. Due to sunlight exposure, persons with severe vitamin D deficiency are few in most tropical countries. However, this is not the case in Gulf counties due to harsh climatic conditions. In these countries, severe hypovitaminosis D increased the number of people with symptomatic COVID syndrome needing ICU care, complications and deaths from SARS.CoV-2 virus (Neher et al., 2020; Wang, 2020).
Apart from Africa, most low and middle-income countries (LMICs-emerging economies and developing countries) located in the tropics. In contrast to the western- industrialised countries, LMICs have less international travel and large gatherings in confined spaces (e.g., arenas, stadiums, etc.). Moreover, percentage of younger people are higher than older people (Chung et al., 2020).
For the mentioned reasons, generally, there is a lower prevalence of COVID-19 and deaths in tropical LMICs.
Specific issues related to India and China-vulnerability to COVID-19:
Neither India nor China is considered wholly tropical countries, although both have some states and provinces deemed tropical. In these two countries, the states with higher average sunlight had lower complications and mortality rates from COVID-19 but higher mortality reported in the northern states.
In India and Sri Lanka, the third wave of COVID-19 could have mitigated if they had the right policies and taken affirmative actions timely. In contrast to Sri Lanka, air pollution is very high in cities and suburbs in India and China, which filter the UVB rays reaching the earth surface. Besides, in both countries, the prevalence of vitamin D deficiency exceeds 70% when measured by conventional standards, either with 20 ng/mL or 30 ng/mL thresholds.
No country has more than a fraction of its population with serum 25(OH)D concentrations 50 ng/mL or more, all year round: regarding the immune system, the true measure of vitamin D repletion. Unlike India, despite China has widespread vitamin D deficiency, the government has managed to control COVID-19 through strict quarantine and compulsory public health measures, and most recently with vaccination. However, the local vaccines likely to fail to control the emerging new variants of SARS.CoV-2, China is expected to see a delayed third wave spreading across the country due to mutated viruses.
India and China both have a high incidence of air pollution in suburban regions and poverty in rural areas, and a high prevalence of vitamin D deficiency. Consequently, both countries have an increased risk of getting an uncontrollable third wave of COVID-19 extending to the winter period driven by new variants [such as Beta (South African variant), Gamma (Brazilian variant), Delta (Indian variant), and future virulent strains).
In addition to being more infectious, some new mutants are less vulnerable to the antibody-mediated immunity induced by prior infection and/or by AstraZeneca and Sinopharm (or other two Chinese) vaccines. Thus, vulnerability to develop complications and deaths will be significantly high in those with vitamin D deficiency. Neither country, however, are proactively addressing this issue.
Reasons for fewer deaths in homeless people
Despite rampant malnutrition and major socioeconomic disadvantages, in many countries, such as northern parts of the USA and the UK, homeless people and farmers had significantly less symptomatic COVID-19 and deaths than those who live in the suburbs. This unexpected significant trend explained by their daily routine exposure to sunlight. A similar phenomenon observed among slum-dwellers in India and Brazil: some groups had over 50% antibodies against SARS.CoV-2, but the death rate was less than 0.2% (Yadaver, 2020).
The above-unexpected phenomena most likely due to the robust vitamin D status of mentioned groups through their outdoor lifestyles. As a result, they have a higher average serum 25(OH)D concentrations than suburban populations and better innate immunity to fight against coronaviruses. Besides high fertility rates (larger family size) and lower life expectancy, these groups have fewer elderly. Moreover, most homeless people in the USA and UK consist of African and Hispanic origin and Asians (i.e., darker skin, non-white ethnic groups). The melanin-rich skinned people who live in suburban areas in northernly located countries had a high death rate due to COVID-19. In contrast, despite having darker skin, fewer homeless people died from COVID.
Link between hypovitaminosis D and severe complications from COVID-19
In the USA, UK, and a few other countries, melanin-rich skin population racial/ ethnic groups had a significantly higher mortality rate from COVID-19 than whites (Raisi-Estabragh et al., 2020; Trivedy et al., 2020). These data should have prompted immediate public health actions to alleviate hypovitaminosis D. Instead, uninformed authorities and conflicted scientists continue to refute these data than accept them readily and take steps to rectify the root cause leading to prevent unnecessary deaths. Strengthening the population immunity through natural means significantly reduce the incidence, severity, and deaths and the need for ICU admissions (Wimalawansa, 2020c, 2020f, 2020c).
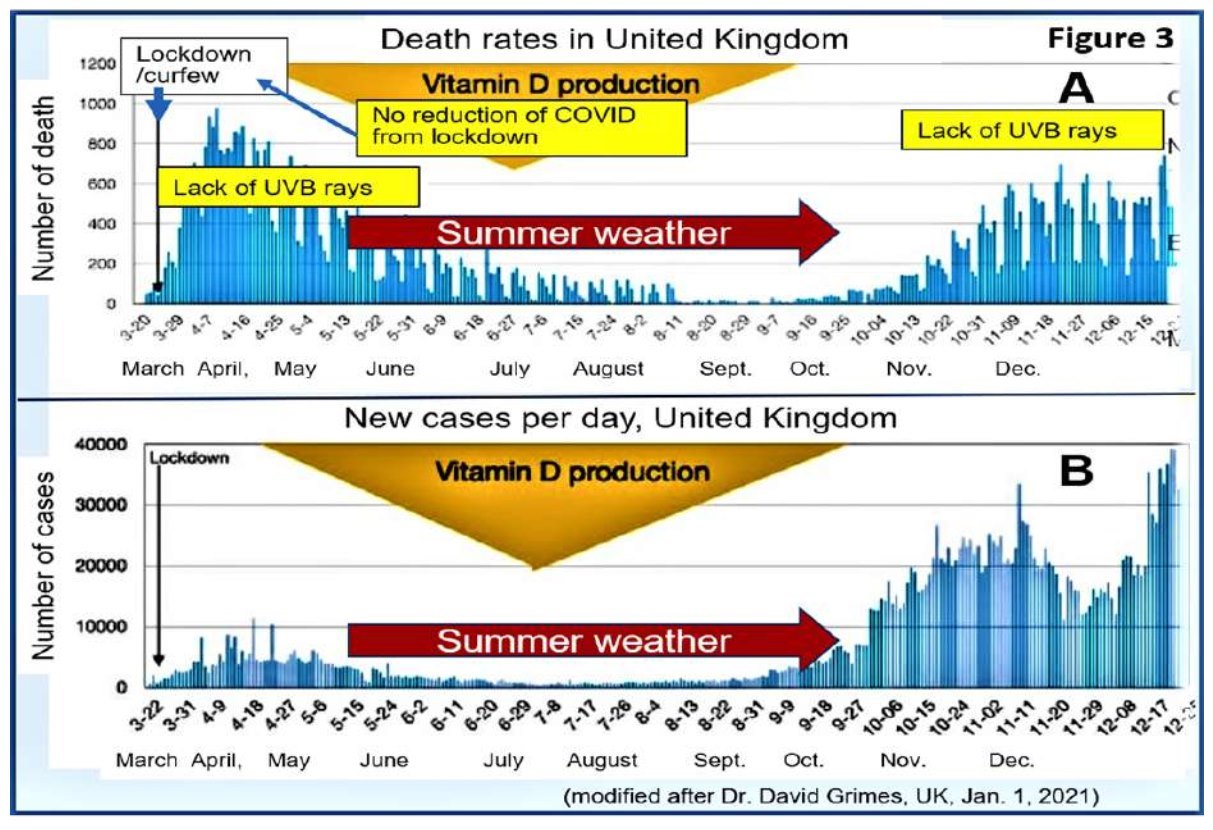
The example in Figure 3 illustrates the unique beneficial effects of exposure to UV-B rays during the summertime in the UK. So as in the USA, the rate of infection and deaths were significantly lower during the summer
months of 2020: a similar effect will occur in 2021, with potential outbreaks of COVID-19 from November 2021. The reported decline of COVID-19 incidence and deaths in 2021 in the USA and UK is due to the summer effect and vaccination. Nevertheless, both administrations entirely rely on vaccines to eliminate COVID-19, which is highly unlikely to happen.
Importance of strengthening the immune system to overcome viral infections
Human immune systems depend on a balanced diet rich in vitamins (D in particular), minerals (zinc and selenium) and other essential components, such as omega-3 fatty acids (ratio), quercetin, magnesium, etc. Vitamin D deficiency weakens the innate immune system, as its driver most immune functions. It increases the vulnerability to contracting microbial infections, including viruses and developing complications.
(modified after Dr David Grimes. UK. Jan 1.2021)
Vitamin D3 is arguably not a vitamin since people can synthesise it themselves. However, as noted above, foods, including fortified foods, do not provide adequate quantities of vitamin D. The situation aggravated due to sun avoiding behaviour and inadequate UVB exposure year-round. Thus, even a “balanced diet” does not provide vitamin D to support the immune system for most people unless supplemented with vitamin D.
The 5,000 IU per day D3 is generally sufficient for 70 kg non-obese adult to attain these levels, yet some viewed it as a large dose due to misunderstanding. However, one IU is 1/40th of a microgram-the amount a six-gram baby rat requires per day to avoid rickets. 5,000 IU/day or a single 50,000 IU capsule every ten days equals 0.125 mg of vitamin D a day (this equal to one gram administered, once every 22 years). Pharma grade D3 costs $2.50 per gram, ex-factory, in 1 kg lots: so, it is not an expensive compound.
The role of vitamin D in the immune system
Innate and adaptive immunity from Immune cells, anti-inflammatory and antioxidative regulatory functions rely on having over 50 ng/mL circulating 25(OH)D concentration (physiological levels needed for cells). This applies to the modulatory effects of all immune cells and autocrine (within) and paracrine (adjacent) signalling systems. Therefore, serum 25(OH)D concentration is an easily monitored, excellent indicator of overall immune status.
Vitamin D dependent release of products and actions from immune cells include antibodies and anti-microbial peptides, macrophage-, dendritic cells, WBC-mediated removals of pathogens, overcoming excessive inflammation and oxidative stress. Conversely, hypovitaminosis-induced weakened immune systems (the frontline defence system) make people vulnerable to viruses, such as SARS- CoV-2 and increases the risks for developing complications and deaths.
The successful autocrine and paracrine immune signalling occurs when the serum 25(OH)D concentrations are above 50 ng/ mL. Such levels not only provide the most robust and fastest possible innate and adaptive defences against pathogens. In addition, it enables Th1 lymphocytes and other types of regulatory immune cell to function correctly. In contrast, when autocrine/paracrine signalling systems fail, Th1 lymphocytes remain stuck in their initial pro-inflammatory program long after they should have switched to their antiinflammatory shutdown program.
This failure in Th1 lymphocytes from the lungs of hospitalised COVID-19 patients - and other similar failures is arguably the cause of severe COVID-19, as the dysregulated inflammatory attack damages the pulmonary endothelium. This gives rise to hyper-coagulative blood and micro-embolism, which causes hypoxia and lead to the formation of larger clots that damage the lungs, heart, central nervous system, and kidneys. The exact molecular processes by which these Th1 lymphocytes fail, elucidated by McGregor et al. in July 2020 (McGregor, 2020).
Properly functioning immune cells also control oxidative stress by improving mitochondrial functions, suppressing the expression of inflammatory cytokines, and subduing the renin-angiotensin hormonal system (RAS) (Adams et al., 1989; Grant, Lahore, et al., 2020). Furthermore, they increase the secretion of anti-inflammatory cytokines and anti-microbial peptides, cathelicidin and defensin, while stimulating all immune cells (Antal, Dombrowski, Koglin, Ruzicka, & Schauber, 2011), including macrophages.
A lost opportunity (lessons to learn)
In mid-March 2020, we suggested to the government of Sri Lanka a comprehensive national program to accomplish the above through sun expose and vitamin D supplementation for the entire adult population. This would have cost only a half of one day’s opportunity costs of curfew. Such a program would have prevented the need for curfew entirely and associated economic calamities. Taking such proactive measures needs an understanding of the big picture. It would have mitigated most economic losses and avoided lengthy lockdown and curfews (Wimalawansa, 2020b, 2020c). Supporting primary and emerging clinical data including RCTs, and recent reviews and meta-analyses, provided strong evidence of the benefit from vitamin D supplements. However, in March 2020, we needed the knowledge to make the right decision (Wimalawansa, 2020a, 2020d, 2020e).
These include (A) a significantly higher percentage of people who died from COVID-19 had severe vitamin D deficiency, (B) countries with lower population vitamin D status (winter effect) had the highest COVID- 19-related mortality, and (C) a strong, inverse linear relationship exists between lower serum 25(OH)D concentrations of individuals and the severity of complications and deaths from COVID-19 (see the section below under clinical and RCT studies) (Castillo, 2020; Kaufman et al., 2020; Maghbooli et al., 2020; Meltzer et al., 2020; Panagiotou et al., 2020; Zemb et al., 2020).
Overall data strongly suggests a significant overall benefit of population-wide vitamin D supplementation to boost and strengthen innate immunity, especially in viral epidemics and pandemic. Together with safe sun exposure guidance, and mandatory wearing of facemasks and keeping social distance, it would have the most tangible benefit on minimising the spread of COVID-19 and deaths at least cost and economic disruption (Wimalawansa, 2020c, 2020d). Mentioned interventions are, however, not mutually exclusive.
Reasons for the inability to achieve herd immunity against SARS.CoV-2
Based on data, one could envisage that strengthening the immune system of everyone would prevent (or eliminate) the infection, reduce risks of severe complications and deaths. However, it is not glamorous for politicians and administrators and is not as profitable for companies as expensive antiviral agents, monoclonal drugs, and vaccines. In contrast, vaccines strengthen a narrow area of the acquired immune system, while vitamin D predominantly strengthens the immune system as a whole at a fraction of the cost.
For several scientific reasons, we predicted that, as per the current approach by the WHO and other authorities and the greed of pharmaceutical companies refusing to relax patent rights, it would be impossible to achieve herd immunity against SARS.CoV-2. Instead of eliminating the virus, the industry wants COVID vaccines to become a profitable annual event. Thus, relying on vaccines and eliminating public health and nutritional aspects is a grave miscalculation. Instead, the most sensible approach is to achieve population vitamin D sufficiency to strengthen innate immunity and use a broader vaccination program to induce acquired immunity.
CLINICAL STUDIES AND RANDOMISED CONTROL STUDY DATA
Over 60 randomised clinical studies are currently registered in clinical trial registries and are in progress: results will be available in mid to late 2021. Emerging data suggest that sufficient vitamin D levels in individuals and populations would protect against infections like SARS.CoV-2 (Wimalawansa, 2020a, 2020c). Several recent prospective clinical studies reported that vitamin D supplements prevented complications (Ilie e al., 2020; Merzon et al., 2020), such as cytokine storm (Crane-Godreau et al., 2020) and deaths from COVID-19.
For persons infected with COVID-19, the need for admitting to intensive care units (ICU) was significantly (between 60 and 80%) reduced in those who are vitamin D sufficient or supplemented with high-dose vitamin D or calcifediol administered earliest possible (Castillo, 2020; Grasselli et al., 2020) (Annweiler et al., 2020; Bäcker, 2020; Cangiano et al., 2020; Castillo, 2020; Chaccour et al., 2020; Kaufman et al., 2020; Maghbooli et al., 2020; Meltzer et al., 2020; Merzon et al., 2020; Radujkovic et al., 2020; Rastogi et al., 2020; Tan et al., 2020; Vassiliou et al., 2020). Detailed critical published studies related to vitamin D and COVID-19 are available on the following websites:
https://vdmeta.com/
https://VitaminDWiki.com/
https://c19study.com/d
https://vitamind4all.org/letter.html
https://c19ivermectin.com/
VitaminD.Wiki-Prof.Wimalawansa- COVID overview-Dec. 2020
https://c19study.com/
https://aminotheory.com/cv19/#vc
https://vitamindstopscovid.info/
The benefit of vitamin D in other viral diseases:
Vitamin D deficiency is associated with increased risks not only from COVID-19 but also from other viral illnesses. Adequate circulating 25(OH)D reduces the severity and subdued hyper-inflammatory syndrome and related complications (Guo et al., 2020; Wimalawansa, 2020b). Several studies reported that vitamin D sufficiency reduced the severity and deaths from dengue fever (Loke et al., 2002; Villamor et al., 2017), especially relevant for Sri Lanka. Others have noted that supplements of 4,000 IU for 10-days were significantly more effective than 1,000 IU in reducing dengue virus replication and controlling pro-inflammatory cytokine-induced hyper-reaction (Arya & Dwivedi, 2020). However, bolus D3 or, better still, a single 1 mg dose of calcifediol would have been more effective in a rapid recovery from dengue and reducing complications.
Examples of highly expressed pro- inflammatory cytokines are TNF-a, INF-y, IL-1p, IL-6, -12, -17, -19 and -33 and NFkB. The expression of inflammatory cytokines and chemokines suppressed by higher circulating 25(OH)D, zinc, and PUFAs (Story, 2021). Mentioned cytokines are released mainly from mast cells, neutrophils, dendritic cells, white cells, Th-1, -2, -18, and Treg cells, causing generalised inflammation. These inflammatory cytokines are present in high concentrations in the circulation, especially in those who are developing complications and dying from COVID-19.
Mentioned inflammatory markers could use for early identification of the worsening condition. For example, to identify those who would benefit from the administration of potent glucocorticoids. These inflammatory markers are also present in moderate levels in the circulation in ageing, uncontrolled diabetes, chronic pulmonary and cardiovascular diseases, and cancer. However, in persons with COVID-19 complications, these are at much higher concentrations than in the aforementioned conditions (Story, 2021).
SCIENTIFIC EXPLANATIONS FOR THE LINK BETWEEN VITAMIN D DEFICIENCY AND COVID-19 OUTCOMES
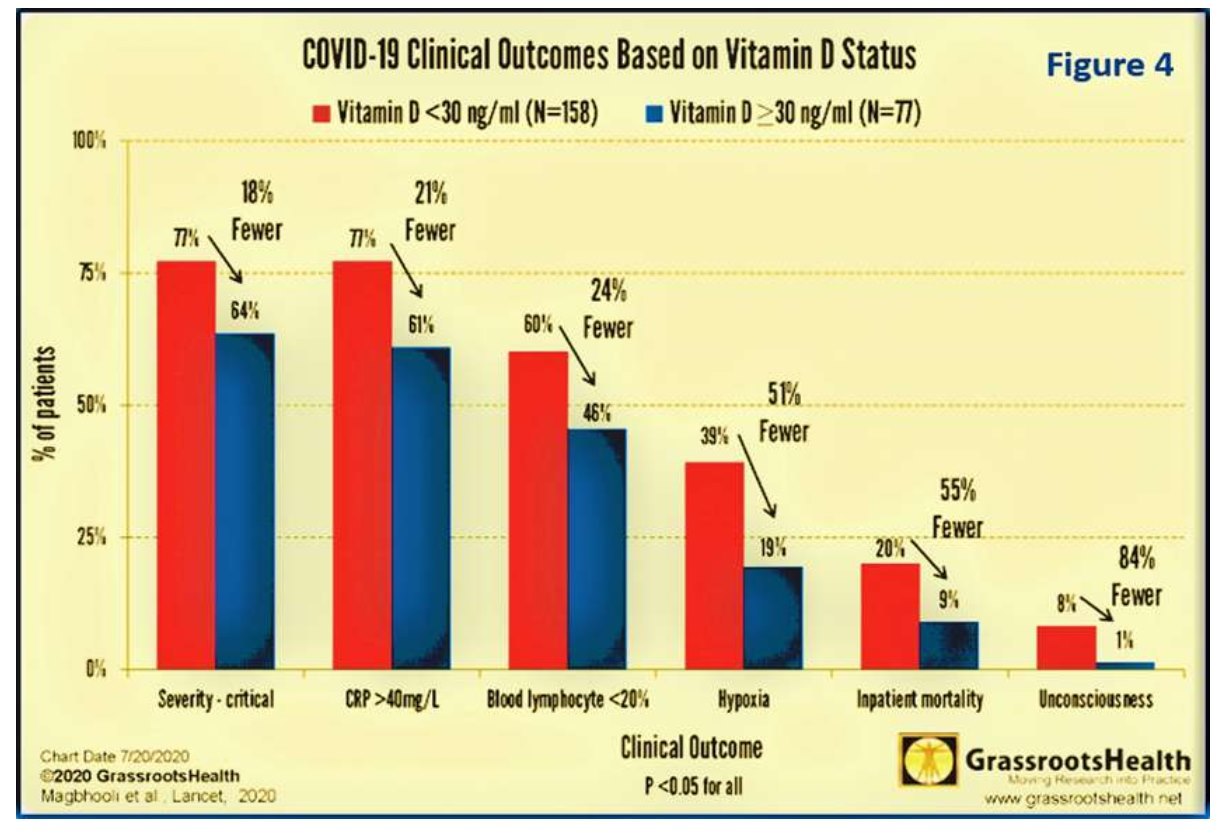
A study by Grassrootshealth reported that better clinical outcomes, including low levels of biochemical markers of inflammations, are associated with higher serum 25(OH) D concentrations. Figure 4 illustrates the vitamin D status based clinical outcomes and associations using a cut-off point of 30 ng/mL as the normal: (A) Less severe COVID-19 and lower lymphocyte counts (a marker of mildmoderate cases); (B) Less inflammation (lower CRP) and possibly reduced risk of cytokine storm; (C) 50% fewer cases of hypoxia, 84% fewer cases of unconsciousness, and 55% fewer deaths (Figure 4: after, Grassrootsthealth, 2020)
Innate immunity, vitamin D, and COVID-19
The ability to control invading microbes is dependent on robust innate immunity. In these circumstances, a rapid immune cells- mediated production of anti-microbial peptides and neutralising antibodies occur, together with chaperoning microbes to macrophage and dendritic cells to destroy them (Wang & Wang, 2004). Furthermore, the peptides defensins and cathelicidin (LL37; a 37 amino acid cationic peptide generated by extracellular cleavage of hCAP18 protein by serine proteases) (Kahlenberg & Kaplan, 2013) neutralise the cellular invasion of COVID-19. Therefore, irrespective of age, gender, or skin colour, severe complications following COVID-19 (e.g., cytokine storm, ARDS, and death) occur at the highest among those with severe vitamin D deficiency (Crane-Godreau et al., 2020).
A previous meta-analysis (2017; a multicountry, 25 RCTs) reported favourable outcomes from viral respiratory tract infections in those with vitamin D3 supplements (Martineau et al., 2017). In parallel, serum 25(OH)D concentrations inversely correlated with clinical outcomes from COVID-19 syndrome (El Zowalaty & Jarhult, 2020; Wimalawansa, 2020c). Persons with severe vitamin D deficiency have weak innate immunity, the highest risk of contracting COVID-19 infection (Ghasemian, 2020; Molloy & Murphy, 2020; Wimalawansa, 2020d) and dying from it (AlSafar et al., 2021).
Vitamin D enhances resistance to viral illnesses
Several clinical studies in acute respiratory tract infections have demonstrated significant clinical improvements following vitamin D supplementation (AlSafar et al., 2021; Grant, Lahore, et al., 2020; Hribar et al., 2020; Martineau et al., 2017). In addition, studies reported that higher serum 25(OH)D concentrations strengthen immune responses to seasonal and winter-associated respiratory tract infections: a significant, 50% reduction of incidence and duration of the illness (Boucher, 2012; Kumar, 2021; Sabetta et al., 2010).
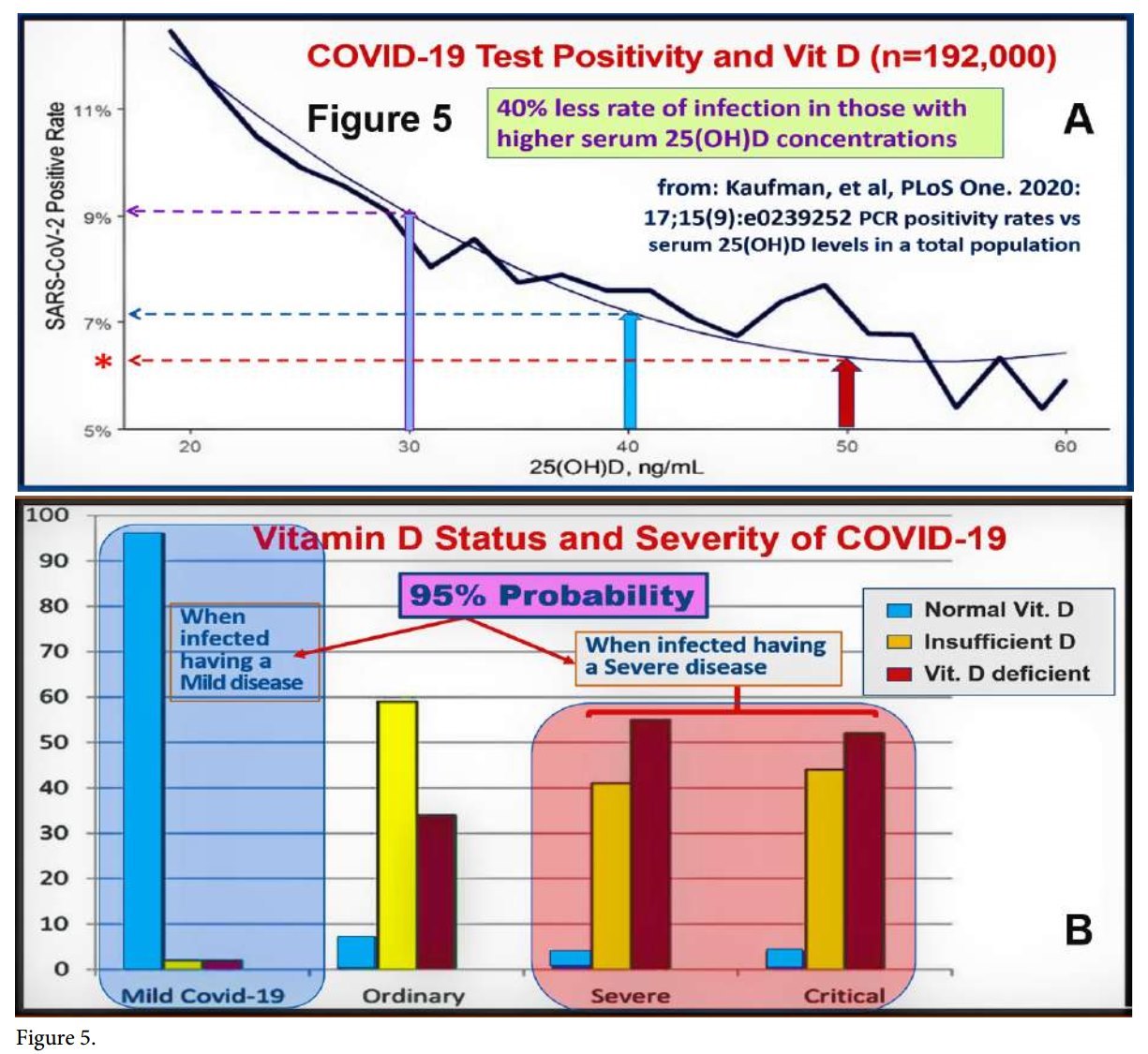
Another meta-analysis revealed that 89% of persons infected with COVID-19 had low serum 25(OH)D concentrations [46% were deficient, and 43% were insufficient (Ghasemian, 2020) (https://vdmeta.com/). Furthermore, an inverse relationship reported between vitamin D status and the recovery/ mortality rates from COVID-19 in different countries. Figure 5 illustrates the relationship between serum 25(OH)D concentration and (A) PCR positivity persons (i.e., increased probability of getting infected), and (B) the relationship probability of getting infected versus serum 25(OH)D status.
Acute respiratory distress syndrome (ARDS) is one of the most common lethal condition in persons with COVID-19; generally, patients develop it in ICUs: a single dose of calcifediol can prevent that (Castillo, 2020; Nogues, 2021). Moreover, 1,25(OH)2D establishes a balance between lipopolysaccharide (LPS) induced damage and subdue the RAS in persons with ARDS that helps recovery (Xu, 2017).
However, to achieve such protection, the minimum serum 25(OH)D concentration needed is 40 ng/mL 100 nmol/L)(Grant et al., 2020; Holick, 2009). However, Quraishi et al. study confirmed (Quraishi et al., 2015) that in the presence of any infections, the immune functions are most effective when serum 25(OH)D concentration maintained above 50 ng/mL. As mentioned above, the dose range needed to achieve such varies between 5,000 to
IU/day (a longer-term intake need) or ?????
IU once a week.
Most effective serum 25(OH) D concentration to overcome COVID-19
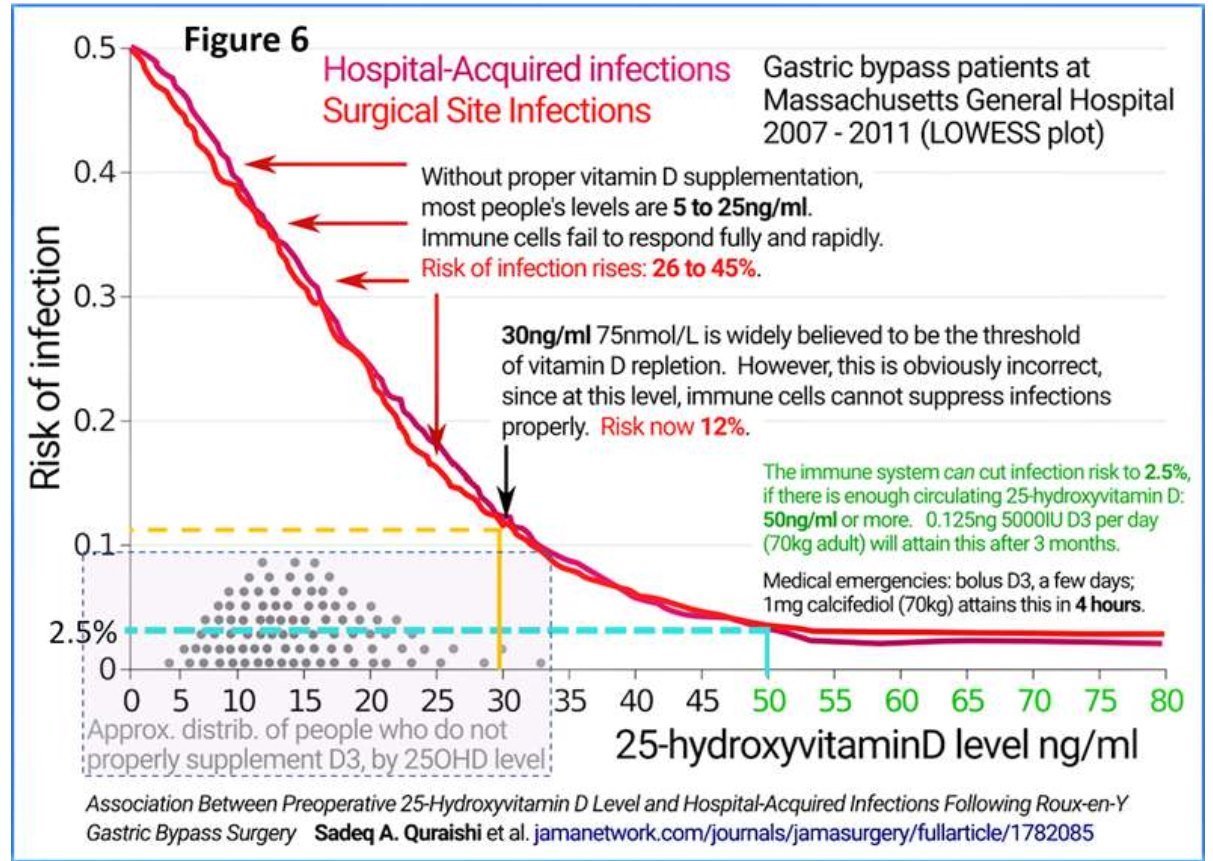
Quraishi et al. also demonstrated that the minimum serum 25(OH)D concentration necessary to generate optimum immune responses against infections is 50 ng/mL. Such sustained serum 25(OH)D concentration will maintain robust innate and adaptive responses against invading pathogens. It also prevents pro-inflammatory responses and immune dysregulation. These data directly apply to severe COVID-19 syndrome, Kawasaki- like syndromes, multi-system inflammatory syndrome, sepsis, and ARDS. The minimum serum 25(OH)D concentration needed to control mentioned disorders is above 50 ng/mL (125 nmol/L.) (Quraishi et al., 2014) (Figure 6).
The root causes of COVID-19 complications and death
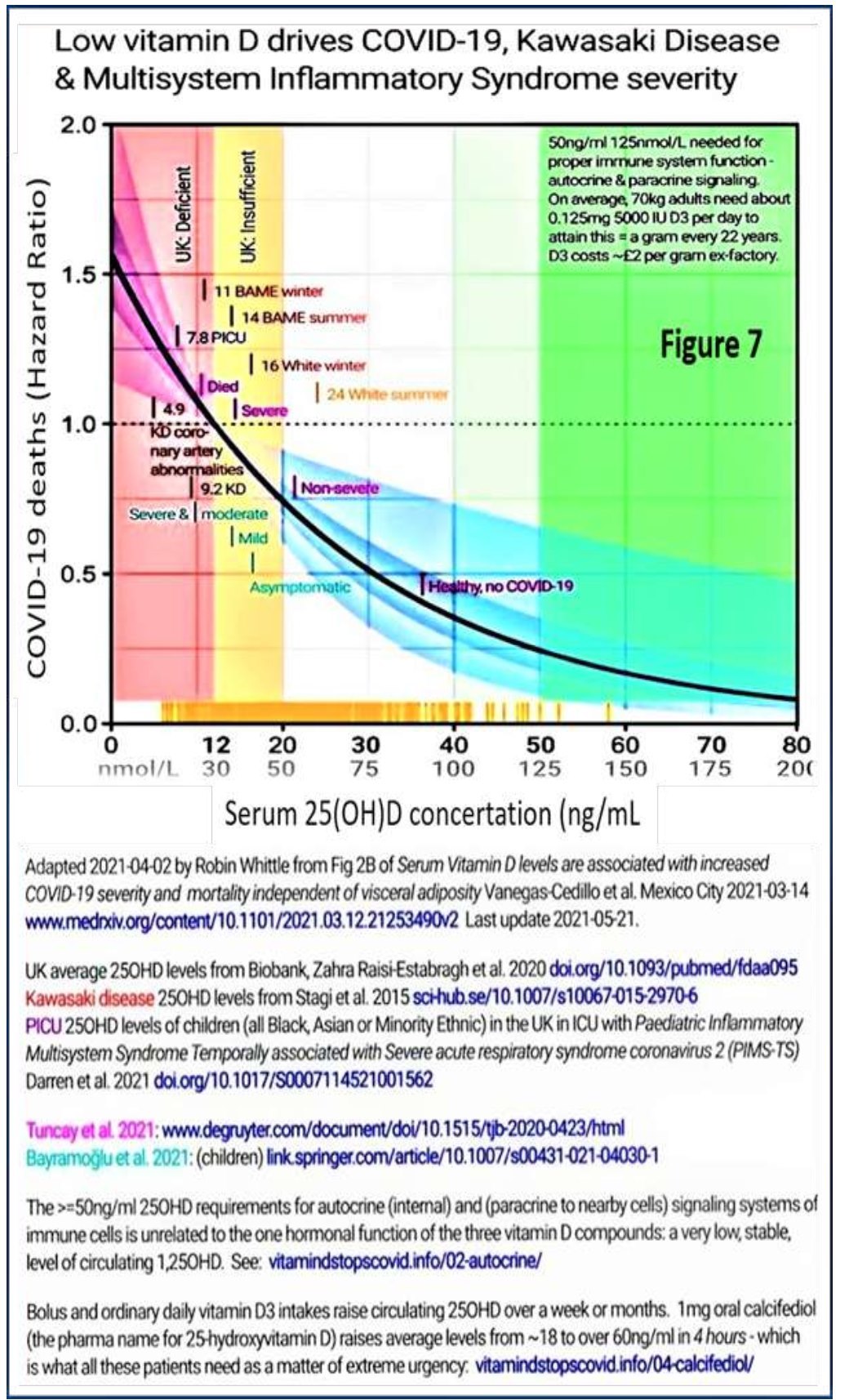
Vitamin D deficiency weakens innate and adaptive immune responses. The risk reduced to less than 2.5% (optimised immune responses) when the serum 25(OH)D concentrations are kept above 50 ng/mL. Figure 7 illustrates COVID-19 risks reported by several data sets based on circulating 25(OH)D levels.
The accepted minimal concentration of vitamin D sufficiency, as considered by the (American) Endocrine Society guidelines of 30 ng/mL developed to ensure the kidneys produce sufficient hormonal 25(OH)D. However, this concentration is not high enough to adequately diffuse into immune cells. Most patients admitted with COVID-19 have 25(OH)D concentrations between 5 and 20 ng/mL, which is insufficient for immune protection (Figure 6). Those who develop severe complications or die typically have less than 12 ng/mL (AlSafar et al., 2021; Radujkovic et al., 2020).
Vitamin D protects people from COVID-19:
Observational and retrospective clinical studies have reported strong associations between serum 25(OH)D concentration and the incidence, severity, and deaths from COVID-19 (Garg et al., 2020; Molloy & Murphy, 2020; Shiravi et al., 2020; Stohs & Aruoma, 2020). Nevertheless, there are other reasons for low serum 25(OH)D concentrations in hospitalised patients.
Above include prolonged hospitalisation or institutionalisation for any reason and lack
Low vitamin D drives COVID-19, Kawasaki Disease & Multisystem Inflammatory Syndrome severity
Serum 25(OH)D concertation (ng/mL
Adapted 202104-02 by Robin Whittle from Fig 2B of Serum Vitamin D levels are associated with increased COVID-19 seventy and mortality independent of visceral adiposity Vanegas-Cedilloetal. Mexico City 2021-03-14 www.medrxiv org/content/101101/20210312.21253490v2 Last update 2021 -05-21.
UK average 250HD levels from Bobartk, Zahra Raisi-Estabragh et al 2020 doi.org/101093/pubmed/fdaa095 Kawasaki dsease 250HD levels from Stagi et al 2015 sohub.se/10.1M7/s10067-015-29706 P1CU 250HD levels of children (all Black, Asian or Minority Ethnic) in the UK in ICU with Paediatric Inflammatory Multisystem Syndrome Temporally associated with Severe acute respiratory syndrome coronams 2 (PIMS- TS) Darren etal 2021 dot.org/101017/S0007114521001562
Tuncaytlal 2021 wwwdegruyter com/document/doi/l 01515/tjb-20200423/html Bayiwrx;’u et ai 2021 (children) Imfcspringercom/artide/10.1007/s0043l -021-040301
The >=50ng/ml 250HD requirements for autocnne (internal) and (paracnne to nearby cells) signaling systems of immune cells s unrelated to the one hormonal function of the three vitamin D compounds: a very low, stable, level of circulating 1.250HD See vitam»idstopscovid.info/02-autocrine/
Figure 7.
Bolus and ordinary daily vitamin D3 intakes raise arculating 250HD over a week or months 1 mg oral calcifediol (the pharma name for ZShydroxyvitamin D) raises average levels from -18 to over 60ng/ml n 4 hours ■ which is what all these patients need as a matter of extreme urgency: vitarrwxistopscovrd. info/04-calcrfed»ol/
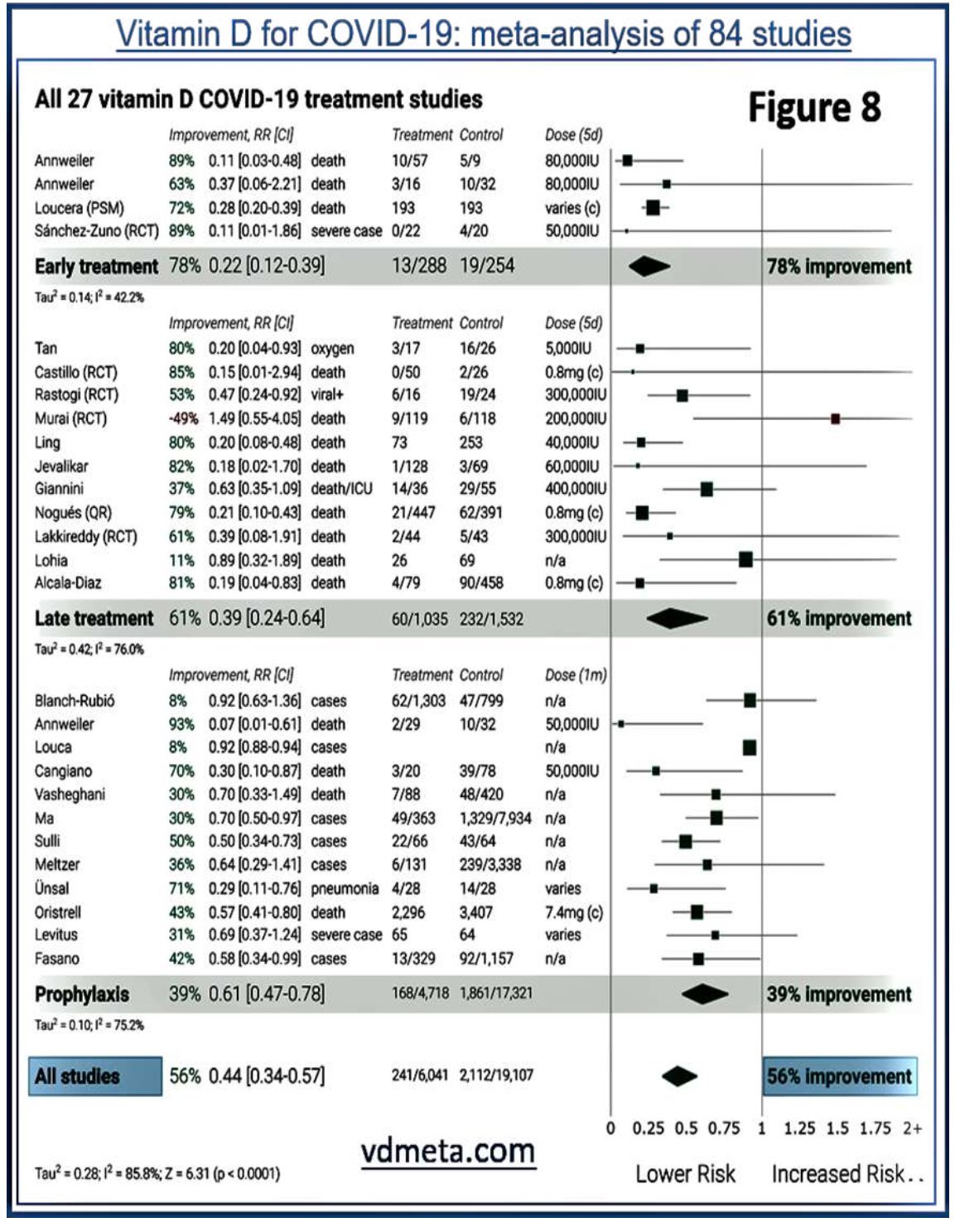
exposure to sunlight or pre-existing vitamin D deficiency: these cascade vulnerabilities to infections. A slight reduction in circulating 25(OH)D can also arise from its consumption by immune cells during illness, especially grossly dysregulated, self-destructive/ autophagy, inflammatory responses. Figure 8 illustrates data from a recent meta-analysis
from 84 clinical studies, demonstrating the efficacy of vitamin D in the early and later stages of COVID-19 (from: vdmeta.com).
In addition to the diurnal variation of serum 25(OH)D concentrations, reduced levels can also occur following severe acute illnesses, including COVID-19: this known as reverse
Vitamin D for COVID-19: meta-analysis of 84 studies
causality (French et al., 2019). In addition, other acute and chronic diseases could also modulate serum 25(OH)D concentration (Hastie et al., 2020; Martineau et al., 2015). To overcome these, some suggested using the 25(OH)D concentrations measured at least two weeks before developing symptoms of COVID-19 to avoid confounders (French et al., 2019). Nevertheless, the effects of COVID-19 on lowering serum 25(OH)D concentration insignificant.
Hypovitaminosis D increases RAS activity and thus, worsen COVID-19 outcomes
In addition to lesser sun exposure, the skin’s ability to produce vitamin D from UVB gradually decreases after 50 years (Gasmi et al., 2020; Grant et al., 2020). Therefore, it is common to have low serum 25(OH)D concentrations in the elderly. For example, in northern Italy and Spain, older persons have a high prevalence of severe vitamin D deficiency correlated with death rates (Saaf et al., 2011; Yousef et al., 2019). Furthermore, several studies reported that the geographic distribution of patients with severe complications from COVID-19, related to less sun exposure, has been attributed to vitamin D deficiency (Bäcker, 2020).
Consequently, above 70% of the USA nursing homes (Okan et al., 2020), developmental disability facilities (Grant et al., 2015), and institutionalised people (Wimalawansa, 2012), and those with chronic renal failure on dialysis shown to have severe vitamin D deficiency and at high risk for viral diseases (Ilie et al., 2020; Kennel et al., 2010). Because of the high incidence of severe vitamin D deficiency, 40% of deaths from COVID in the USA occurred in the above groups.
Reduced 25(OH)D concentrations are typical in those with metabolic disorders such as obesity, diabetes, hypertension, COPD, cardiovascular diseases, and those who get less exposure to sunlight. Therefore, they have a higher risk for COVID-19 and developing related complications and deaths. Besides having darker skin, synthesising vitamin D is reduced in those who live in higher and lower latitudes (Grant et al., 2020; Hastie et al., 2020). Hypovitaminosis D not only causes weaken innate immunity and aggravates common systemic diseases, such as diabetes, obesity and metabolic syndrome.
Poor COVID-19 outcomes are mostly due to hypovitaminosis D
Evidence of point toward a strong association between vitamin D status and COVID-19 complications and mortality. Despite mounting evidence, many governments, medical associations, and some sponsored scientists downplay the beneficial role effects of vitamin D in COVID syndrome due to conflicts of interest. Either they dismiss by stating that there is no evidence of vitamin D benefits or unwilling to recommend vitamin D supplements.
These ill-informed sources and biased recommendations assumed that evidence to link vitamin D and COVID-19 is insufficient, theoretical, and emphasise the possible harm from the extremely low incidence of toxicity (https://vitamind4all.org/letter.html) (Hunter et al., 2020; Rhodes et al., 2020). Figure 9 illustrates the effectiveness of vitamin D in different states of COVID-19 syndrome.
UTILISING VITAMIN D AS A PROPHYLACTIC AND THERAPEUTIC TOOL
Minimum serum levels of vitamin D needed to overcome disorders
Research suggests that 40-60 ng/mL is needed to prevent non-musculoskeletal, including viral respiratory infections. Others recommend maintaining concentrations between 50-80 ng/mL to reverse hypertension and cardiovascular diseases [47] and minimise infections (Quraishi et al., 2014). In a randomised controlled trial, subjects with vitamin D insufficiency who took 10,000 IU/ day for three years did not develop any adverse effects (Burt et al., 2019).
Figure 9.
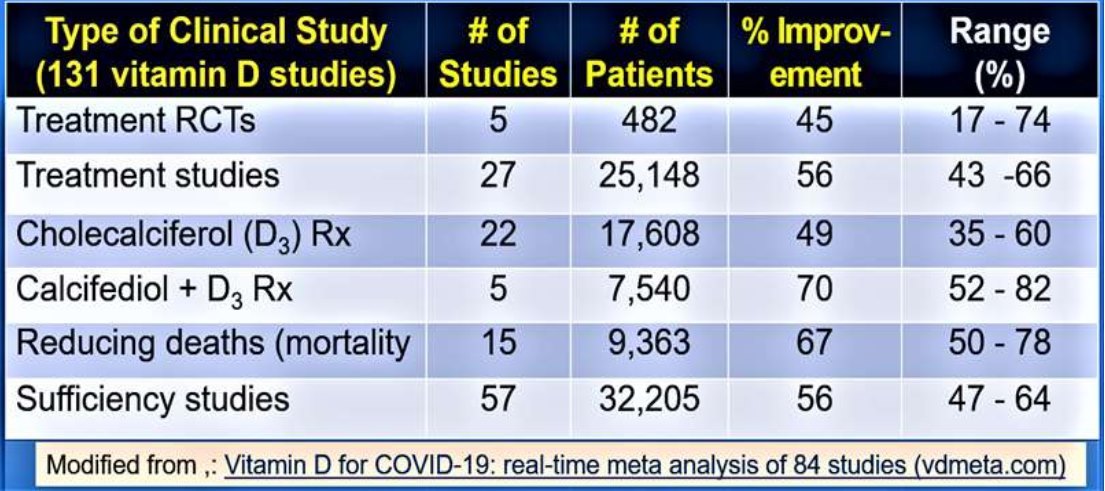
While 400 IU/day treated group did not increase the mean 25(OH)D concentrations, those who took 4,000 and 10,000 IU/day increased their serum levels by 53 ng/mL and 58 ng/mL, respectively (Burt et al., 2019). Another meta-analysis of vitamin D supplementation indicated that long-term daily supplementation of vitamin D, 5,000 IU/ day was safe and effective in preventing acute respiratory infections (Kakodkar et al., 2020; Martineau et al., 2017). Table 1 summarised outcomes from 131 clinical studies, intervene with vitamin D in persons with COVID-19, up to the end of April 2021.
Potential Adverse Effects of Vitamin D
Vitamin D toxicity occurs only in serum concentrations above 150 ng/mL and associated with hypercalcemia-related signs and symptoms (Hribar et al., 2020; Marcinowska-Suchowierska et al., 2018) (Kimball et al., 2017). Historically, toxic levels of vitamin D have almost exclusively resulted from industrial errors (inaccurate doses of supplements) and a few cases of deliberate overuse. Stopping the supplement and maintaining hydration is generally adequate to reverse the situation (Lim & Thadhani, 2020; Spiller et al., 2016).
Table 1.
[ Modified from Vitamin D for COVID-19: real-time meta analysis of 84 studies (vdmeta.com) ~] even when compared to those with vitamin D insufficiency (p<0.02) and infrequent administration (Martineau et al., 2017).
Clinical studies reported that vitamin D supplementation is exceptionally safe, even when using short-term high doses and longer-term moderate doses (Martineau et al., 2017). However, clinical investigations must be conducted based on the serum 25(OH)D concentrations achieved, not the amount of vitamin D administered or consumed (Fabbri et al., 2020; Haq et al., 2018; Pludowski et al., 2018; Wimalawansa et al., 2017).
Randomised control clinical trial on prevention of COVID-19 in Sri Lanka
Previous studies reported that vitamin D supplements prevent acute respiratory tract infections, reduce the severity [66], and have a dose-related effect on subduing respiratory infections (Martineau et al., 2017). Also, vitamin D deficiency increases the severity of acute respiratory infections and mortality in children and adults (Esposito & Lelii, 2015; Martineau et al., 2017). Vitamin D also exerts a broader immunomodulation function (Chirumbolo et al., 2017) and improves defences by suppressing anti-microbial peptides in response to vitamin D (Han et al., 2017).
The innate immune system is activated within two to three days of administration of high doses of vitamin D and protects from viral respiratory tract infection. Therefore, we hypothesised that high-dose vitamin D reduces the incidence, morbidity, and mortality from COVID-19 infection. Furthermore, with its known efficacy in reducing the risks of respiratory viral diseases, data support that this approach to control COVID-19 is highly cost- effective (Wimalawansa, 2020a). However, to achieve the intended benefits, serum 25(OH) D concentration be raised quickly to boost the innate immune system (Wimalawansa, 2020c), necessitating the administration of upfront higher doses.
Therefore, based on the need, we designed two randomised controltrials (RCTs) to evaluate cost-effective ways of preventing and treating persons with COVID 19. The prevention study was to be at selected quarantine centres in Sri Lanka, enrolling 2,000 subjects. This RCT was designed to assess whether using a single dose of vitamin D, 400,000 IU, would significantly reduce people getting infected with COVID-19 at the quarantine centres. This RCT would have been the first COVID prevention study in the world.
The second study was designed to evaluate the comparative efficacy of approved agents, vitamin D, hydroxychloroquine, and ivermectin in the early stages of COVID-19. In addition, clinical outcomes of the mentioned three active groups to compare with a group that received a placebo. Based on the Power Analysis, the goal was to recruit 900 PCR positive subjects per group (total n=3,600): at the “early” stage of the disease, admitted to the IDH hospital. It was the first and the largest COVID hospital in Sri Lanka.
Both studies had adequate statical power to achieve the primary endpoints with confidence. We sought ethical approval (ERC/ IRB) from the Sri Lanka Medical Association and the national medical regulatory authority (NMRA). Moreover, administrative permissions sought from the heath minister/ secretary, director of health, director at the IDH, and the director of the COVID task force to conduct the mentioned two RCTs. RCTs were self-funded, and there was no cost to the government or health department. To date, neither of these two RCTs formally approved by the administration. Sri Lanka missed an excellent opportunity to lead the global COVID clinical research to place it on the top of the research map.
By the end of 2020, these two complementary RCTs would have generated precious data for the world on cost-effective ways of preventing and treating COVID-19. Instead of interfering with progress and erecting barriers, governments and bureaucrats should have practical and sensible to facilitate essential clinical research in emergencies. The administrators’ theme, however, was how to block the progress and not how to facilitate it. Instead of raising barriers, administrators, including review boards (ERCs) and NMRA, should seek ways to facilitate clinical research in the future.
Latest break though in the prevention of complications from COVID-19
Most recent RCTs demonstrated that chemical formulation of 25(OH)D, calcifediol (a partially activated vitamin D), can be used orally at a dose between 0.5 and 1.0 mg, that raises the serum 25(OH)D concentration by four hours, instead of an average, three days (Castillo, 2020; Nogues X., 2021). This preparation can immediately overcome the few day delays in raising the blood 25(OH) D concentration with oral vitamin D. This is a significant advance that has already been a lifesaver in persons with COVID-19. Calcifediol is available as an over-the-counter medication at a reasonable cost in many counties, including the USA, the UK, Canada, and Australia.
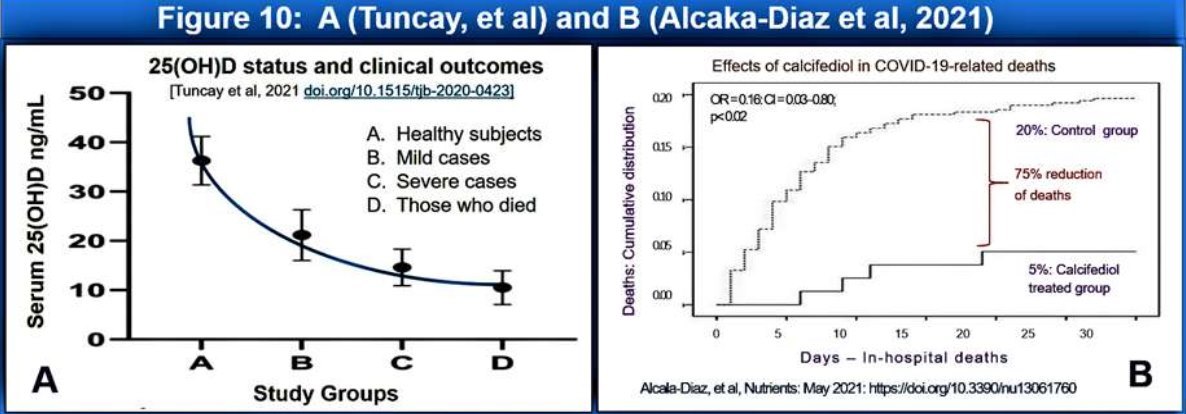
Calcifediol to be used when a person with PCR positive or suspected of infected with SARS.CoV-2 virus (or any other virus) admitted (i.e., at the earliest opportunity). Figure 10 illustrates the high efficacy of 0.523 mg of calcifediol in persons with moderate COVID-19 syndrome, preventing deaths (75% reduction of deaths than in the control group) (i.e., early and late interventions and prophylactic preventative use.
Calcifediol can be used in hospitalised patients at the time of admission or immediately after the diagnosis of SARS.Cov-2 virus, and in those with COVID-19 syndrome associate complications, as effectively demonstrated in two RCTs recently in Cordoba, Italy (Castillo, 2020) (Figure 10). Therefore, the single-dose calcifediol should be a standard (life-saving) therapy immediately after becoming PCR positive or with COVID-19 to minimise the risks of complications and deaths. This should be followed up with 200,000 IU of vitamin D (or 50,000 IU daily for four days) as a single dose or divided doses over four days to maintain the benefits of calcifediol.
CONCLUSIONS
Apart from avoiding exposure and reducing the viral load by using effective facemasks, the most critical strategies against COVID-19 syndrome is strengthening the immune system at the earliest possible. The first is achieved through adhering to public health measures (especially wearing protective face masks and avoid crowd gatherings), a balanced diet, physical activities, etc. A robust immune system can be achieved and maintained by taking high dose vitamin D and appropriate COVID vaccines.
Neither of these strategies requires expensive or expansion of expensive hospital equipment, protocols, quarantine centres, creating more ICU beds, task forces, or lockdowns and curfew. These expansions are needed when administrations failed to manage COVID properly. Lockdowns and curfews may temporarily reduce community exposure, as evident in Australia, New Zealand, and China, but at the expense of multiple adverse outcomes such as increased poverty, hunger, suicides, community deaths, spousal and child abuse, depression, other psychosocial factors.
This paper examined broader aspects of preventing COVID-19 using public health measures and approved cost-effective medications. Presented data overwhelmingly support and strengthen initial observations made in early 2020, that (A) those with vitamin D sufficiency rarely develop symptomatic disease or death, (B) vitamin D deficiency increases the risk of COVID-19, complications and deaths, (C) high dose vitamin D, significantly reduces the complications and deaths, and (D) 1.0 mg calcifediol is highly beneficial in preventing complications and deaths from COVID-19.
Inefficient immunity, secondary to vitamin D deficiency, causes a cascade of negative consequences following COVID-19. Meanwhile, there is no credible evidence to date that antiviral treatments, monoclonal antibodies, plasma therapy etc., have a significant effect in reducing complications and deaths. Therefore, we recommend an appropriate single dose of combined vitamin D and calcifediol in PCR positive persons or suspected of having COVID and prevent community spread of the pandemic. Furthermore, the significant impact of using vitamin D supplementation and calcifediol together with wearing facemasks is highly cost- effective in preventing the spread of COVID-19 and reduce the severity of SARS-CoV-2.
REFERENCES
Adams, J. S., Modlin, R. L., Diz, M. M., & Barnes, P. F. (1989). Potentiation of the macrophage 25-hydroxyvitamin D-1-hydroxylation reaction by human tuberculous pleural effusion fluid. J Clin Endocrinol Metab, 69(2), 457-460. doi:10.1210/jcem-69-2-457 Aldridge, R. W, Lewer, D., Katikireddi, S. V., Mathur, R., Pathak, N., Burns, R., . . . Hayward,(2020). Black, Asian and Minority Ethnic groups in England are at increased risk of death from COVID-19: indirect standardisation of NHS mortality data. Wellcome Open Res, 5, 88. doi:10.12688/wellcomeopenres.15922.2
AlSafar, H., Grant, W. B., Hijazi, R., Uddin, M., Alkaabi, N., Tay, G., . . . Al Anouti, F. (2021). COVID-19 Disease Severity and Death in Relation to Vitamin D Status among SARS- CoV-2-Positive UAE Residents. Nutrients, 13(5). doi:10.3390/nu13051714
Annweiler, G., Corvaisier, M., Gautier, J., Dubee, V., Legrand, E., Sacco, G., & Annweiler, C. (2020). Vitamin D Supplementation Associated to Better Survival in Hospitalised Frail Elderly COVID-19 Patients: The GERIA-COVID Quasi-Experimental Study. Nutrients, 12(11). doi:10.3390/nu12113377
Antal, A. S., Dombrowski, Y., Koglin, S., Ruzicka, T., & Schauber, J. (2011). Impact of vitamin D3 on cutaneous immunity and anti-microbial peptide expression. Dermatoendocrinol, 3(1), 18-22. doi:10.4161/derm.3.1.14616
Arya, A., & Dwivedi, V. D. (2020). Synergistic effect of vitamin D and remdesivir can fight COVID-19. J Biomol Struct Dyn, 1-2. doi:10.1 080/07391102.2020.1773929
Auler, A. C., Cassaro, F. A. M., da Silva, V. O., & Pires, F. (2020). evidence that high temperatures and intermediate relative humidity might favor the spread of COVID-19 in tropical climate: A case study for the most affected Brazilian cities. Sci Total Environ, 729, 139090. doi:10.1016/j. scitotenv.2020.139090
Bäcker, A. (2020). Slower COVID-19 morbidity and mortality growth at higher solar Iirradiance and elevation. Social Science Research Network, Rochester, NY. doi:10.2139/ssrn.3604729
Bäcker A, M. M. (2020). Double COVID-19 confirmed case fatality rate in countries with high elderly female vitamin D deficiency prevalence. Social Science Research Network, Rochester, NY. doi:10.2139/ssrn.3623662
Bishop, E., Ismailova, A., Dimeloe, S. K., Hewison, M., & White, J. H. (2020). Vitamin D and immune regulation: antibacterial, antiviral, anti-inflammatory. JBMR Plus. doi:10.1002/ jbm4.10405
Boucher, B. J. (2012). The problems of vitamin d insufficiency in older people. Aging Dis, 3(4), 313-329.
Braiman, M. (2020). Latitude Dependence of the COVID-19 Mortality Rate—A Possible Relationship to Vitamin D Deficiency? . Social Science Research Network. doi:10.2139/ ssrn.3561958
Brown, R. A. (2020). Preventing a covid-19 pandemic-COVID-19: Vitamin D deficiency; and, death rates; are both disproportionately higher in elderly Italians, Spanish, Swedish Somali, and African Americans? A connection. BMJ, Letter to the Editor.
Burt, L. A., Billington, E. O., Rose, M. S., Raymond, D. A., Hanley, D. A., & Boyd, S. K. (2019). Effect of High-Dose Vitamin D Supplementation on Volumetric Bone Density and Bone Strength: A Randomised Clinical Trial. JAMA, 322(8), 736745. doi:10.1001/jama.2019.11889
Cangiano, B., Fatti, L. M., Danesi, L., Gazzano, G., Croci, M., Vitale, G., . . . Bonomi, M. (2020). Mortality in an Italian nursing home during COVID-19 pandemic: correlation with gender, age, ADL, vitamin D supplementation, and limitations of the diagnostic tests. Aging (Albany NY), 12(24), 24522-24534. doi:10.18632/aging.202307
Cao, Z., Wu, Y., Faucon, E., & Sabatier, J. M. (2020). SARS-CoV-2 & Covid-19: Key-Roles of the ‘Renin-Angiotensin’ System / Vitamin D Impacting Drug and Vaccine Developments. Infect Disord Drug Targets, 20(3), 348-349. doi: 10.2174/1871526520999200505174704
Castillo, M. E., Costa, L.M.E, Barrios, J.M.V, Diaz, J.F.A, Miranda, J.L, Bouillon, R, Gomez, J.M.,Q. (2020). Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalised for COVID-19: A pilot randomised clinical study. J Steroid Biochem Mol Biol, 203(203), 105751, https://www.sciencedirect.com/science/article/ abs/pii/S0960076020302764/. doi:10.1016/j. jsbmb.2020.105751
Chaccour, C., Ruiz-Castillo, P., Richardson, M. A., Moncunill, G., Casellas, A., Carmona-Torre, F.,Rabinovich, R. (2020). The SARS-CoV-2 Ivermectin Navarra-ISGlobal Trial (SAINT) to Evaluate the Potential of Ivermectin to Reduce COVID-19 Transmission in low risk, nonsevere COVID-19 patients in the first 48 hours after symptoms onset: A structured summary of a study protocol for a randomised control pilot trial. Trials, 21(1), 498. doi:10.1186/ s13063-020-04421-z
Chirumbolo, S., Bjorklund, G., Sboarina, A., & Vella, A. (2017). The Role of Vitamin D in the Immune System as a Pro-survival Molecule. Clin Ther, 39(5), 894-916. doi:10.1016/j. clinthera.2017.03.021
Christakos, S., Dhawan, P, Verstuyf, A., Verlinden, , & Carmeliet, G. (2016). Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol Rev, 96(1), 365408. doi:10.1152/physrev.00014.2015
Chung, C., Silwal, P, Kim, I., Modlin, R. L., & Jo, E. K. (2020). Vitamin D-Cathelicidin Axis: at the Crossroads between Protective Immunity and Pathological Inflammation during Infection. Immune Netw, 20(2), e12. doi:10.4110/ in.2020.20.e12
Conticini, E., Frediani, B., & Caro, D. (2020). Can atmospheric pollution be considered a cofactor in extremely high level of SARS-CoV-2 lethality in Northern Italy? Environ Pollut, 261, 114465. doi:10.1016/j.envpol.2020.114465
Crane-Godreau, M. A., Clem, K. J., Payne, P, & Fiering, S. (2020). Vitamin D Deficiency and Air Pollution Exacerbate COVID-19 Through Suppression of Antiviral Peptide LL37. Front Public Health, 8, 232. doi:10.3389/ fpubh.2020.00232
Ebadi, M., & Montano-Loza, A. J. (2020). Perspective: improving vitamin D status in the management of COVID-19. Eur J Clin Nutr, 74(6), 856-859. doi:10.1038/s41430-020-0661-0
El Zowalaty, M. E., & Jarhult, J. D. (2020). From SARS to COVID-19: A previously unknown SARS- related coronavirus (SARS-CoV-2) of pandemic potential infecting humans - Call for a One Health approach. One Health, 9, 100124. doi:10.1016/j.onehlt.2020.100124
Esposito, S., & Lelii, M. (2015). Vitamin D and respiratory tract infections in childhood. BMC Infect Dis, 15, 487. doi:10.1186/s12879-015- 1196-1
Fabbri, A., Infante, M., & Ricordi, C. (2020). Editorial - Vitamin D status: a key modulator of innate immunity and natural defense from acute viral respiratory infections. Eur Rev Med Pharmacol Sci, 24(7), 4048-4052. doi:10.26355/ eurrev20200420876
French, C. B., McDonnell, S. L., & Vieth, R. (2019). 25-Hydroxyvitamin D variability within- person due to diurnal rhythm and illness: a case report. J Med Case Rep, 13(1), 29. doi:10.1186/ s13256-018-1948-9
Garg, M., Al-Ani, A., Mitchell, H., Hendy, P, & Christensen, B. (2020). Low population mortality from COVID-19 in countries south of latitude 35 degrees North-supports vitamin D as a factor determining severity. Authors’ reply. Aliment Pharmacol Ther, 51(12), 14381439. doi:10.1111/apt.15796
Gasmi, A., Noor, S., Tippairote, T., Dadar, M., Menzel, A., & Bjorklund, G. (2020). Individual risk management strategy and potential therapeutic options for the COVID-19 pandemic. Clin Immunol, 215, 108409. doi:10.1016/j.clim.2020.108409
Ghasemian, R., Amir Shamshirian, Heydari, K, Malekan, M, Alizadeh-Navaei, R, Ali Ebrahimzadeh, M, Jafarpour, H, Rezaei Shahmirzadi, A, Khodabandeh, M, Seyfari, B, Sedaghat, M, Motamedzadeh, A, Dadgostar, E, Aalinezhad, M, Behnamfar, M, Asadi, A, Zarandi, B, Razzaghi, N, Yaghoubi Naei, V, Hessami, A, Azizi, S, Mohseni, AR, Shamshirian, D. (2020). The role of vitamin D in the age of COVID-19: A systematic review and meta-analysis along with an eological approach. medRxiv, (Preprint).
Grant, W. B., Al Anouti, F., & Moukayed, M. (2020). Targeted 25-hydroxyvitamin D concentration measurements and vitamin D3 supplementation can have important patient and public health benefits. Eur J Clin Nutr, 74(3), 366-376. doi:10.1038/s41430-020-0564- 0
Grant, W. B., Lahore, H., McDonnell, S. L., Baggerly, C. A., French, C. B., Aliano, J. L., & Bhattoa, H. P. (2020). Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients, 12(6), 1626. doi:10.3390/ nu12040988
Grant, W. B., Wimalawansa, S. J., Holick, M. F., Cannell, J. J., Pludowski, P, Lappe, J. M., . . . May, P. (2015). Emphasising the health benefits of vitamin D for those with neurodevelopmental disorders and intellectual disabilities. Nutrients, 7(3), 1538-1564. doi:10.3390/nu7031538 Grasselli, G., Zangrillo, A., Zanella, A., Antonelli,
M., Cabrini, L., Castelli, A., . . . Viola, U. (2020). Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA. doi:10.1001/jama.2020.5394
Guo, L., Ren, L., Yang, S., Xiao, M., Chang, Yang, F., . . . Wang, J. (2020). Profiling Early Humoral Response to Diagnose Novel Coronavirus Disease (COVID-19). Clin Infect Dis, 71(15), 778-785. doi:10.1093/cid/ciaa310
Han, J. E., Alvarez, J. A., Jones, J. L., Tangpricha, V., Brown, M. A., Hao, L., . . . Ziegler, T. R. (2017). Impact of high-dose vitamin D3 on plasma free 25-hydroxyvitamin D concentrations and antimicrobial peptides in critically ill mechanically ventilated adults. Nutrition, 38, 102-108.
doi:10.1016/j.nut.2017.02.002 Haq, A., Wimalawansa, S. J., Pludowski, P, & Anouti, F. A. (2018). Clinical practice guidelines for vitamin D in the United Arab Emirates. J Steroid Biochem Mol Biol, 175, 4-11. doi:10.1016/j.jsbmb.2016.09.021 Hastie, C. E., Mackay, D. F., Ho, F., Celis-Morales, C. A., Katikireddi, S. V., Niedzwiedz, C. L., . . . Pell, J. P. (2020). Vitamin D concentrations and COVID-19 infection in UK Biobank. Diabetes Metab Syndr, 14(4), 561-565. doi:10.1016/j. dsx.2020.04.050
Hastie, C. E., Pell, J. P., & Sattar, N. (2020). Vitamin D and COVID-19 infection and mortality in UK Biobank. Eur J Nutr. doi:10.1007/s00394- 020-02372-4
Holick, M. F. (2009). Vitamin D status: measurement, interpretation, and clinical application. Ann Epidemiol, 19(2), 73-78. doi:10.1016/j. annepidem.2007.12.001
Holmes, L., Jr., Enwere, M., Williams, J., Ogundele,, Chavan, P, Piccoli, T., . . . Dabney, K. W. (2020). Black-White Risk Differentials in COVID-19 (SARS-COV2) Transmission, Mortality and Case Fatality in the United States: Translational Epidemiologic Perspective and Challenges. Int J Environ Res Public Health, 17(12). doi:10.3390/ijerph17124322
Hribar, C. A., Cobbold, P H., & Church, F. C. (2020). Potential Role of Vitamin D in the Elderly to Resist COVID-19 and to Slow Progression of Parkinson’s Disease. Brain Sci, 10(5). doi:10.3390/brainsci10050284
Hunter, J., Arentz, S., Goldenberg, J., Yang, G., Beardsley, J., Mertz, D., & Leeder, S. (2020). Rapid review protocol: Zinc for the prevention or treatment of COVID-19 and other coronavirus-related respiratory tract infections. Integr Med Res, 9(3), 100457. doi:10.1016/j. imr.2020.100457
Ilie, P C., Stefanescu, S., & Smith, L. (2020). The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin Exp Res, 32(7), 1195-1198. doi:10.1007/ s40520-020-01570-8
Kahlenberg, J. M., & Kaplan, M. J. (2013). Little peptide, big effects: the role of LL-37 in inflammation and autoimmune disease. J Immunol, 191(10), 4895-4901. doi:10.4049/ jimmunol.1302005
Kakodkar, P, Kaka, N., & Baig, M. N. (2020). A Comprehensive Literature Review on the Clinical Presentation, and Management of the Pandemic Coronavirus Disease 2019 (COVID-19). Cureus, 12(4), e7560. doi:10.7759/cureus.7560
Kaufman, H. W, Niles, J. K., Kroll, M. H., Bi, , & Holick, M. F. (2020). SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels. PLoS One, 15(9), e0239252. doi:10.1371/journal.pone.0239252
Kennel, K. A., Drake, M. T., & Hurley, D. L. (2010). Vitamin D deficiency in adults: when to test and how to treat. Mayo Clin Proc, 85(8), 752757; quiz 757-758. doi:85/8/752 [pii] 10.4065/mcp.2010.0138
Kimball, S. M., Mirhosseini, N., & Holick, M. F. (2017). Evaluation of vitamin D3 intakes up to 15,000 international units/day and serum 25-hydroxyvitamin D concentrations up to 300 nmol/L on calcium metabolism in a community setting. Dermato-Endocrinology, 9(1), e1300213-e1300213. doi:10.1080/193819 80.2017.1300213
Kumar, R., Rathi, H., Haq, A, Wimalawansa, S.J, Sharma, A. (2021). Putative roles of vitamin D in modulating immune response and immunopathology associated with COVID-19. Virus Res, 292, 198235. doi:10.1016/j.virusres.2020.198235
Lim, K., & Thadhani, R. (2020). Vitamin D Toxicity. J Bras Nefrol, 42(2), 238-244. doi:10.1590/2175- 8239-JBN-2019-0192
Loke, H., Bethell, D., Phuong, C. X., Day, N., White, N., Farrar, J., & Hill, A. (2002). Susceptibility to dengue hemorrhagic fever in vietnam: evidence of an association with variation in the vitamin d receptor and Fc gamma receptor IIa genes. Am J Trop Med Hyg 67(1), 102-106. doi:10.4269/ajtmh.2002.67.102
Lowe, H., Cusano, N. E., Binkley, N., Blaner, W. S., & Bilezikian, J. P (2011). Vitamin D toxicity due to a commonly available “over the counter” remedy from the Dominican Republic. J Clin Endocrinol Metab, 96(2), 291-295. doi:10.1210/ jc.2010-1999
Maghbooli, Z., Sahraian, M. A., Ebrahimi, M., Pazoki, M., Kafan, S., Tabriz, H. M., . . . Holick, M. F. (2020). Vitamin D sufficiency, a serum 25-hydroxyvitamin D at least 30 ng/ mL reduced risk for adverse clinical outcomes in patients with COVID-19 infection. PLoS One, 15(9), e0239799. doi:10.1371/journal. pone.0239799
Marcinowska-Suchowierska, E., Kupisz-Urbanska, M., Lukaszkiewicz, J., Pludowski, P, & Jones, G. (2018). Vitamin D Toxicity-A Clinical Perspective. Front Endocrinol (Lausanne), 9, 550. doi:10.3389/fendo.2018.00550
Martelletti, L., & Martelletti, P. (2020). Air Pollution and the Novel Covid-19 Disease: a Putative Disease Risk Factor. SN Compr Clin Med, 1-5. doi:10.1007/s42399-020-00274-4
Martineau, A. R., Hanifa, Y., Witt, K. D., Barnes, N. C., Hooper, R. L., Patel, M., . . . Griffiths, C. J. (2015). Double-blind randomised controlled trial of vitamin D3 supplementation for the prevention of acute respiratory infection in older adults and their carers (ViDiFlu). Thorax, 70(10), 953-960. doi:10.1136/ thoraxjnl-2015-206996
Martineau, A. R., Jolliffe, D. A., Hooper, R. L., Greenberg, L., Aloia, J. F., Bergman, P., . . . Camargo, C. A., Jr. (2017). Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and metaanalysis of individual participant data. BMJ, 356, i6583. doi:10.1136/bmj.i6583
McGregor, E., Kazemian M, Afzali, B, et al. (2020). An autocrine Vitamin D-driven Th1 shutdown program can be exploited for COVID-19 https://www.biorxiv.org/ content/10.1101/2020.07.18.210161v1.
Mecenas, P, Bastos, R., Vallinoto, A. C. R., & Normando, D. (2020). Effects of temperature and humidity on the spread of COVID-19: A systematic review. PLoS One, 15(9), e0238339. doi:10.1371/journal.pone.0238339
Meltzer, D. O., Best, T. J., Zhang, H., Vokes, T., Arora, V., & Solway, J. (2020). Association of Vitamin D Deficiency and Treatment with COVID-19 Incidence. medRxiv.doi:10.1101/2020.05.08.20095893
Merzon, E., Tworowski, D., Gorohovski, A., Vinker, S., Golan Cohen, A., Green, I., & Frenkel Morgenstern, M. (2020). Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: an Israeli population- based study. FEBS J. doi:10.1111/febs.15495
Meschia, J. F., Barrett, K. M., Brown, R. D., Jr., Turan, T. N., Howard, V. J., Voeks, J. H., . . . Brott, T. G. (2020). The CREST-2 experience with the evolving challenges of COVID-19: A clinical trial in a pandemic. Neurology, 95(1), 29-36. doi:10.1212/WNL.0000000000009698
Molloy, E. J., & Murphy, N. (2020). Vitamin D, Covid-19 and Children. Ir Med J, 113(4), 64.
Neher, R. A., Dyrdak, R., Druelle, V., Hodcroft, E. B., & Albert, J. (2020). Potential impact of seasonal forcing on a SARS-CoV-2 pandemic. Swiss Med Wkly, 150, w20224. doi:10.4414/ smw.2020.20224
Nogues X., O. D., Quesada-Gomez J.M., Bouillon R. (2021). Calcifediol treatment and COVID-19- related outcomes.]http://dx.doi.org/10.2139/
ssrn.3771318.
Okan, F., Okan, S., & Zincir, H. (2020). Effect of Sunlight Exposure on Vitamin D Status of Individuals Living in a Nursing Home and Their Own Homes. J Clin Densitom, 23(1), 2128. doi:10.1016/j.jocd.2018.12.005
Osmancevic, A., Demeke, T., Gillstedt, M., Angesjo, E., Sinclair, H., Abd El-Gawad, G., & Landin- Wilhelmsen, K. (2016). Vitamin D treatment in Somali women living in Sweden-Two randomised, placebo-controlled studies. Clin Endocrinol (Oxf), 85(4), 535-543. doi:10.1111/ cen.13097
Panagiotou, G., Tee, S. A., Ihsan, Y., Athar, W, Marchitelli, G., Kelly, D., . . . Quinton, R. (2020). Low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalised with COVID-19 are associated with greater disease severity. Clin Endocrinol (Oxf). doi:10.1111/cen.14276
Pludowski, P., Holick, M. F., Grant, W. B., Konstantynowicz, J., Mascarenhas, M. R., Haq, A., . . . Wimalawansa, S. J. (2018). Vitamin D supplementation guidelines. J Steroid Biochem Mol Biol, 175, 125-135. doi:10.1016/j. jsbmb.2017.01.021
Quraishi, S. A., Bittner, E. A., Blum, L., McCarthy, C. M., Bhan, I., & Camargo, C. A., Jr. (2014). Prospective study of vitamin D status at initiation of care in critically ill surgical patients and risk of 90-day mortality. Crit Care Med, 42(6), 1365-1371. doi:10.1097/ CCM.0000000000000210
Quraishi, S. A., De Pascale, G., Needleman, J. S., Nakazawa, H., Kaneki, M., Bajwa, E. K., . .
. Bhan, I. (2015). Effect of Cholecalciferol Supplementation on Vitamin D Status and Cathelicidin Levels in Sepsis: A Randomised, Placebo-Controlled Trial. Crit Care Med, 43(9), 1928-1937. doi:10.1097/ CCM.0000000000001148
Radujkovic, A., Hippchen, T., Tiwari-Heckler, S., Dreher, S., Boxberger, M., & Merle, U. (2020). Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients, 12(9). doi:10.3390/nu12092757
Raisi-Estabragh, Z., McCracken, C., Bethell, M. S., Cooper, J., Cooper, C., Caulfield, M. J., . . Petersen, S. E. (2020). Greater risk of severe COVID-19 in Black, Asian and Minority Ethnic populations is not explained by cardiometabolic, socioeconomic or behavioural factors, or by 25(OH)-vitamin D status: study of 1326 cases from the UK Biobank. J Public Health (Oxf). doi:10.1093/pubmed/fdaa095
Rastogi, A., Bhansali, A., Khare, N., Suri, V., Yaddanapudi, N., Sachdeva, N., . . . Malhotra, P. (2020). Short term, high-dose vitamin D supplementation for COVID-19 disease: a randomised, placebo-controlled, study (SHADE study). Postgrad Med J, https://pmj. bmj.com/content/early/2020/2011/2012/ postgradmedj-2020-139065 doi:10.1136/ postgradmedj-2020-139065
Rhodes, J. M., Subramanian, S., Laird, E., & Kenny, R. A. (2020). Editorial: low population mortality from COVID-19 in countries south of latitude 35 degrees North supports vitamin D as a factor determining severity. Aliment Pharmacol Ther, 51(12), 1434-1437. doi:10.1111/apt.15777
Saaf, M., Fernell, E., Kristiansson, F., Barnevik Olsson, M., Gustafsson, S. A., & Bagenholm, G. (2011). Severe vitamin D deficiency in pregnant women of Somali origin living in Sweden. Acta Paediatr, 100(4), 612-614. doi:10.1111/j.1651- 2227.2011.02134.x
Sabetta, J. R., DePetrillo, P., Cipriani, R. J., Smardin, J., Burns, L. A., & Landry, M. L. (2010). Serum 25-hydroxyvitamin d and the incidence of acute viral respiratory tract infections in healthy adults. PLoS One, 5(6), e11088. doi:10.1371/ journal.pone.0011088
Shiravi, A. A., Saadatkish, M., Abdollahi, Z., Miar, P., Khanahmad, H., & Zeinalian, M. (2020). Vitamin D can be effective on the prevention of COVID-19 complications: A narrative review on molecular aspects. Int J Vitam Nutr Res, 1-13. doi:10.1024/0300-9831/a000676
Spiller, H. A., Good, T. F., Spiller, N. E., & Aleguas, A. (2016). Vitamin D exposures reported to US poison centers 2000-2014: Temporal trends and outcomes. Hum Exp Toxicol, 35(5), 457461. doi:10.1177/0960327115595685
Stohs, S. J., & Aruoma, O. I. (2020). Vitamin D and Wellbeing beyond Infections: COVID-19 and Future Pandemics. J Am Coll Nutr, 1-2. doi:10.1 080/07315724.2020.1786302
Story, M. J. (2021). Essential sufficiency of zinc, omega-3 polyunsaturated fatty acids, vitamin D and magnesium for prevention and treatment of COVID-19, diabetes, cardiovascular diseases, lung diseases and cancer. Biochimie. doi:10.1016/j.biochi.2021.05.013
Swami, S., Krishnan, A. V., & Feldman, D. (2011). Vitamin D metabolism and action in the prostate: Implicationsforhealthand disease. Mol Cell Endocrinol. doi:S0303-7207(11)00251-6 [pii] 10.1016/j.mce.2011.05.010
Tan, C. W, Ho, L. P., Kalimuddin, S., Cherng, B. P. Z., Teh, Y. E., Thien, S. Y., . . . Ng, H. J. (2020). Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B12 in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrition, 79-80, 111017. doi:10.1016/j. nut.2020.111017
Temgoua, M. N., Kuate, L. M., Ngatchou, W., Sibetcheu, A., Toupendi, Z. N., Belobo, G., . . . Kingue, S. (2020). COVID-19 pandemic: do we need systematic screening of patients with cardiovascular risk factors in Low and Middle- Income Countries (LMICs) for preventing death? Pan Afr Med J, 35(Suppl 2), 11. doi:10.11604/pamj.2020.35.2.22947
Trivedy, C., Mills, I., & Dhanoya, O. (2020). The impact of the risk of COVID-19 on Black, Asian and Minority Ethnic (BAME) members of the UK dental profession. Br Dent J, 228(12), 919-922. doi:10.1038/s41415-020-1781-6
Vassiliou, A. G., Jahaj, E., Pratikaki, M., Orfanos, S. E., Dimopoulou, I., & Kotanidou, A. (2020). Low 25-Hydroxyvitamin D Levels on Admission to the Intensive Care Unit May Predispose COVID-19 Pneumonia Patients to a Higher 28-Day Mortality Risk: A Pilot Study on a Greek ICU Cohort. Nutrients, 12(12). doi:10.3390/nu12123773
Villamor, E., Villar, L. A., Lozano, A., Herrera, V. M., & Herran, O. F. (2017). Vitamin D serostatus and dengue fever progression to dengue hemorrhagic fever/dengue shock syndrome. Epidemiol Infect, 145(14), 29612970. doi:10.1017/S0950268817002059
Wang J, T. K., Feng K, X Lin, W Lv, K Chen, L Wang. (2020). High temperature and high humidity reduce the transmission of COVID-19. SSRN Electron J, 19(13), 1-13. doi:10.2139/ ssrn.3551767
Wang, Z., & Wang, G. (2004). APD: the Antimicrobial Peptide Database. Nucleic Acids Res, 32(Database issue), D590-592. doi:10.1093/nar/gkh025
Wimalawansa, S. J. (2012). Vitamin D in the new millennium. Curr Osteoporos Rep, 10(1), 4-15. doi:10.1007/s11914-011-0094-8 Wimalawansa, S. J. (2020a). Achieving population vitamin D sufficiency will markedly reduce healthcare costs. EJBPS, 7:3 136-141.
Wimalawansa, S. J. (2020b). Commonsense approaches to minimising risks from COVID-19. Open J Pulm Respir Med, 2, 28-37, https://doi.org/10.36811/ojprm.32020.110010 doi:10.36811/ojprm.2020.110010
Wimalawansa, S. J. (2020c). COVID-19: Evolution and prevention. Trends Telemed E-Health, 2(3), 1-5. doi:10.31031/TTEH.2020.02.000539
Wimalawansa, S. J. (2020d). Global epidemic of coronavirus—COVID-19: What can we do to minimise risks? European J. Biomed & Pharma Sci., 7(3), 432-438. doi:https://storage. googleapis.com/journal-uploads/ejbps/article_ issue/volume7marchissue3/1584436192. pdf
Wimalawansa, S. J. (2020e). Modes of preventing secondary community spread and future epidemics of COVID-19. J. Immuno Biol, 5(1), 1-5. doi:10.37421/JIB.2020.5.147 Wimalawansa, S. J. (2020f). Reducing risks from COVID-19: Cost-effective ways of strengthening individual’s and the population immunity with vitamin D. J Endocrinol Sci, 2(2), -13.
Wimalawansa, S. J. (2020c). Fighting against COVID-19: Boosting the immunity with micronutrients, stress reduction, physical activity, and vitamin D. Nutrition and Food Science Journal (Sci Literature), 3(1)(126), 1-4.
Wimalawansa, S. J., Razzaque, D. M.S, Al-Daghri, N.M. (2017). Calcium and Vitamin D in Human Health: Hype or Real? J Steroid Biochem Mol Biol, 180, 4-14. doi:10.1016/j. jsbmb.2017.12.009
Xu J, Y. J., Chen J, Luo Q, Zhang Q, Zhang H. (2017). Vitamin D alleviates lipopolysaccharide- induced acute lung injury via regulation of the renin-angiotensin system. Mol Med Rep, 16, 7432-7438
Yadaver, S. (2020). 57% of Mumbai slum dwellers were exposed to Covid, developed antibodies, finds sero survey. Retrieved from https:// theprint.in/health/57-of-mumbai-slum- dwellers-were-exposed-to-covid-developed- antibodies-finds-sero-survey/470120/
Yousef, S., Elliott, J., Manuel, D., Colman, I., Papadimitropoulos, M., Hossain, A., . . . Wells, G. A. (2019). Study protocol: Worldwide comparison of vitamin D status of immigrants from different ethnic origins and native-born populations-a systematic review and metaanalysis. Syst Rev, 8(1), 211. doi:10.1186/ s13643-019-1123-4
Zemb, P., Bergman, P., Camargo, C. A., Jr., Cavalier, E., Cormier, C., Courbebaisse, M., . . . Souberbielle, J. C. (2020). Vitamin D deficiency and the COVID-19 pandemic. J Glob Antimicrob Resist, 22, 133-134. doi:10.1016/j. jgar.2020.05.006
Zhu, Y., Xie, J., Huang, F., & Cao, L. (2020). Association between short-term exposure to air pollution and COVID-19 infection: Evidence from China. Sci Total Environ, 727, 138704. doi:10.1016/j.scitotenv.2020.138704
