MASLD (NAFLD, Non-Alcoholic Liver Disease) and Vitamin D - meta-analysis
Vitamin D Supplementation for Steatotic Liver Disease: an Updated Systematic Review and Dose-Response Meta-analysis of Randomized and Nonrandomized Interventional Studies
Current Dev. in Nutrition 10 (2026) https://doi.org/10.10167j.cdnut.2025.107631 Iran
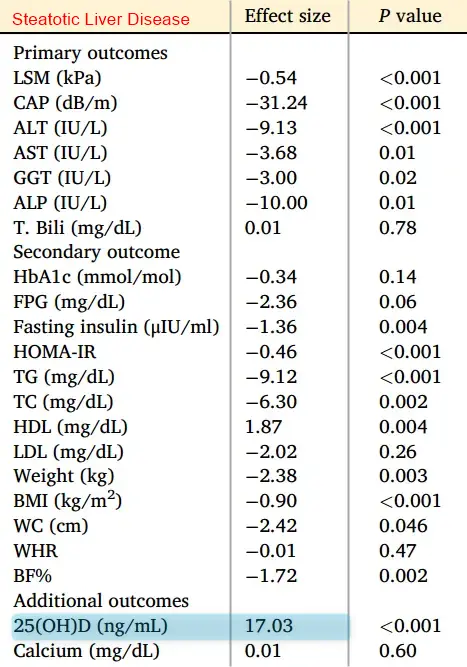
Vitamin D has by far the strongest effect size
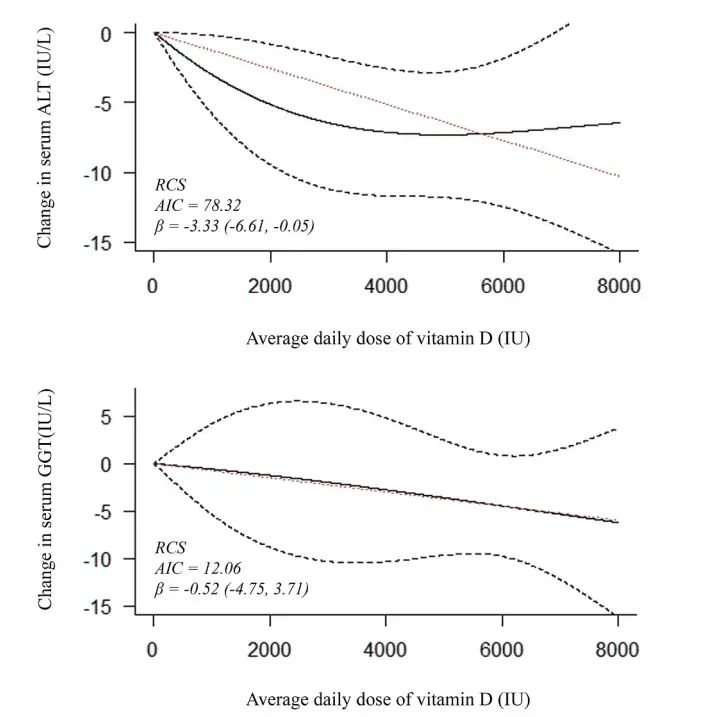
Metabolic dysfunction-associated steatotic liver disease (MASLD) is among the leading causes of chronic liver disease, with few approved treatment options. Vitamin D supplementation has been proposed as a safe and efficacious supplement intervention for MASLD. The current study aimed to systematically evaluate the effect of vitamin D supplementation in any preparation on hepatic (histological, radiological, and biomarker) and metabolic parameters (glucose regulation, lipid profile, and indices of obesity) in patients diagnosed with steatotic liver disease. These effects were compared with patient baseline and/or placebo response, where available. MEDLINE, Scopus, Web of Science, the Cochrane Library, clinicaltrials.gov, and International Clinical Trials Registry Platform were systematically searched for relevant randomized or nonrandomized studies of intervention. Screening and data extraction were completed by independent pairs of reviewers. Effects were pooled using random-effects meta-analyses. One-stage dose-response analysis was performed for alanine aminotransferase (ALT) and y-glutamyl transferase. The effect of baseline vitamin D, ALT, body mass index, and vitamin D response on treatment response was explored via metaregression. Treatment efficacy was evaluated in subgroup analyses according to patient and intervention characteristics.
A total of 28 studies (21 randomized controlled trials) were analyzed. Statistically significant improvements were noted in FibroScan parameters, liver enzymes, insulin resistance, serum triglyceride, and high-density lipoprotein. However, the magnitude of effect regarding these improvements was smaller than thresholds for clinical benefit, and analyses demonstrated inconsistencies. Subgroup analyses failed to identify a specific subset of patients with MASLD benefiting from supplementation. Vitamin D supplementation was safe and well-tolerated, but no meaningful clinical benefit was identified for hepatic or metabolic parameters of interest in MASLD.
Related in VitaminDWiki
- NAFLD and Vitamin D - many studies
- Fewer liver problems (NAFLD, MASLD) if higher Vitamin D -Dec 2025
- NAFLD treated by Vitamin D restoration of gut microbiota
- Liver Fibrosis (NAFLD) 5X less if not obese and good Vitamin D
- Vitamin D is one of only 2 ways to easily prevent NAFLD (380,000 people)
- NAFLD is treated by Vitamin D, Omega-3, Curcumin, Silymarinm, etc.
- Non-alcoholic fatty liver disease (NAFLD) reduced somewhat by 50,000 IU vitamin D every 2 weeks – RCT
- Glutathione fights: Diabetes, Alz, PD, NAFLD, Kidney, UC, IBD, HIV, Fatigue, etc,
- Omega-3 helps NAFLD and other liver problems
People with NAFLD may benefit from gut-friendly forms of Vitamin D
People with NAFLD commonly have measurably “unhealthy” gut features (dysbiosis and increased permeability), but there is almost no direct evidence that they need a special “gut‑friendly” form of vitamin D solely because of NAFLD; standard oral vitamin D still works in most trials, though alternative routes or formulations may be reasonable in selected individuals. pmc.ncbi.nlm.nih
Gut status in NAFLD
Multiple human and animal studies show clear gut microbiota alterations (dysbiosis) in NAFLD compared with healthy controls, including shifts in major phyla (Firmicutes, Bacteroidetes, Proteobacteria, Actinobacteria) and reduced microbial diversity. These dysbiotic patterns are linked to inflammation, toxic metabolite production, and progression toward NASH and cirrhosis, suggesting that NAFLD is partly a gut–liver axis disease. frontiersin
Meta‑analyses and mechanistic reviews indicate that a substantial subset of NAFLD patients have increased intestinal permeability (“leaky gut”), with higher odds of permeability abnormalities compared with healthy controls. This impaired barrier function, combined with dysbiosis, contributes to endotoxemia and hepatic inflammation, though not every NAFLD patient shows the same degree of permeability change. nature
Vitamin D, NAFLD, and the gut
Observational and interventional work links low vitamin D status to NAFLD severity, and several trials report that oral vitamin D supplementation (conventional preparations) can improve liver enzymes and inflammatory/metabolic markers in NAFLD patients. In animal models of diet‑induced NAFLD, vitamin D supplementation not only improves liver pathology but also partially normalizes gut microbiota composition and diversity, suggesting bidirectional effects between vitamin D and the gut–liver axis. pmc.ncbi.nlm.nih
These data show that vitamin D can act as a modulator of dysbiosis and barrier function in NAFLD, rather than being clearly limited by them. However, they do not directly quantify intestinal absorption efficiency of vitamin D in NAFLD, and standard biochemical responses (25(OH)D increases) in trials imply that oral absorption is generally adequate for most NAFLD patients. pmc.ncbi.nlm.nih
Do NAFLD patients specifically need “gut‑friendly” vitamin D?
There is currently no strong clinical trial evidence that NAFLD patients, as a group, require specialized “gut‑friendly” formulations (e.g.,
- liposomal
- nanoemulsion
- buccal, or
- transdermal) to correct vitamin D status or obtain liver benefits. Existing NAFLD vitamin D trials have mostly used standard oral cholecalciferol and still achieved improvements in liver enzymes and metabolic markers, implying that intestinal uptake is usually sufficient. sciencedirect
That said, NAFLD frequently coexists with obesity, diabetes, small intestinal bacterial overgrowth, or more severe barrier dysfunction, which may blunt fat‑soluble vitamin absorption in some individuals; in such cases, higher oral doses, emulsified/liposomal forms, or non‑GI routes (e.g., intramuscular, UVB exposure) could be rational individualized strategies, even though direct comparative evidence in NAFLD is lacking. In practice, the more evidence‑based priorities are: diagnose and treat NAFLD’s metabolic drivers, address gut dysbiosis/permeability with diet and lifestyle, and monitor 25(OH)D responses to oral dosing rather than assuming a special formulation is mandatory. frontiersin
