Kidney disease helped by active or high dose Vitamin D
Kidney research, such as summarized in the editorial and meta-analsys on the page, seems to have focused only on Vitamin D and its analogs
For Chronic Kidney Disease there are several options to increase active Vitamin D
- Large dose of vitamin D and help the body activate Vitamin D without the Kidney
Time release active vitamin D - which has been patented (unaware of FDA approval)
A purple herb has a precursor of active vitamin D - which has been used for farm animals for many years
See VitaminDWiki
Many ways to increase Vitamin D: Should use Calcitriol if poor kidney
click on chart for details
Kidney failure – still debating what form of vitamin D to use – April 2016
Vitamin D level can be high, but little benefit: due to kidney, genes, low Magnesium etc.
Peritoneal Dialysis nicely treated by active vitamin D – July 2013
Kidney Dialysis clinics reluctant to add vitamin D treatment as they are not reimbursed – Oct 2012
4X more Chronic Kidney disease patients are now using vitamin D – March 2014
Kidney Inflammation: both prevented and reduced by Vitamin D – review Jan 2014
Adding Vitamin D decreased kidney deaths by 4x – Dec 2010 way back then some of the trials were terminated as they felt it
- morally wrong to not give vitamin D to all of the Kidney patients
Items in both Kidney and Calcitriol categories in VitaminDWiki:
{category}
The beneficial impact of vitamin D treatment in CKD patients: what's next? - Editorial Comment
Clin Kidney J (2015) 8:38-40 doi: 10.1093/ckj/sfu135
Patients with chronic kidney disease (CKD) have markedly higher rates of severe vitamin D deficiency and reduced ability to convert 25-(OH)vitamin D into the active form, 1,25-dihydroxyvitamin D [1]. In the setting of CKD, secondary hyperparathyroidism develops as a consequence of reduced renal production of active vitamin D and phosphate retention resulting in hypocalcaemia and hyperpho- sphataemia. This is a process that is dangerously linked with metabolic bone disease, arterial calcifications and cardiovascular mortality [2]. Therefore, the conventional rationale for vitamin D treatment in CKD is to slow the progression of secondary hyperparathyroidism.
In addition to the classical pathway for activation of 25-(OH)vitamin D to 1,25-(OH)2 vitamin D, a peripheral autocrine pathway exists and results in calcitriol synthesis in a variety of peripheral extra-renal tissues [3]. By binding with its intracellular vitamin D receptor (VDR) in these tissues, calcitriol can regulate cellular proliferation and differentiation, inflammation, the immune system and the endocrine system, including insulin resistance, lipid metabolism and renin-angiotensin system (RAS) [4]. Interestingly, active vitamin D analogues have shown demonstrably favourable effects on proteinuria, likely through interference with RAS [5, 6]. The discovery of this non-classical pathway has brought new significance to the importance of addressing nutritional vitamin D deficiency [7].
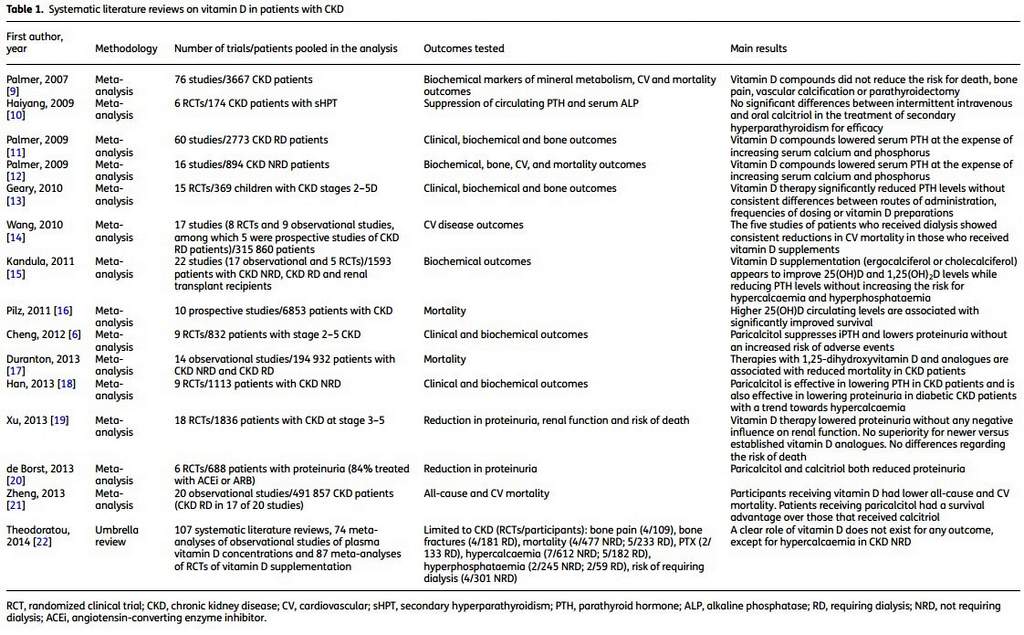
Vitamin D deficiency has been associated with all-cause and cardiovascular mortality in patients with CKD, whereas therapies with vitamin D and analogues have been associated with reduced mortality, recently also in meta-analysis of observational studies (Table 1). However, evidence from randomized controlled trials (RCTs) supporting a survival benefit from active and/or pre-active vitamin D administration in CKD patients is still lacking. Moreover, it is not even known whether different types of active vitamin D, selective or non-selective VDR activators, or precursors have a diversified effect on mortality in the CKD population.
In the present issue of the Clinical Kidney Journal, Mann et al. [8] present a meta-analysis of RCTs to investigate the effect of oral vitamin D therapy versus placebo on mortality and cardiovascular outcomes among adults with CKD, whereas vitamin D supplementation was not found to exert any significant effect on these hard outcomes. Analysis of pooled data displayed a substantial overlap in confidence intervals and homogeneity between study results. Stratification of trials by CKD stage, weekly vitamin D dose, proportion of diabetic subjects and vitamin D compound displayed similar results. In detail, 13 trials that, overall, enrolled 1469 patients with CKD stage 1-5D were selected for analysis and none of them had mortality as a primary outcome. These studies were mainly designed to test biochemical or bone histological end points and consequently had a rather short follow-up. On the whole 41 all-cause deaths (2.8%) were recorded during a follow-up ranging from 3 to 104 weeks (mean 41 weeks). Of note, about two-thirds of the patients (n = 1087) had been followed for <1 year (mean 21, range 3-48 weeks), registering 17 all-cause deaths (41%), 8 cardiovascular deaths (62%) and 18 cardiovascular events (86%). Only two trials (total patient number = 233) had a follow-up time up to 2 years, but they registered only 11 deaths of which, 5 had a cardiovascular cause. Taken together, these observations could indicate that the duration of follow-up may have been insufficient to capture possible differences in mortality, as correctly stated from the authors in the limitation section and as well as suggested by the relatively low number of events displayed.
Moreover, not negligible differences are also present in patient populations (End Stage Renal Disease in 5 of 13 trials) and in interventions, above all considering the heterogeneity in administered vitamin D compounds and dosages.
In conclusion, it is not the time to say that interventions based on vitamin D may reduce mortality in patients with CKD, but the opposite cannot be said yet beyond all reasonable doubt. In fact, given the paucity of good quality data, the reliability of the pooled results is still uncertain.
📄 Download the Editorial PDF from VitaminDWiki
Effect of oral vitamin D analogs on mortality and cardiovascular outcomes among adults with chronic kidney disease: a meta-analysis
Clin Kidney J (2015) 8:41-48 doi: 10.1093/ckj/sfu122 Advance Access publication 2 December 2014
Michelle C. Mann1, Amy J. Hobbs2, Brenda R. Hemmelgarn2,3, Derek J. Roberts2’4, Sofia B. Ahmed1’3 and Doreen M. Rabi2 department of Medical Sciences, Cumming School of Medicine, University of Calgary, Calgary, AB, Canada, department of Community Health Sciences, Cumming School of Medicine, University of Calgary, Calgary, AB, Canada, division of Nephrology, Cumming School of Medicine, University of Calgary, Foothills Medical Centre, Calgary, AB, Canada and department of Surgery, Cumming School of Medicine, University of Calgary, Foothills Medical Centre, Calgary, AB, Canada
Correspondence to: Michelle C. Mann; E-mail: mcmann@ucalgary.ca
Background. Vitamin D deficiency is highly prevalent in patients with chronic kidney disease (CKD) and has been associated with all-cause and cardiovascular mortality in observational studies. However, evidence from randomized controlled trials (RCTs) supporting vitamin D supplementation is lacking. We sought to assess whether vitamin D supplementation alters the relative risk (RR) of all-cause and cardiovascular mortality, as well as serious adverse cardiovascular events, in patients with CKD, compared with placebo.
Methods. PubMed/MEDLlNE, EMBASE, Cochrane Library, and selected nephrology journals and conference proceedings were searched in October 2013. RCTs considered for inclusion were those that assessed oral vitamin D supplementation versus placebo in adults with CKD (<60 mL/min/1.73 m2), including end-stage CKD requiring dialysis. We calculated pooled RR of mortality (all-cause and cardiovascular) and that of cardiovascular events and stratified by CKD stage, vitamin D analog and diabetes prevalence.
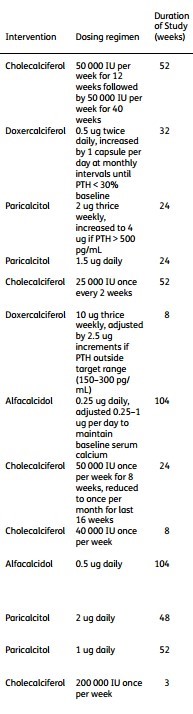
Results. The search identified 4246 articles, of which 13 were included. No significant treatment effect of oral vitamin D on all-cause mortality (RR: 0.84; 95% CI: 0.47,1.52), cardiovascular mortality (RR: 0.79; 95% CI: 0.26, 2.28) or serious adverse cardiovascular events (RR: 1.20; 95% CI: 0.49, 2.99) was observed. The pooled analysis demonstrated large variation in trials with respect to dosing (0.5 ug-200 000 IU/week) and duration (3-104 weeks).
Conclusions. Current RCTs do not provide sufficient or precise evidence that vitamin D supplementation affects mortality or cardiovascular risk in cKd. While its effect on biochemical endpoints is well documented, the results demonstrate a lack of appropriate patient-level data within the CKD literature, which warrants larger trials with clinical primary outcomes related to vitamin D supplementation.
📄 Download the Meta-analysis PDF from VitaminDWiki
Types of Vitamin D in meta-analyses
Note: Calcitriol is not listed!!
(A few columns of table 1 in the PDF)