Vitamin D improves sleep and reduces pain – review of studies
The interfaces between vitamin D, sleep and pain
J Endocrinol. 2017 Jul;234(1):R23-R36. doi: 10.1530/JOE-16-0514. Epub 2017 May 23.
Daniela Leite de Oliveira, Camila Hirotsu, Sergio Tufik and Monica Levy Andersen
Department of Psychobiology, Universidade Federal de Sao Paulo, Sao Paulo, Brazil
Correspondence should be addressed to C Hirotsu milahirotsu@gmail.com
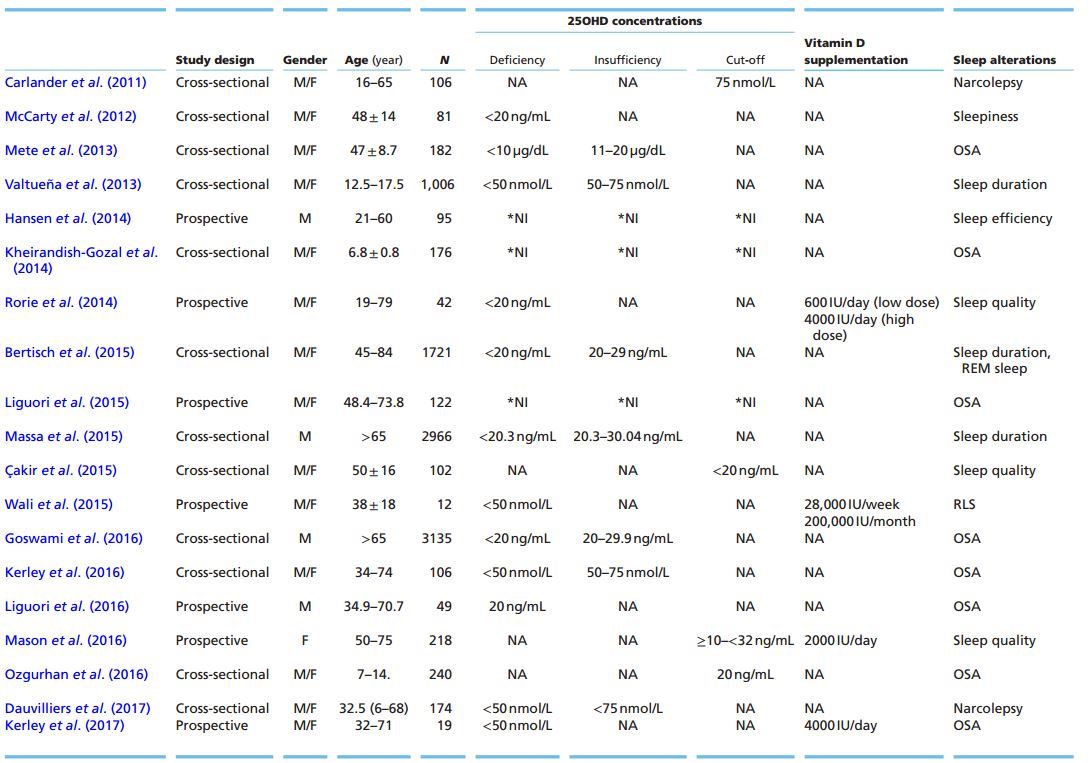
T1: Studies of Vitamin D and Sleep
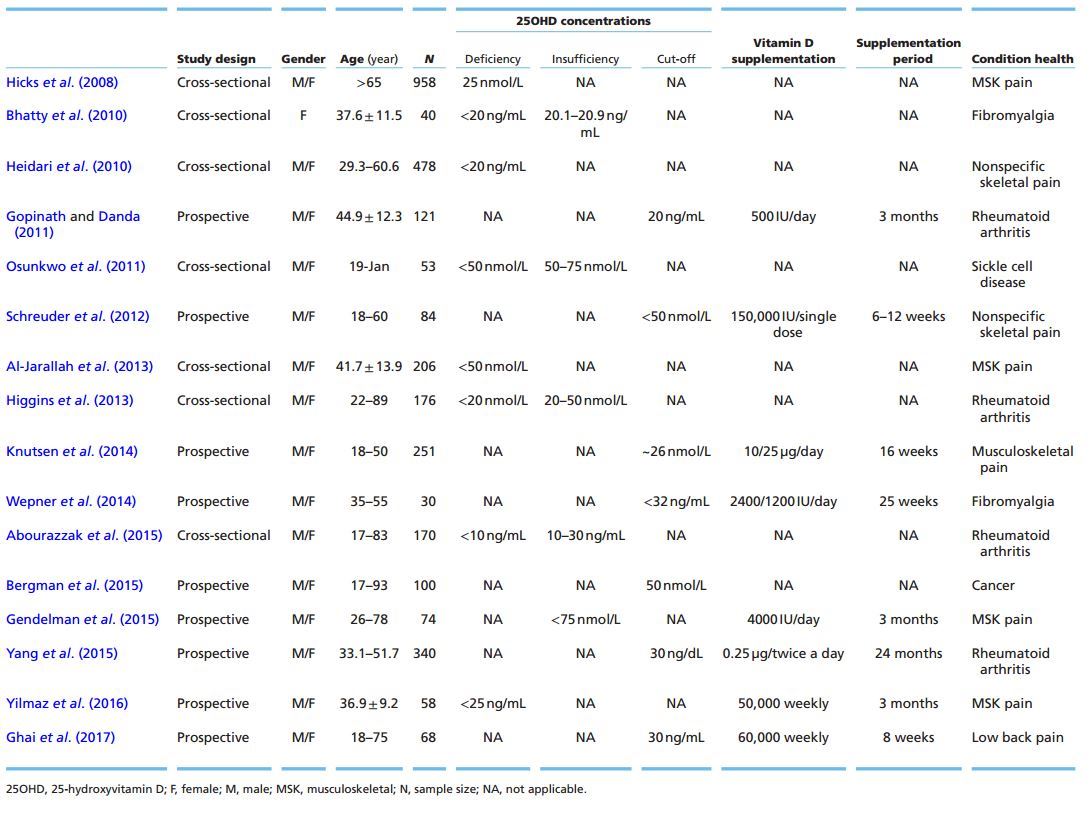
T2: Studies of Vitamin D and Pain
The role of vitamin D in osteomineral metabolism is well known. Several studies have suggested its action on different biological mechanisms, such as nociceptive sensitivity and sleep-wake cycle modulation. Sleep is an important biological process regulated by different regions of the central nervous system, mainly the hypothalamus, in combination with several neurotransmitters. Pain, which can be classified as nociceptive, neuropathic and psychological, is regulated by both the central and peripheral nervous systems. In the peripheral nervous system, the immune system participates in the inflammatory process that contributes to hyperalgesia. Sleep deprivation is an important condition related to hyperalgesia, and recently it has also been associated with vitamin D. Poor sleep efficiency and sleep disorders have been shown to have an important role in hyperalgesia, and be associated with different vitamin D values. Vitamin D has been inversely correlated with painful manifestations, such as fibromyalgia and rheumatic diseases. Studies have demonstrated a possible action of vitamin D in the regulatory mechanisms of both sleep and pain. The supplementation of vitamin D associated with good sleep hygiene may have a therapeutic role, not only in sleep disorders but also in the prevention and treatment of chronic pain conditions.
Introduction
The musculoskeletal benefits of vitamin D are widely known: they promote the calcification of bone matrix and increase the absorption of calcium and phosphate in the intestine. Vitamin D deficiency can cause rickets in children and osteomalacia in adults. It also contributes to bone frailty and consequent fracture in the elderly.
The endogenous synthesis of vitamin D occurs in the epidermis from the precursor 7-dehydrocholesterol (7-DHC) through ultraviolet B radiation (wavelengths between 290 and 315 nm). Vitamin D2 (ergosterol) and vitamin D3 (cholecalciferol) can be obtained from vegetable and animal sources, such as shiitake mushrooms and salmon, respectively (Holick & Chen 2008, IOM 2011). When both ergosterol and cholecalciferol reach the liver and kidney through circulation, two consecutive hydroxylations occur mediated by cytochrome (CYP) P450. In the kidney, 25-hydroxyvitamin D (25OHD) is converted to its metabolically active form: 1-a,25-dihydroxyvitamin D, also known as calcitriol (1,25(OH)2D). This active molecule (1,25(OH)2D) can also be produced in different cells and tissues, such as skin, colon, placenta and prostate (Holick & Chen 2008, EFSA 2016). Calcitriol binds to the vitamin D receptor (VDR), a transcription factor that translocates to the cell nucleus, in which it is heterodimerized with the retinoid X receptor (RXR). Then, the calcitriol-VDR-RXR complex couples to a specific sequence of DNA known as the vitamin D response element (VDRE), which is associated with promoters of several target genes and co-activator molecules (Holick et al. 2011).
Many conditions interfere with the endogenous synthesis of vitamin D. The lower the amount of sunlight, the lower the endogenous vitamin D production. Age is also relevant, but not determinant to the reduction of 7 DHC production due to increased thickening of the skin in elderly individuals (Need et al. 1993, Holick & Chen 2008). Additionally, skin pigmentation can modulate the synthesis of vitamin D, since the amount of melanin restricts the radiation of photons that are necessary for its endogenous production. Consequently, dark/black skin individuals need longer exposure to the sun to get optimal levels of vitamin D (Hagenau et al. 2009, Gordon et al. 2012, Correia et al. 2014).
Vitamin D status is based on the serum levels of 25OHD, the metabolite found in the human body in higher concentration. Currently, there is a debate about the optimal reference value of vitamin D, which is primarily based on bone health parameters. Different scientific committees' panels indicate that 25OHD serum levels lower than 20 ng/mL are appropriate for good health in the general population (IOM 2011, EFSA 2016, SACN 2016). On the other hand, the Endocrine Practice Guideline suggests a screening in groups at risk for vitamin D deficiency (Holick et al. 2011). In this guideline, individuals with 25OHD serum levels of 20 ng/mL are considered as deficient for vitamin D, while serum levels above 30 ng/mL are considered sufficient to provide health benefits (Holick et al. 2011). Manson and colleagues (Manson et al. 2016) have addressed this issue by emphasizing that the suggested values for 25OHD sufficiency of >30 ng/mL are due to a misinterpretation and misunderstanding of the reference value indicated by the IOM. For clinical practice, it is important to establish a cut-off point. However, this requires more standardized studies, since most experimental designs available in the literature were not well controlled for confounders and based solely on bone health
.
25OHD serum levels have been associated with several comorbidities such as infectious, autoimmune and neurological diseases, as well as neuromuscular disorders, which can lead to increased pain sensitivity (Dhesi et al. 2002, Orme et al. 2013, Lachmann et al. 2015). Regarding the mechanisms of pain sensitization, vitamin D seems to stimulate anti-inflammatory processes in some cases and thus to relieve the painful sensation of many diseases (Adorini & Penna 2008, Osunkwo et al. 2011, Le Goaziou et al. 2014). It is important to mention that the anti-inflammatory actions on pain conditions are not specific for vitamin D. Recently, researchers have suggested the participation of vitamin D in regulatory mechanisms of sleep-wake cycle, based on the evidence suggesting that the reduction of this vitamin is also associated with sleep disorders (McCarty et al. 2012, Mete et al. 2013, Qakir et al. 2015, Kerley et al. 2016).
This proposed ability of vitamin D to act on both the sleep-wake cycle and the nociceptive process may be due to the fact that they share some neurotransmitters in similar pathways. In this review, we aim to explore the current knowledge about the association between sleep and pain and to consider the role that vitamin D may play in this relationship. Finally, we will highlight new perspectives about the potential applicability vitamin D may have in sleep disorders and chronic painful conditions.
Vitamin D and sleep
Sleep regulation
Sleep is a complex physiological state, and although often thought as an inactivity period, it comprises a period of intense metabolic activity. Its function is still not fully understood, although it is known that this biological phenomenon participates in several processes, e.g., learning, consolidation of memory, inflammation, and metabolic disorders, being essential to good mental and physical health (Walker & Stickgold 2004, Van Cauter et al. 2008, Hirotsu et al. 2012, Ruiz et al. 2012, Garcia et al. 2013, Davis et al. 2017).
Sleep is classified into the rapid eye movement (REM) stage, also known as paradoxical sleep, and the non-REM (NREM) or slow-wave sleep stage. NREM sleep is characterized by cortical synchronization, muscular hypotonia and endocrine regulation. REM sleep presents cortical desynchronization, muscle atonia, rapid eye movements and dream activity (Jouvet 1967, Carskadon & Dement 2017).
The sleep-wake cycle is modulated by different regions of the central nervous system (CNS). Its main regulation occurs in the ventrolateral preoptic nucleus (VLPO) of the hypothalamus through interaction with circadian and homeostatic factors. A set of neurons from the suprachiasmatic nucleus is activated by circadian influences such as absence and/or presence of light, while hypnogenic homeostatic factors such as adenosine accumulation in the forebrain are induced by prolonged wakefulness or decreased by sleep (Saper et al. 2005). Another important factor in the regulation of sleep is the presence of melatonin, an endogenous hormone produced by the pineal gland and responsible for the regulation of circadian rhythms. The absence of light stimulates melatonin production, leading to sleep facilitation. During the daytime, the light stimulus is processed by the retina and sent to the suprachiasmatic nucleus, inhibiting melatonin synthesis and promoting wakefulness (McArthur et al. 1991, Cajochen et al. 2003).
During wakefulness, due to its higher behavioral complexity, numerous neural systems are activated in the brainstem reticular formation, which is a network of cells and fibers that project throughout the cerebral cortex, producing cortical activity and controlling the waking state (Jones 2011). Acetylcholine, norepinephrine, histamine and hypocretin neurotransmitters participate in the cortical activation process, while dopamine and serotonin participate in both cortical and behavioral activation. Conversely, y-aminobutyric acid (GABA), an inhibitory neurotransmitter, is associated with sleep. From the VLPO, neuronal GABAergic projections inhibit the release of hypocretin, acetylcholine, norepinephrine, serotonin and dopamine, resulting in brain synchronization. During REM sleep, however, there is continued inhibition of these neurotransmitters, except for acetylcholine, which provides cortical activity during REM sleep (Espana & Scammell 2011).
The advance of technology and increased urbanization over the 20th and 21st centuries caused the suppression of melatonin synthesis by artificial light produced at night. This has led to extended periods of wakefulness and sleep curtailment. Sleep deprivation induced by exposure to artificial light causes several impairments in the regulation of biological rhythms, cognition and brain development, leading to long-term negative impacts on quality of life (Thomas et al. 2000, Durmer & Dinges 2005, Tufik et al. 2009).
The most common sleep disorders in the general population are obstructive sleep apnea (OSA) and insomnia, followed by others of lower prevalence such as sleep-related movement disorders, parasomnias and central hypersomnolence disorders. OSA is a sleep disordered breathing condition that affects one-third of the general population and is characterized by complete or partial obstruction of the upper airways resulting in a higher apnea-hypopnea index (AHI>15), snoring, morning headaches and dry mouth (Tufik et al. 2010). Risk factors for OSA include obesity and being male and older. The cardiovascular system is also impaired in OSA due to intermittent hypoxia, sleep fragmentation and reduction of intrathoracic pressure. This condition may lead to vascular injury contributing to the increase ofsystemic blood pressure (Drager et al. 2013, Franklin & Lindberg 2015).
Insomnia is characterized by a difficulty in initiating and/or maintaining sleep or early awakening with a frequency of more than 3 times a week over a period of 3 months (ICSD-3, 2014), and affects around 15-45% of the general population. Individuals with insomnia frequently have a non-restorative sleep and may present early awakenings. Among the predisposing and risk factors are female gender, anxiety and depression (Castro et al. 2013).
Vitamin D and sleep
The recent interest in the role of vitamin D as a sleep modulator is based on studies in both animal models and humans, indicating the neuronal expression of CYP27B1 and VDR in different brain areas, especially in regions that also regulate the sleep-wake cycle, such as the hypothalamus (Stumpf & O'Brien 1987, Perez- Fernandez et al. 1997, Prufer et al. 1999, Eyles et al. 2005, 2014).
The association between sleep disorders and 25OHD values has increased in the last decade (Andersen & Tufik 2012, Bozkurt et al. 2012, Kheirandish-Gozal et al. 2014, McCarty et al. 2014, de Oliveira et al. 2015, Evatt 2015). The possible mechanisms involved in this association are not clear, suggesting that additional studies are required to understand the potential role of vitamin D in physiological sleep mechanisms. Table 1 summarizes the current findings from the clinical literature. One of the first studies showed a significant association between excessive daytime sleepiness and serum values of 25OHD <20 ng/mL only in black individuals (McCarty et al. 2012). Valtuena et al. (2013) found that lower levels of 25OHD concentrations were associated with shorter sleep duration (<8 h) in female teenagers. In this study, sleep duration was assessed by self-administered questionnaires, which only provide subjective information about the sleep pattern of subjects. Recently, other studies have evaluated the relationship between serum 25OHD levels and sleep using objective parameters. Low 25OHD serum levels (<30 ng/mL) were associated with shorter sleep duration (<5 h) measured by actigraphy among men (Massa et al. 2015). Similarly, Bertisch et al. (2015) reported that shorter sleep duration (373.3 ± 80.7 min) measured by polysomnography was significantly associated with 25OHD serum levels lower than 20 ng/mL, while a lower proportion of REM sleep was weakly correlated with 25OHD values between 20 and 29 ng/mL. In addition, a study using actigraphy in Review d l de oliveira and others Sleep and pain: the role of 234:1 xxxx male forensic patients demonstrated that sleep latency decreased in the group who was submitted to a fish- enriched diet compared to the control group. Moreover, 25OHD serum levels increased with fish consumption, and a positive correlation between vitamin D and sleep efficiency was found (Hansen etal. 2014). It is important to highlight the heterogeneous characteristics of the sample in this study, such as age, gender and the presence of comorbidities, which hamper the establishment of a clear relationship between vitamin D and sleep maintenance.
Other studies have demonstrated that in patients with OSA, the higher the body mass index (BMI) and AHI, the lower the 25OHD levels (Mete et al. 2013, Kerley et al. 2016). Additionally, Goswami and coworkers (2016) found that BMI and neck circumference could be a confounding factor in the association observed between 25OHD serum levels <20 ng/mL and some risk factors for OSA. A further study revealed that the use of continuous positive airway pressure (CPAP), the gold standard for OSA treatment, promoted increased levels of 25OHD (from 19.21 ± 9.45 ng/mL to 21.03 ± 9.50 ng/mL) (Table 1) in men with OSA after 7 days of CPAP use (Liguori et al. 2015). The CPAP therapy was also effective in increasing 25OHD serum concentrations from 16.05 ± 7.74 ng/mL to 25.73 ± 12.91 ng/mL after 1 year of treatment in both obese and non-obese OSA patients (Liguori et al. 2016). The increase in serum 25OHD levels and decrease in both sleepiness and AHI were mainly observed in the obese patients compared to non-obese OSA men treated with CPAP. A pilot study assessing Caucasian OSA individuals submitted to cholecalciferol supplementation (4000 I U/ day) during 15 weeks (Kerley et al. 2017) showed an increase in 25OHD serum levels in the supplemented group vs placebo (100.8 ± 39.9 nmol/L x 43.0 ± 21.4 nmol/L, respectively), and a decrease in metabolic parameters such as low-density lipoprotein (LDL) and lipoprotein- associated phospholipase A2 (Lp-PLA2). Some subjects from both groups used CPAP, but the data were not controlled, hampering the interpretation about the benefits of the supplementation since there is current evidence that CPAP use can increase 25OHD levels (Liguori et al. 2015).
In pediatric patients, the clinical condition is similar. Obese children present lower serum levels of 25OHD (83.6 ± 19.2 ng/mL) compared to non-obese children (97.1 ± 20.9 ng/mL); and obese children with OSA have lower 25OHD levels than obese children without OSA (Kheirandish-Gozal 2014). Assessing the risk of developing OSA using the Pittsburg Sleep Quality Index (PSQI) questionnaire, Ozgurhan and coworkers (2016) found that children of both sexes with serum 25OHD levels ranging 13.10 ± 4.02 ng/mL had a higher risk of developing OSA than the control group (27.12 ± 5.63 ng/ mL). The group at high risk of developing OSA presented a higher BMI compared to those at low risk.
Restless legs syndrome (RLS) is a sleep-related movement disorder that has also been linked to lower levels of vitamin D. An urge to move the legs characterizes this syndrome, mainly during the end of the day, which ameliorates with movement and whose physiopathology is associated with a possible dysfunction in dopaminergic neurotransmission. RLS affects sleep quality by increasing both sleep latency and daytime sleepiness. Using the PSQI questionnaire, RLS was found to be more frequent in individuals with serum 25OHD levels <20 ng/mL compared to controls (serum 25OHD >20 ng/mL) (Qakir et al. 2015) (Table 1). Vitamin D supplementation (28,000 IU/week; 200,000/month) showed a decrease in RLS severity in patients compared to their baseline (serum 25OHD at baseline: 21.7 nmol/L (13.45-57.4); serum 25OHD after supplementation: 61.8 nmol/L (42.58-95.9)) (Table 1) (Wali et al. 2015). 1,25(OH)2D acts on dopaminergic neurons and its in vitro action increases the expression of N-cadherin, a molecule that participates in the neurogenesis of dopaminergic neurons and that possibly regulates the expression of tyrosine hydroxylase enzyme, a rate-limiting molecule in the dopamine synthesis pathway (Cui et al. 2013, 2015, Jiang et al. 2014a,b).
In individuals diagnosed with narcolepsy with cataplexy, a central hypersomnolence disorder, the serum 25OHD levels were lower compared to individuals without the disease (59.45 nmol/L x 74.73 nmol/L, respectively) (Table 1) (Carlander et al. 2011). Recently, Dauvilliers and coworkers (2017) did not find any association between narcolepsy individuals (77.46 (21.10-209.9) nmol/L) and serum 25OHD levels <20 ng/mL compared to the control group (77.21 (3.73-437.25) nmol/L). Narcolepsy is a sleep disorder characterized by excessive sleepiness that can be or not associated with cataplexy (reduced muscle tonus without loss of consciousness) and hallucinations. The mechanism of narcolepsy is attributed to the decrease in the concentration of hypocretin, a neuropeptide responsible for wakefulness in the CNS.
In a placebo-controlled randomized clinical trial with postmenopausal women participating in a weight loss program (Table 1), Mason and coworkers (2016) reported that individuals undergoing cholecalciferol supplementation (2000 IU/day for 12 months) had impaired subjective sleep quality as assessed by the PSQIvitamin D questionnaire. However, another randomized study reported cholecalciferol supplementation (4000 IU/day for 12 weeks) benefitted sleep (Table 1) in chronic urticaria patients. The authors found a trend of improvement in sleep quality in the high-vitamin D supplemented group compared to the low-vitamin D supplemented one (600 IU/day) (Rorie et al. 2014). The different designs of both studies make it difficult to conclude whether vitamin D supplementation has beneficial effects on sleep quality.
Vitamin D and pain
Pain regulation
According to the International Association for the Study of Pain (IASP), pain is an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage (IASP 1994). It can be classified into nociceptive, originating in the mechanical, thermal or chemical nociceptors (sensory neurons) of the physical area in which the injury occurs; neuropathic, caused by injury or disease of the nervous system; or psychological, from an emotional origin. Acute pain comes from inflammation or tissue damage and is related to tissue repair and the healing process. Chronic pain persists for the period of injury, causing damage to the individual's quality of life (Schaible & Richter 2004).
The most important nerve centers responsible for the transmission of nociceptive information are the thalamus, midbrain, limbic system and reticular formation. The nociception process consists of 3 stages: stimulus transduction by first-order neurons of the spinal cord, where neurotransmitters such as glutamate, substance P and calcitonin are released and activate second-order neurons. These, in turn, transmit information to the thalamus, where the pain sensitivity occurs. Third-order neurons connect the thalamus to the cortex, activating it and resulting in pain perception (Schaible & Richter 2004, Klaumann et al. 2008).
Several neurotransmitters play a role in the pain sensitization process. Norepinephrine, dopamine and serotonin modulate the descending pain pathway by inhibition or excitation of their target receptors. Nitric oxide acts on nociceptive sensitivity (Meller & Gebhart 1993, Millan 2002). Prostaglandin E2 (PGE2) contributes to inflammatory pain (Kawabata 2011).
Another important component of pain mechanism is the immune system. Because of injury, inflammation of the damaged tissue may cause hyperalgesia. Activation of macrophages in the damaged peripheral tissues results in the release of proinflammatory cytokines, such as tumor necrosis factor alpha (TNF-a), interleukin-1 beta (IL-1p), neuronal growth factor (NGF), nitric oxide and PGE2. Neutrophils are also recruited in this process by inflammation-producing substances such as cytokines and chemokines. The accumulation of these cells in inflammatory processes leads to opioid expression, with antinociceptive effects (Marchand et al. 2005, Ren & Dubner 2010, Shipton & Shipton 2015).
In the last decades, studies have suggested the action of the active form of vitamin D (1,25OH2D) in several cell types of the immune system. It acts in Th2 cells by increasing the synthesis of interleukin-4 (IL-4) and transforming growth factor-p (TGF-p) (Deluca & Cantorna. It is known that TGF-p decreases the expression of proinflammatory cytokines, such as interferon-y (IFN-y), IL-1 and TNF-a. Both TGF-p and IL-4 are found in microglial cells (Garcion et al. 2002). Vitamin D inhibits the action of PGE2 by inhibiting its precursor, cyclooxygenase-2 (COX- 2) (Moreno et al. 2006). Finally, studies have shown that 1,25(OH)2D precludes the synthesis of inducible nitric oxide synthase (iNOS), an enzyme that produces large amounts of nitric oxide when stimulated (Garcion et al.
. These regulatory pathways are involved in pain sensitization and 1,25(OH)2D appears to regulate important molecules involved in this process, suggesting it has a potential neuro-immunomodulatory action.
Vitamin D and pain
Different anatomical regions may be the targets of musculoskeletal pain such as the lower limbs (legs and knees) and back. This chronic pain condition causes socioeconomic and psychological harms. Moreover, the cause of these disorders is often associated with several factors such as age, gender, diet and psychological status (McBeth & Jones 2007). Researchers have shown that women with 25OHD serum levels <20 ng/mL have a higher chance of developing pain (Table 2) (Hicks et al. 2008, Heidari et al. 2010). Several studies have indicated that vitamin D supplementation (Table 2) is effective in reducing complaints in specific or nonspecific musculoskeletal pain disorders (Schreuder et al. 2012, Gendelman et al. 2015, Yilmaz et al. 2016, Ghai et al. 2017). In a recent meta-analysis (Table 2), Wu and colleagues (Wu et al. 2016) reported, in a subgroup analysis of vitamin D group vs placebo, that vitamin D supplementation for an average time duration of 3 months reduced pain in conditions such as myalgia, arthritis and chronic musculoskeletal pain. This result was observed in hospitalized patients with pain-related medical conditions. Although some studies have shown a negative association between musculoskeletal pain and serum 25OHD, little is known about the mechanisms involved in this relationship (Table 2) (Al-Jarallah et al. 2013, Knutsen et al. 2014).
In fibromyalgia , a diffuse chronic pain syndrome, there are reports of an inverse relationship between serum 25OHD and pain. When evaluating the 25OHD concentrations among women with fibromyalgia, a study showed lower levels in younger compared to older women. There was no information about a control group (Table 2) (Bhatty et al. 2010). This study was performed in Pakistan, a region where women's typical clothing is the burqa, which hinders endogenous vitamin D synthesis. Wepner and coworkers (2014) supplemented individuals with fibromyalgia using 2400 or 1200 I U/ day to reach serum 25OHD levels of 32-48 ng/mL (Table 2). According to the visual analog scale (VAS), the pain score of the treated group reduced over the weeks of supplementation. Another point to be considered is that vitamin D deficiency causes osteomalacia, a painful bone disease, which can often be confused or misdiagnosed as fibromyalgia (Holick 2003, Plotnikoff & Quigley 2003).
In rheumatic diseases , such as rheumatoid arthritis or osteoarthritis, the results for the inverse correlation between vitamin D and pain complaints are in the same direction (Table 2) (Gopinath & Danda 2011, Higgins et al. 2013, Abourazzak et al. 2015, Yang et al. 2015). These disorders are also characterized as autoimmune diseases. For this reason, another hypothesis suggests that pain relief in these patients could be modulated by the action of 1,25(OH)2D in the immune system and not only in the pain-regulating mechanism. Vitamin D also presents a potential immunomodulatory response in autoimmune diseases. A prospective study, including men and women with an age range of 18-71 years, reported that an elevation of 10 nmol/L on vitamin D status was associated with lower incidence of diseases such as multiple sclerosis, type 1 diabetes, Crohn's disease and thyrotoxicosis (Skaaby et al. 2015). It is known that 1,25(OH)2D modulates some actions of the immune system. It suppresses the proinflammatory response (Th1) and stimulates the anti-inflammatory response (Th2), which leads to an immunological tolerance and consequently attenuates the clinical condition of the autoimmune disease (Kamen & Tangpricha 2010).
Other comorbidities can also cause chronic pain. In young individuals with sickle cell disease, both pain and increased bone frailty were associated with serum 25OHD levels <20 ng/mL (Table 2) (Osunkwo et al. 2011). In individuals under treatment for cancer, serum 25OHD levels were related to the use of opioids. Low levels of 25OHD (<20 ng/mL) led to higher doses of opioids for pain relief, while high serum 25OHD levels were a predictor of longevity (Table 2) (Bergman et al. 2015).
Sleep and pain
Changes in sleep pattern are common in the population and contribute to several impairments of the physiological system, leading to increased cardiovascular risk, reduction in immune defense, cognitive impairment and increased pain sensitivity (Meier-Ewert et al. 2004, Durmer & Dinges 2005, Nascimento et al. 2007, Zager et al. 2007, Roizenblatt et al. 2015).
The use of animal models has provided a better understanding of the effects of sleep on pain and vice- versa, mimicking the physiological and behavioral manifestations observed in humans (Andersen et al. 2006). In rats with chronic constrictive injury, lower sleep efficiency, a higher number of awakenings and longer sleep latency were observed, as well as a reduction in NREM sleep (Andersen & Tufik 2003). These results showed that pain was associated with intrinsic changes in sleep architecture. These findings have been confirmed in humans. A survey evaluating the impact of dysmenorrhea on sleep profile showed that women who had menstrual cramps presented longer REM sleep latency (Araujo et al. 2011). Changes in sleep pattern have also been found in women with fibromyalgia, who have shown intrusion of high-frequency brainwaves during NREM sleep in association with hypersensitivity and increased subjective pain (Smith et al. 2009). The reduction in both sleep efficiency and slow wave sleep are associated with fatigue in this disease. The treatment of pain conditions may be helpful for sleep quality. A randomized placebo-controlled study evaluated the efficacy of pregabalin use, a drug for fibromyalgia treatment, on the management of pain (Arnold et al. 2015). Women were more prevalent in this cohort (93%), and the participants were taking antidepressant medication early in the study, such as selective serotonin reuptake inhibitor (SSRI) or serotonin/norepinephrine reuptake inhibitor (SNRI). The results showed that the treated group had a decrease in mean pain score at the end of the study, and sleep quality was improved compared to placebo.
Sleep changes can also trigger painful conditions. Acute reduction of sleep time in healthy pain-free individuals caused hyperalgesia in the following morning (Roehrs et al. 2006). This study also found that REM sleep deprivation decreased the finger withdrawal latency for pain sensation compared to individuals that had 8 h of normal sleep. A meta-analysis corroborates these findings, revealing that sleep deprivation impairs pain perception (Schrimpf et al. 2015). Positive insomnia symptoms, at baseline assessment, were predictive of developing a painful condition (Finan et al. 2013). Insomnia frequency and severity among men and women ranging from 30 to 87 years old are also related to pain sensitivity. Sivertsen and coworkers (2015) demonstrated a higher hazard ratio (52%) to painful conditions among individuals from both sexes who reported insomnia more than once a week. Just as it has been shown that decreasing pain improves sleep, it has also been demonstrated that a restorative sleep is a predictor of pain resolution. Davies and coworkers (2008), assessing chronic widespread pain and sleep quality through questionnaires, reported that restorative sleep was associated with chronic pain resolution among younger participants compared to individuals with chronic persistent pain. After controlling for confounders, restorative sleep remained as the sleep component associated with pain resolution.
In general, pain can cause changes in sleep pattern resulting in a non-restorative, superficial and fragmented sleep, with longer onset latency. Some painful conditions are frequently studied regarding sleep disorders including fibromyalgia, rheumatoid arthritis, low back pain, systemic lupus erythematosus, as well as those associated with comorbidities, such as dysmenorrhea, tumor, and cardiac or neurological diseases. A prospective study showed that participants (~55% women) with sleep complaints had an increased probability (from 14% to 38%) of developing chronic pain 3 years later (Bonvanie et al. 2016). Although many researchers support the hypothesis of a bidirectional relationship between sleep and pain, others suggest a unidirectional association, in which alterations in the sleep pattern predict the occurrence of pain (Finan et al. 2013). Both longitudinal and cross-sectional studies have shown that sleep problems are a preponderant factor for the development of pain.
Sleep and pain share neurotransmitters in their physiological processes. Compared to wakefulness, during normal sleep the release of serotonin is decreased; under conditions of sleep deprivation, its release is further reduced (Blanco-Centurion & Salin-Pascual 2001, Roehrs et al. 2006), which contributes to nociceptive pain stimulation. Thus, the use of drugs, such as SSRI, SNRI or tricyclic antidepressants, can improve both sleep and pain problems as its mechanism of action inhibits serotonin reuptake, resulting in elevated extracellular levels of this neurotransmitter.
Pain is also present in individuals with OSA. In a crosssectional study with individuals of both sexes, morning headaches were more frequent in OSA patients compared to those without OSA (Kristiansen et al. 2011). In a prospective study, Johnson and coworkers (2012) reported that headaches in individuals with OSA improved after CPAP treatment. Unfortunately, the duration of CPAP treatment in this study was not clear.
Future perspectives: vitamin D, sleep and pain
Based on the literature, vitamin D status seems to have an important role in the bidirectional relationship observed between sleep and pain. A study evaluating the effects of cholecalciferol supplementation (1200 I U/ day to individuals with serum 25OHD between 20 and 29 ng/mL and 50,000 IU/weekly to those with serum 25OHD <20 ng/mL) in veterans with multiple areas of chronic pain found a decrease in self-reported pain in both supplemented groups, as well as an improvement in sleep pattern, with decreased sleep latency and increased sleep duration (Huang et al. 2013). In another study, patients with nonspecific chronic pain selected from a sleep clinic showed serum 25OHD levels <20 ng/mL (McCarty et al. 2013). The risk factors for the decrease of vitamin D status in this study were ethnicity, obesity and age. In patients with hyperparathyroidism supplemented with vitamin D after parathyroidectomy, a decrease in RLS symptoms (from 56.2% to 21.0%) and pain relief were observed (Santos et al. 2016). Although it resulted in an increase in serum 25OHD levels, there was no significant correlation between 25OHD levels and RLS severity. xxxx
The data presented in Tables 1 and 2 highlight the most relevant studies linking the relationship between vitamin D, pain and sleep. The different study designs and cutoffs of vitamin D, as well as the distinct vitamin D supplementation protocols, make the establishment of adequate doses of vitamin D still difficult. Nevertheless, this review brings important information regarding the benefits of vitamin D for pain-related conditions and sleep disturbances. Thus, we can hypothesize that suitable vitamin D supplementation combined with sleep hygiene may optimize the therapeutic management of pain-related diseases such as fibromyalgia (Fig. 1). As 1,25(OH)2D stimulates an anti-inflammatory immune response, this mechanism may induce an antinociceptive action. Sleep hygiene consists of several interventions to improve sleep, i.e. changes in the habits that precede sleep, such as avoiding the consumption of alcoholic and caffeinated drinks; reducing lighting in the bedroom and use of electronic appliances; and maintaining regular sleep schedules. For individuals with insomnia, pharmacological treatment combined with other techniques such as cognitive behavioral therapy is helpful in promoting sleep quality and reducing pain sensitivity (Finan et al. 2014). Finally, OSA treatment with CPAP can improve both pain perception and 25OHD levels. These data indicate the importance of considering the sleep- pain-vitamin D inter-relationship during anamnesis by clinicians, sleep physicians and neurologists, so that the best intervention is chosen to provide the patient with a better quality of life.
