Mucosal membranes (mouth, lungs, nose, intestines, etc) can activate Vitamin D
Impact of the Local Inflammatory Environment on Mucosal Vitamin D Metabolism and Signaling in Chronic Inflammatory Lung Diseases
Front. Immunol., 10 July 2020 | https://doi.org/10.3389/fimmu.2020.01433
Jasmijn A. Schrumpf*, Anne M. van der Does and Pieter S. Hiemstra
Department of Pulmonology, Leiden University Medical Center, Leiden, Netherlands
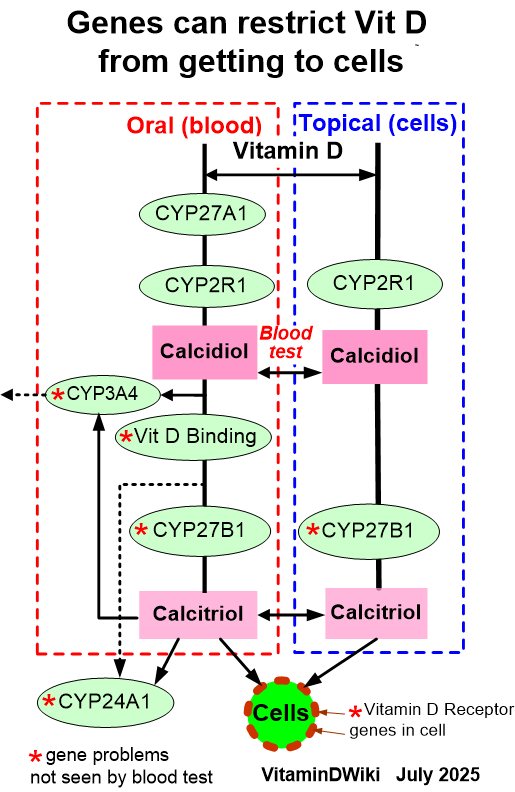 click on image for details
See also Inhaled
* Inhaled vitamin D help lungs - many studies
* Inhaled nanoemulsion of Vitamin D killed lung bacteria – Sept 2017
* Perhaps drugs such as inhaled vitamin D can get directly to the brain – March 2018
* Inhaling Vitamin D nanoemulsion through nose gets lots more to the brain (mice) – July 2020
* Vitamin D thru the mouth appears to be >1000X better at increasing Vitamin D in the lungs than oral Vitamin D
* Inhaled Vitamin D thru the nostrils appears to greatly increase Vitamin D in the brain
See also Inflammation
* Inflammatory diseases: review of vitamin D, with many tables – May 2014
* Inflammation is associated with low vitamin D in at least 15 diseases – April 2020
---
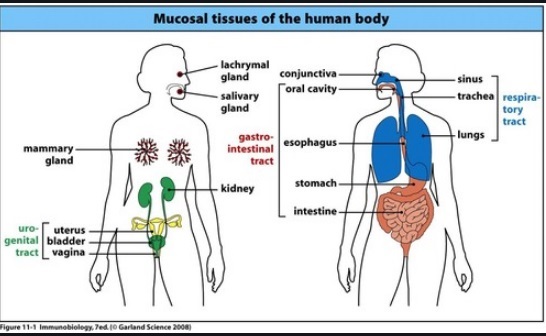
Mucous membrane entry in Wikipedia has the following sub-entries
Bronchial mucosa and the lining of vocal folds, Endometrium: the mucosa of the uterus
Esophageal mucosa, Gastric mucosa, Intestinal mucosa, Nasal mucosa. Olfactory mucosa
Oral mucosa, Penile mucosa, Respiratory mucosa, Vaginal mucosa, Frenulum of tongue
Tongue, Anal canal, Palpebral conjunctiva
Mucosal Tissues
click on image for details
See also Inhaled
* Inhaled vitamin D help lungs - many studies
* Inhaled nanoemulsion of Vitamin D killed lung bacteria – Sept 2017
* Perhaps drugs such as inhaled vitamin D can get directly to the brain – March 2018
* Inhaling Vitamin D nanoemulsion through nose gets lots more to the brain (mice) – July 2020
* Vitamin D thru the mouth appears to be >1000X better at increasing Vitamin D in the lungs than oral Vitamin D
* Inhaled Vitamin D thru the nostrils appears to greatly increase Vitamin D in the brain
See also Inflammation
* Inflammatory diseases: review of vitamin D, with many tables – May 2014
* Inflammation is associated with low vitamin D in at least 15 diseases – April 2020
---
Mucous membrane entry in Wikipedia has the following sub-entries
Bronchial mucosa and the lining of vocal folds, Endometrium: the mucosa of the uterus
Esophageal mucosa, Gastric mucosa, Intestinal mucosa, Nasal mucosa. Olfactory mucosa
Oral mucosa, Penile mucosa, Respiratory mucosa, Vaginal mucosa, Frenulum of tongue
Tongue, Anal canal, Palpebral conjunctiva
Mucosal Tissues
📄 Download the PDF from VitaminDWiki
Vitamin D plays an active role in the modulation of innate and adaptive immune responses as well as in the protection against respiratory pathogens. Evidence for this immunomodulatory and protective role is derived from observational studies showing an association between vitamin D deficiency, chronic airway diseases and respiratory infections, and is supported by a range of experimental studies using cell culture and animal models. Furthermore, recent intervention studies have now shown that vitamin D supplementation reduces exacerbation rates in vitamin D-deficient patients with chronic obstructive pulmonary disease (COPD) or asthma and decreases the incidence of acute respiratory tract infections.
The active vitamin D metabolite, 1,25-dihydroxy-vitamin D (1,25(OH)2D), is known to contribute to the integrity of the mucosal barrier, promote killing of pathogens (via the induction of antimicrobial peptides), and to modulate inflammation and immune responses. These mechanisms may partly explain its protective role against infections and exacerbations in COPD and asthma patients.
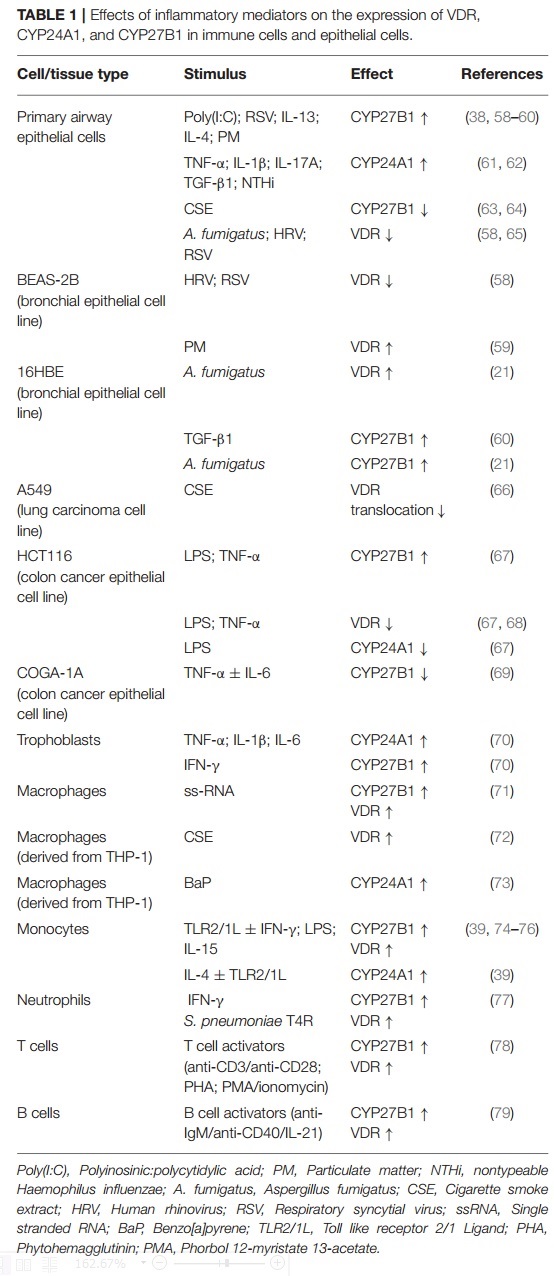
The respiratory mucosa is an important site of local 1,25(OH)2D synthesis, degradation and signaling, a process that can be affected by exposure to inflammatory mediators.
As a consequence, mucosal inflammation and other disease-associated factors, as observed in e.g., COPD and asthma, may modulate the protective actions of 1,25(OH)2D.
Here, we discuss the potential consequences of various disease-associated processes such as inflammation and exposure to pathogens and inhaled toxicants on vitamin D metabolism and local responses to 1,25(OH)2D in both immune- and epithelial cells. We furthermore discuss potential consequences of disturbed local levels of 25(OH)D and 1,25(OH)2D for chronic lung diseases. Additional insight into the relationship between disease-associated mechanisms and local effects of 1,25(OH)2D is expected to contribute to the design of future strategies aimed at improving local levels of 1,25(OH)2D and signaling in chronic inflammatory lung diseases.
Related abstract - Sept 2020
Mucosal vitamin D signaling in inflammatory bowel disease
Review Autoimmun Rev. 2020 Sep 14;102672. doi: 10.1016/j.autrev.2020.102672 (behind a paywall)
Lauge Kellermann 1, Kim Bak Jensen 2, Fredrik Bergenheim 3, John Gubatan 4, Naomi D Chou 5, Alan Moss 6, Ole Haagen Nielsen 3
Epidemiological studies have identified vitamin D (25(OH)D) deficiency to be highly prevalent among patients with inflammatory bowel disease (IBD), and low serum levels correlate with a higher disease activity and a more complicated disease course. The link to IBD pathogenesis has been subject of investigations, primarily due to the distinct immunological functions of vitamin D signaling, including anti-inflammatory and anti-fibrotic actions. Vitamin D is a pleiotropic hormone that executes its actions on cells through the vitamin D receptor (VDR). A leaky gut, i.e. an insufficient intestinal epithelial barrier, is thought to be central for the pathogenesis of IBD, and emerging data support the concept that vitamin D/VDR signaling in intestinal epithelial cells (IECs) has an important role in controlling barrier integrity. Here we review the latest evidence on how vitamin D promotes the interplay between IECs, the gut microbiome, and immune cells and thereby regulate the intestinal immune response. On the cellular level, vitamin D signaling regulates tight junctional complexes, apoptosis, and autophagy, leading to increased epithelial barrier integrity, and promotes expression of antimicrobial peptides as part of its immunomodulating functions. Further, intestinal VDR expression is inversely correlated with the severity of inflammation in patients with IBD, which might compromise the positive effects of vitamin D signaling in patients with flaring disease. Efforts to reveal the role of vitamin D in the pathophysiology of IBD will pave the road for the invention of more rational treatment strategies of this debilitating disease in the future.
