Response and adverse effects of 20,000 and 30,000 IU Vitamin D weekly
Small safety study of women at high risk of getting breast cancer
Safety, Feasibility, and Biomarker Effects of High-Dose Vitamin D Supplementation Among Women at High Risk for Breast Cancer
International Journal of Food Science, Nutrition and Dietetics (1JFS) ISSN 2326-3350
Crew KD1,2,3*, Xiao T2, Thomas PS4, Terry MB1,3, Maurer M2,3, Kalinsky K2,3, Feldman S3,5, Brafman L3, Refice SR3, Hershman DL1,2,3
Department of Medicine, College of Physicians and Surgeons, Columbia University, New York, NY, USA.
Department of Epidemiology, Mailman School of Public Health, Columbia University, New York, NY, USA.
Herbert Irving Comprehensive Cancer Center, Columbia University, New York, NY, USA.
Department of Clinical Cancer Prevention, MD Anderson Cancer Center, Houston, TX, USA.
Department of Surgery, College of Physicians and Surgeons, Columbia University, NewYork, NY, USA.
Vitamin D deficiency is a potentially modifiable risk factor that may be targeted for breast cancer prevention. We examined the safety, feasibility, and biomarker effects of high-dose vitamin D among women at high risk for breast cancer. Forty high-risk women, defined as a 5-year breast cancer risk >1.67% per the Gail model, lobular or ductal carcinoma in situ, were assigned to a 1-year intervention of vitamin D3 20,000 IU or 30,000 IU weekly. Participants were monitored for toxicity every 3 months, underwent serial blood draws at baseline, 6 and 12 months, and a digital mammogram at baseline and 12 months. Biomarker endpoints included serum 25-hydroxyvitamin D [25(OH)D], 1,25-dihydroxyvitamin D [1,25(OH)2D], parathyroid hormone (PTH), insulin-like growth factor (IGF-1), IGF binding protein (IGFBP-3), and mammographic density (MD) using Cumulus software. From November 2007 to January 2011, we enrolled 40 women; 37 were evaluable at 6 months and 30 at 12 months. One patient was taken off study for hypercalciuria; otherwise, the intervention was well tolerated. From baseline to 12 months, mean serum 25(OH)D and 1,25(OH)2D rose from 20.0 to 46.9 ng/ml and 69.7 to 98.1 pg/ml, respectively (p<0.01). Serum PTH decreased by 12% at 6 months and IGF-1/IGFBP-3 ratio decreased by 4.3% at 12 months (p<0.05). There was no significant change in MD regardless of menopausal status or dose level. We demonstrated that 1 year of high-dose vitamin D3 was associated with a significant increase in circulating vitamin D levels and favorable effects on IGF signaling, but no significant change in MD.
Response of unactivated (standard) Vitamin D:
85% >40 ng in 6 months with both 20,000 and 30,000 IU weekly
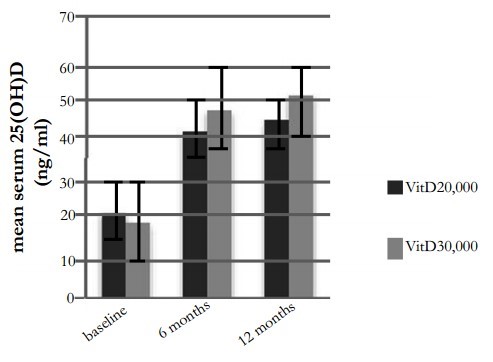
Response by Active Vitamin D
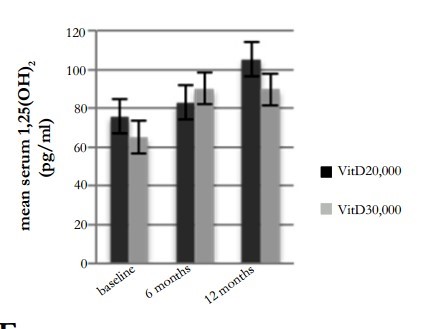
Strange that 30,000 IU/week decreased acitive Vitamin D between the 6th month to 12th month test
Mild adverse effects: >10% of women @ 20,000/week >18% of women @ 30,000 IU/week
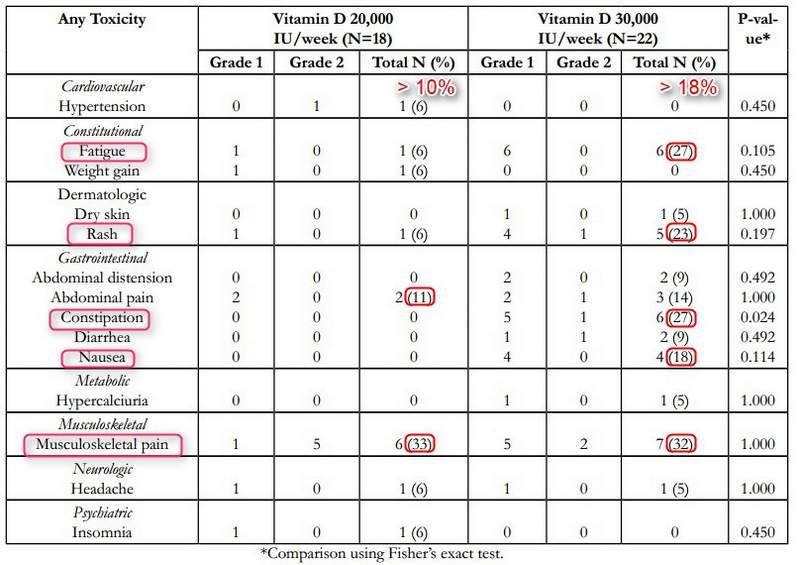
Occasionally people taking vitamin D supplements have previously reported adverse effects
No indication in this study as to when the 2 or 3 pills were taken (all at once, with/without meal, etc.)
Adverse effects shown here appear to be dose-dependant - so the red dye in the pill can be ignored
Often the Adverse effects appear to have been due to a lack of magnesium - which shows up when enough vitamin D is taken
Soluion - be ready to add magnesium when taking lots of vitamin D
📄 Download the PDF from VitaminDWiki
See also VitaminDWiki
Constipation - Magnesium, Calcium and cofactors Lots of Vitamin D and not enough Magnesium ==> constipation
Overview Rare Allergic reaction to vitamin D which include the following
- Try a different form of vitamin D: bioemulsified, liquid, sublingual,
- Try sun or UV instead of vitamin D (but not if you have an allergic reaction to the sun - which sometimes due to being very low on vitamin D)
Try taking it just after the largest meal of the day
Side Effects and Warning for vitamin D
- Excessive thirst, Metal taste in mouth, Poor appetite, Weight loss, Bone pain, Tiredness, Sore eyes, Itchy skin,
- Vomiting, Diarrhea, Constipation, A frequent need to urinate, Muscle problems
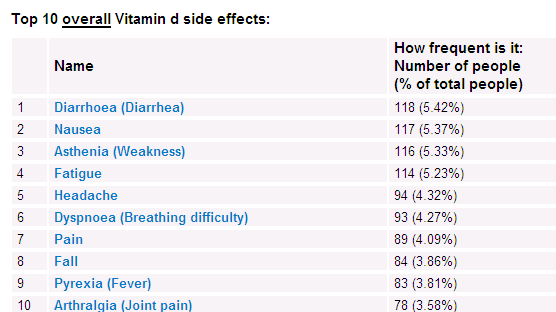
On-line survey of drug uses and side effects – including vitamin D
Which contains the following chart:
Note the following are not the % of people taking vitamin D who got reactions
but rather the % who had any reaction which had a particular reaction