Basketball players low on vitamin D
Low levels of vitamin D in professional basketball players after wintertime: relationship with dietary intake of vitamin D and calcium
Note: PDF is attached - text extracted below with just a single image from the PDF
See also VitaminDWiki
Suggests that college athletes supplement with vitamin D in winter - June 2010
Young Israeli athletes and dancers had just 25 ng of vitamin D – Sept 2010
Nutriciorv Hospitalaria, 2011;26(S):94S-9S1 ISSN 0212-1611 • CODEN NUHOEQ S.VJJ.318
R. Bescos Garcia12 (raul.bescos@inefc.net ) and F. A. Rodriguez Guisado
'National Institute of Physical Exercise of Barcelona. Sport Sciences Research Group. 'Nutritional service of DKV Joventut of Badalona. Spain.
Introduction: Although vitamin D deficiency has a high worldwide prevalence among the general population, very little is known about vitamin status in athletes.
Aim: To investigate serum vitamin D (25[OH]D) levels after wintertime in male elite basketball players, and to relate these levels to the dietary intake of vitamin D and calcium.
Methods: Subjects were 21 players from the same professional Spanish team. Blood samples to assess 25(OH)D levels were collected after wintertime during the 2008/2009 (April) and 2009/2010 (March) seasons. In addition, athletes completed 4-day dietary records to estimate energy consumption and a food frequency questionnaire to determine dietary vitamin D and calcium intake. Serum 25(OH)D levels were 47.8 ± 21.8 nmol/L, with twelve subjects (57%) being vitamin D deficient (< 50 nmol/L).
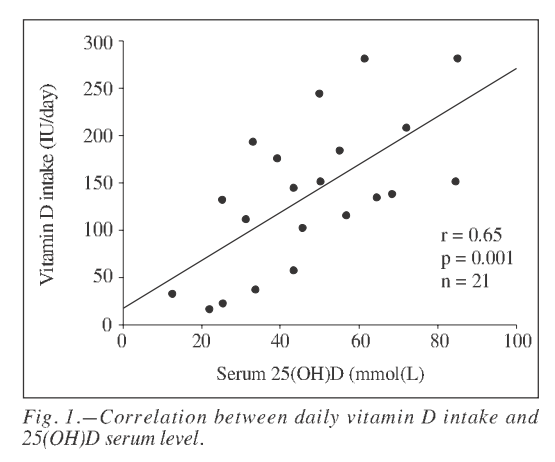
Results: Vitamin D intake was 139 ± 78 IU/day and calcium intake was 948 ± 419 mg/day. Serum 25(OH)D levels correlated with the daily dietary intake of vitamin D (r = 0.65; P = 0.001) and calcium (r = 0.82; P < 0.001).
Conclusion: Professional basketball players are at higher risk of hypovitaminosis D after wintertime. Adequate intake of dietary calcium and vitamin D is required if athletes are to avoid low serum 25(OH)D levels when exposure to sunlight is limited.
Introduction
Unlike most other vitamins, activated vitamin D is a secosteroid hormone rather than a cofactor in an enzymatic reaction or an antioxidant.1 Although hypovita-minosis D can affect an athlete's overall health and ability to train (i.e. affecting bone health, innate immunity, and exercise innate immunity and inflammation)23 this micronutrient has, for many years, been largely ignored in sports nutrition. Indeed, clinicians and nutritionists have generally not focused their attention on vitamin D status, possibly because it is assumed that exposure to sunlight is sufficient to maintain adequate plasma levels of vitamin D. However, any factor (such as regular sunscreen use > 15 sun protection factor, clothing, atmospheric pollution, skin pigmentation, latitude > 35° N or S, or time of the day) that limits the amount or quality of sun exposure can compromise serum vitamin D levels.14 To avoid deficiency when skin production is limited, humans are also able to obtain vitamin D from their diet in the form of vitamin D, (ergocalciferol) or D3 (cholecalciferol), although few available foods, whether natural or fortified, contain this micronutrient.1
Recent studies have reported that hypovitaminosis D is common among the general population15 and vitamin D deficiency may also be a problem among athletes, especially in those who train and compete in indoor sport.23 However, only two studies to date have analysed this question in indoor athletes.67 Both assessed vitamin D status in young female gymnasts and found that a lower concentration of serum vitamin D may be common after wintertime.6 7 However, it is known that many gymnasts have a low energy and nutrient intake in their diet and fail to reach the recom-mendable amount of dietary vitamins and minerals such as vitamin D.8 To the best of our knowledge no other studies have analysed vitamin D status in any other indoor discipline at the end of winter, this being the time when the risk of hypovitaminosis D is higher. In this regard, basketball players are a particularly interesting athletic population to study because the sport is an indoor discipline and exposure to sunlight may be limited during the competitive season (autumn and wintertime). In addition, and in contrast to gymnasts, basketball players presumably have a higher energy diet,9 although research has yet to analyse their intake of dietary vitamin D and calcium, or the relationship of this with serum vitamin D.
At all events it should be remembered that calcium and vitamin D metabolism are connected. Vitamin D plays an important role in the intestinal absorption of calcium by mediating active calcium transport across the intestinal mucosa.5 Moreover, serum levels of vitamin D have been directly related to bone density in both white and black men and women, with maximum density being achieved when the 25-hydroxyvitamin D (25[OH]D) level reaches 100 nmol/L.1 However, there is a lack of research on the relationship between vitamin D serum levels and dietary intake of vitamin D and calcium in athletes.
Given the above, the main aim of this study was to investigate vitamin D status in professional male basketball players after wintertime. A secondary aim was to analyse the relationship between serum vitamin D levels and the dietary intake of calcium and vitamin D. We hypothesised that these athletes would have lower levels of vitamin D during late winter due to the indoor characteristics of their sport. Additionally, although basketball players presumably have a higher energy diet, their consumption of food that is rich in vitamin D, such as fatty fish, may be lower, which would also affect serum levels of vitamin D.
Materials and methods
Subjects
Subjects were 21 male elite basketball players who were enrolled in the same professional Spanish team during the 2008/2009 and 2009/2010 seasons. Thirteen athletes were assessed during the first season, while the remaining eight were evaluated during the second. Five players formed part of the team during both seasons and were thus assessed twice. The sample was multinational, with subjects coming from nine countries. Sixteen athletes were White Caucasian and five players were African American. Somatic characteristics are summarised in table I. During the basketball season, from late August to May, all the players lived in the metropolitan area of Barcelona (41°N, 2°E). The number of sunny hours per month between October and April was 134.7 in the first season (2008/2009) and 124.1 in the second season (2009/2010). Athletic regimes included six to ten weekly training sessions (physical, technical and tactical), plus one or two games per week corresponding to a typical top-level competitive European season: Spanish professional league, the Spanish King's Cup tournament, and the European competition. These training sessions or competitive matches always took place indoors.
Table I
The study was reviewed and approved by the local ethics committee and all subjects gave their informed consent prior to the study. Furthermore, they were all free of medical conditions that could affect vitamin D or calcium status, as determined by a pre-season medical examination, and no relevant clinical symptoms were apparent at the time of the study. No subject was taking medication such as anticonvulsants, glucocorticoids or barbiturates which might also affect vitamin D metabolism.
Vitamin D status
Venous blood samples were drawn during the second week of April 2008 and the third week of March 2009, following a day of rest in both cases. All blood samples (20 mL) were collected in sterile chilled tubes and were allowed to clot at room temperature before being centrifuged at 3,000 rpm for 10minat4°C. The serum samples for 25(OH)D were protected from light during processing and were measured by a competitive two-step chemiluminescence assay method (Liasion® 25-OH Vitamin D Total 310600, Stillwater, USA). Method specificity for vitamins D2 and D3 was 104% and 100%, respectively. The intra-assay coefficient of variation (CV) was 5%, while the inter-assay CV was 8%.
Following the classification of Lips et al.4 three levels of vitamin D deficiency were considered: severe deficiency (< 12.5 mmol/L), moderate deficiency (12.5-25 mmol/L), and mild deficiency or insufficiency (25-50 mmol/L). Values above 50 nmol/L were considered to indicate a vitamin D replete state, i.e. sufficient.124 Values between 75 and 150 nmol/L were considered as the preferred (optimal) values for bone and general health.12510 Finally, serum 25(OH)D over 375 nmol/L was regarded as indicating vitamin D intoxication.1
Nutrient intake assessment
Subjects completed, non-consecutively, 4-day dietary records to estimate energy intake. A food frequency questionnaire was used to analyse dietary intake of calcium and vitamin D. The validity and reliability of both types of dietary review has been previously reported." 12 Players were given verbal and written instructions, including pictures of serving sizes for completion of their dietary records, which had to include all foods and beverages consumed, including vitamin and mineral supplements and products fortified with vitamin D and/or calcium. Subjects were also asked to document how their food was prepared (e.g. boiled, fried, etc.), the time of day at which they ate, and their use of condiments. Nutritional data were analysed by a trained nutritionist using a food composition database (CES-NID, University of Barcelona, Spain).
Statistical analysis
Descriptive data are presented as individual results and standard deviations (± SD). Spearman's rank correlations were used to analyse the relationship between 25(OH)D serum levels and dietary intake of energy, calcium and vitamin D. In addition, the Wilcoxon signed-rank test was applied to compare values of 25(OH)D in five subjects who were assessed twice. Non-parametric methods were used as not all parameters were normally distributed. The significance level was set at P< 0.05.
Results
Serum 25(OH)D Mean and individual results are shown in table II. Twelve subjects (57%) were vitamin D deficient (serum 25(OH)D < 50 nmol/L): two athletes had moderate 25(OH)D deficiency (12.5-25 nmol/L), while ten subjects showed mild deficiency (25-50 nmol/L). The remaining nine players (43%) showed sufficient values (> 50 nmol/L). Only two subjects had a value within the preferred or optimal range (75-150 nmol/L). Mean values of Caucasian subjects were 55.3 ± 16.5 nmol/L, with seven athletes showing mild deficiency. Black players had lower serum 25(OH)D levels (23.7 ± 7.5 nmol/L), with all of them being between moderate (three athletes) and mild deficiency (two athletes). In the subjects who were assessed twice (both seasons), no differences in serum 25(OH)D levels were observed across the two evaluations (table III).
Dietary analysis
Individual and mean daily intake of energy, vitamin D and calcium are summarised in table II. Despite consuming high-energy diets (17,936 ± 2,935 kj/day; 4,284 ± 701 kcal/day), the subjects' vitamin D intake (139 ± 78 IU/day) was below the daily recommended intake for young adults (200 IU/day).13 Individual vitamin D intake values showed that four subjects consumed less than 40 IU/day, and only four players consumed more than 200 IU/day. Interestingly, these players reported a frequent weekly consumption (1-3 times per week) of food rich in vitamin D (fatty fish). Other sources of vitamin D were milk and derived products such as yogurt or cheese, but no subjects consumed foods that were fortified with vitamin D.
Table II
Table III
Values of serum 25(OH)D in players who formed part of the team during both seasons and were assessed twice
(fig. 1)
~~#00F:Notes:
X and Y axis are reversed from normal.
Most had less than 30 ng/ml (75 nmol),
Less than 1,000 IU helps~~
Discussion
The aim of this study was to investigate serum 25(OH)D levels in professional basketball players living in Barcelona and to examine the relationship with dietary energy and the intake of calcium and vitamin D. In agreement with our first hypothesis the results confirm that professional basketball players are at higher risk of hypovitaminosis D (serum 25(OH)D < 50 nmol/L) after wintertime, despite living in a sunny location. Moreover, those athletes who ingested higher amounts of dietary calcium and vitamin D showed high serum 25(OH)D levels independently of dietary energy, thus indicating the importance of consuming an adequate amount of these micronutrients when endogenous production of vitamin D is limited.
Vitamin D status
The current research found mean serum 25(OH)D values of 47.8 ±21.8 nmol/L. Comparison with previous studies of indoor sports shows that the present results are above the mean 25(OH)D levels (33.9 nmol/L) reported by Lehtonen-Veromaa et al.7 in a young group of Finnish female athletes (gymnasts and runners) living in a northern location (60° N) and assessed in midwinter (February-March). By contrast, Lovell et al.6 found substantially higher mean values (56 nmol/L) in eighteen adolescent Australian female gymnasts living in Canberra (Australia) at a latitude 35.3° S and assessed during early May. In outdoor sports, vitamin D status has also been analysed in some populations. Guillemant et al.14 studied 54 male adoles
2,000 -0 20 40 60 80 100
Serum 25(OH)D (mmol(L)
Fig. 2.—Correlation between estimated daily calcium intake and serum 25(OH)D levels.
cents from a jockey training centre located at a slightly higher latitude (49°N) than in the current research, and found that serum 25(OH)D values during March were substantially below those reported here (20.4 ± 6.4 nmol/L). In addition, at two successive wintertime points, 25(OH)D levels were clearly deficient (< 25 nmol/L) in 72% and 68% of the young athletes. A French study which included a group of seven male competitive road cyclists (aged 20-39 years) living in Montpellier (at a similar latitude (43°N) to the present study) showed 25(OH)D levels of 83.4 ± 16.0 nmol/L, although the exact time of year (competitive season) and the number of deficient athletes were not reported.15 Lastly, Hamilton et al.16 recently found lower values (< 30 nmol/L) of 25(OH)D in a mixed sample (indoor and outdoor) of athletes resident in the Middle East (s> 25°N) and who were evaluated across a wide period of time (April to October). However, the authors did not distinguish between indoor and outdoor athletes and this prevents comparison with our results. In summary, although one might assume that athletes would be sufficiently exposed to sunlight so as to maintain adequate endogenous production of vitamin D, there are several reasons why such an assumption should be made with caution, especially in the context of indoor sport. First, indoor courts and gyms are particularly common in sports such as basketball for both training and competition. Second, an urban lifestyle favours indoor living and insufficient exposure to sunlight cannot be ruled out, even in favourable environmental conditions.1 Third, the use of sunscreens and protective clothing (e.g. hats and outdoor garments) is becoming more popular because of their protective effect against skin burns and skin cancer.17
Dietary vitamin D intake
When cutaneous endogenous production is limited, diet is the main source of vitamin D.18 Previous observations in athletic populations show large variability in vitamin D intake. Average consumption, including food and supplements, in a large group of Canadian elite athletes was higher in males (286 IU/day) than in females (185 IU/day).19 Interestingly, a group of elite adolescent Spanish soccer players having a buffet-style diet consumed an average of 128 IU/day, significantly lower than another group having a menu-style diet (236 IU/day), thereby indicating an inadequate self selection of food with a high vitamin D content.20 Surprisingly, and despite the higher energy intake, average estimated vitamin D levels in this study were only 139 ± 78 IU/day; only four subjects ingested more than 200 IU/day, which is the minimum recommended daily intake for adults.13 Based on the quantitative analyses of their diet the most straightforward explanation may be the relatively high consumption of meat and derived products to the detriment of oil-rich fish and other vitamin D-rich products. It is worth noting that an adequate consumption of fish —at least four times per week— maintains 25(OH)D above 65 nmol/L.21 If athletes dislike fatty fish or consume only limited amounts, routine supplementation may be required to avoid vitamin D deficiency.
Dietary calcium intake
There is evidence that an adequate vitamin D status is required to achieve the nutritional benefits of calcium and vice versa.22 The absorptive efficiency of calcium increases in line with serum 25(OH)D up to => 80 nmol/L, while above this level additional increases in vitamin D status have no further effect on absorptive regulation.5 Moreover, there is evidence that adequate dietary calcium intake and, in some cases, supplementation with vitamin D and calcium are needed to avoid 25(OH)D deficiency, particularly in black athletes.13 In the present study, the average daily dietary intake of calcium (948 ± 419 mg/day) was borderline to the recommended intake for men aged 19-50 years.13 These results are consistent with previous studies that reported low calcium intake in athletic populations.23 25 Thus, given the important functions that this mineral has in the body, more accurate assessment of dietary calcium consumption should be carried out in athletes, with supplementation being used when dietary intake fails to achieve the optimal amounts.
Ethnicity and serum 25(OH)D
Low levels of 25(OH)D have been reported in the black general population compared to white subjects, this being due to cutaneous melanin (skin pigment), which acts as an effective, ever-present sunscreen.26 In addition, the African-American population usually presents low levels of calcium and vitamin D consumption.27 Although our sample of black players was smaller the results are in agreement with these findings. Black players showed lower serum 25(OH)D values and ingested smaller amounts of calcium and vitamin D. In this regard, future research is needed to analyse ethnic differences in vitamin D status among professional athletes.
Relationship between serum 25(OH)D levels and nutritional data
Interestingly the current study showed a high correlation between 25(OH)D and dietary vitamin D (r = 0.65; P = 0.001), indicating that sufficient amounts of vitamin D-rich or fortified products and supplements may indeed be necessary to ensure adequate vitamin D status in professional indoor athletes during wintertime. Moreover, dietary calcium was also highly associated with serum 25(OH)D levels (r = 0.82; P < 0.001), indicating a close match between the regulatory mechanisms of vitamin D and calcium at sub-optimal levels.22 Dietary calcium was also associated with dietary vitamin D consumption (r = 0.45; P < 0.039), possibly because some sources of calcium (e.g. natural milk or cheese) are also rich in vitamin D and vice versa.
Study limitations and future lines of research
There are several limitations to the present study. One of the most important concerns the very small sample size and the design. However, in contrast to previous research conducted in younger amateur athletes, this study constitutes the first attempt to analyse vitamin D status in professional athletes (a population that is hard to recruit for scientific studies). We believe that the present data are of interest in that they confirm suggestions made in two recent reviews about vitamin D and sport.2 3 A further limitation is that it would have been interesting to obtain serum 25(OH)D levels at the beginning of the season (August). However, players did not begin training at the same time because some of them were involved in competitive matches during August and September, specifically with their national teams in the Olympic Games (season 2008/2009) or European Championships (season 2009/2010). A further point of note is that they were at different latitudes during summer time and this could affect the serum 25(OH)D level at this point in the year. Lastly, our limited data do not enable a relationship to be established between 25(OH)D and performance and/or injury markers in players. In the early twentieth century, athletes and coaches felt that ultraviolet rays had a positive impact on athletic performance, and there is now increasing evidence in support of this view.2 Both cross-sectional and longitudinal studies allude to a functional role for vitamin D in muscle, and, more recently, the discovery of the vitamin D receptor in muscle tissue provides a mechanistic understanding of the function of vitamin D in this context.28 Further research is needed to analyse the potential impact which vitamin D deficiency may have on underperformance and/or risk of injury in athletes.
Conclusion
We conclude that professional basketball players are at higher risk of hypovitaminosis D after wintertime, despite consuming high caloric diets and living in sunny locations.
Nutritionists responsible for the health of athletes should focus their attention on serum 25(OH)D levels and make appropriate recommendations regarding dietary vitamin D and calcium consumption that will help athletes to achieve an adequate vitamin D status.
Aknowledgements
We are indebted to Robert Amat, Kico Pla, Dr. Victor Lamez and Dr. Jorge Guillen of the DKV Joventut Basketball Club for their support in data collection during the study.
References
Holick MF. Vitamin D deficiency. NEngl J Med 2007; 19: 26681.
Cannell JJ, Hollis BW, Sorenson MB, Taft TN, Anderson JJ. Athletic Performance and Vitamin D. Med Sci Sports Exerc 2009; 41: 1102-10.
Willis KS, Peterson NJ. Larson-Meyer DE. Should we be concerned about the vitamin D status of athletes? Int J Sport Nutr Exerc Metab 2008; 18: 204-24.
Lips P. Which circulating level of 25-hydroxyvitamin D is appropriate? / SteroidBiochem Mol Biol 2004; 90: 611 -14.
Heaney RP. Vitamin D and calcium interactions: functional outcomes. Am J Clin Nutr 2008: 88; 541-44.
Lovell G. Vitamin D status of females in an elite gymnastics program. Clin J Sport Med 2008; 18: 159-61.
Lehtonen-Veromaa M, Mottonen T, Irjala K, Karkkainen M, Lamberg-Allardt C, Hakola P, Viikari J. Vitamin D intake is low and hypovitaminosis D common in healthy 9 to 15 year-old Finnish girls. EurJClinNutr 1999; 53: 746-51.
Jonnalagadda SS. Energy and nutrient intakes of the United States national women's artistic gymnastics team. Int J Sports Nutr 1998; 8: 331-44.
Kleiner SM. Nutrition intervention in an all-star basketball player with fatigue. Curr Sports Med Rep 2003; 2: 187-88.
Chapuy MC, Preziosi P, Maamer M, Arnaud S, Galan P, Her-cberg S, Meunier PJ. Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos Int 1997; 7:439-43.
Block G, Hartman AM. Issues in reproducibility and validity of dietary studies. Am J Clin Nutr 1989; 50: 1133-38.
Taylor C, Lamparello B, Kruczek K, Anderson EJ, Hubbard J, Misra M. Validation of a food frequency questionnaire for determining calcium and vitamin D intake by adolescent girls with anorexia nervosa. J Am Diet Assoc 2009; 109:479-85.
Institute of Medicine. Dietary Reference Intakes: Calcium, Phosporus, Magnesium, Vitamin D, and Fluoride. Washington DC: National Academy Press, 1997. Disponible en: http:// www.nap.edu/openbook.php?record_id=5776
Guillemant J, Le HT, Maria A, Allemandou A, Peres G, Guille-mant S. Wintertime vitamin D deficiency in male adolescents: effect on parathyroid function and response to vitamin D3 supplements. Osteoporos Int 2001; 12: 875-79.
Maimoun L, Manetta J, Couret I, Dupuy AM, Mariano-Goulart D, Micallef JP, Peruchon E, Rossi M. The intensity level of physical exercise and the bone metabolism response. Int J Sports Med 2006; 27: 105-cbll.
Hamilton B, Grantham J, Racinais S, Chalabi H. Vitamin D deficiency is endemic in Middle Eastern sportsmen. Public Health Nutr 13: 1528-34.
Koh HK, Bak SM, Geller AC, Mangione TW, Hingson RW, Levenson SM, Miller DR, Lew RA, Howland J. Sunbathing habits and sunscreen use among white adults: results of a national survey. Am J Public Health 1997; 87: 1214-17.
Holick MF, Biancuzzo RM, Chen TC, Klein EK, Young A, Bibuld D, Reitz R, Salameh W, Ameri A, Tannenbaum AD. Vitamin D2 is as effective as vitamin D3 in maintaining circulating concentrations of 25-hydroxyvitamin D. / Clin Endocrinol Metab 2008;93:677-81.
Lun V, Erdman K, Reimer R. Evaluation of nutritional intake in Canadian high-performance athletes. Clin J Sport Med 2009; 19:405-11.
Garrido G, Webster AL and Chamorro M. Nutritional adequacy of different menu settings in elite Spanish adolescent soccer players. Int.I Sport Nutr Exerc Metab 2007; 17: 421 -32.
Nakamura K, Nashimoto M, Hori Y, Yamamoto M. Serum 25-hydroxyvitamin D concentrations and related dietary factors in peri- and postmenopausal Japanese women. Am J Clin Nutr 2000; 71: 1161-65.
Peterlik M, Cross HS. Vitamin D and calcium insufficiency-related chronic diseases: molecular and cellular pathophysiology. Eur J Clin Nutr 2009; 63,1377-86.
Medelli J, Shabani M, Lounana J, Fardellone P, Campion F. Low bone mineral density and calcium intake in elite cyclists. / Sports Med Phys Fitness 2009; 49: 44-53.
Juzwiak CR, Amancio OM, Vitalle MS, Szejnfeld VL, Pinheiro MM. Effect of calcium intake, tennis playing, and body composition on bone-mineral density of Brazilian male adolescents. Int J Sport Nutr Exer Metab 2008; 18: 524-38.
Valimaki W, Alfthan H, Lehmuskallio E, Loyttyniemi E, Sahi T, Stenman UH, Suominen H, Valimaki M J. Vitamin D status as a determinant of peak bone mass in young Finnish men. / Clin Endocrinol Metab 2004; 89: 76-80.
Yetley EA. Assessing the vitamin D status of the US population. Am J Clin Nutr 2008; 88: 558-64.
Heaney RP. Low calcium intake among African Americans: effects on bones and body weight. JNutr 2006; 136: 1095-98.
Hamilton B. Vitamin D and human skeletal muscle. Scand J Med Sci Sports 2009; 20: 182-90.
Withers RT, Craig NP, Bourdon PC, Norton KI. Relative body fat and anthropometric prediction of body density of male athletes. Eur JAppl Physiol Occup Physiol 1987; 56: 191 -200.
Siri WE. Body composition from fluid spaces and density. Analysis of methods. In: Brozec AJ, Henschel A, editors. Techniques for measuring body composition. Washington DC: National Academy of Sciences, 1961, pp. 223-244.
