Colon Cancer survival increased by MEG3 (MEG3 is increased by Vitamin D in lab) – 2018
MEG3 Activated by Vitamin D Inhibits Colorectal Cancer Cells Proliferation and Migration via Regulating Clusterin
ebiomedicine.com March(?) 2018 DOI: https://doi.org/10.1016/j.ebiom.2018.03.032
📄 Download the PDF from VitaminDWiki
High level of MEG3 - (MEG3 is increased by Vitamin D)
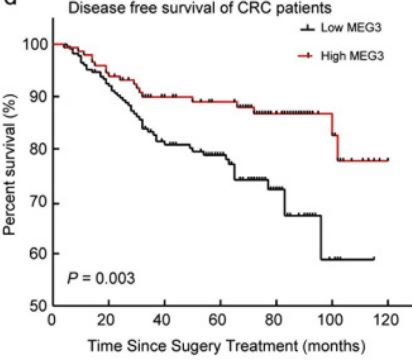
Note: The study does not seem to indicate by how much Vitamin D increases MEG3
Highlights
MEG3 serves as a novel CRC prognosis biomarker and a potential therapeutic target.
MEG3 over-expression represses CRC cells proliferation and metastatic features.
MEG3 has a role in the down-regulation of Clusterin expression and activity at transcriptional and post-transcriptional levels.
VDR activated MEG3 expression via directly binding to MEG3 promoter.
The long non-coding RNA maternally expressed gene 3 (MEG3) is frequently dysregulated in human cancers ; however, its roles in colorectal cancer (CRC) development are largely unknown. Here, we reported that MEG3 was down-regulated in CRC tissues and CRC patients with lower MEG3 showed poorer overall survival and disease-free survival than those with higher MEG3 level. MEG3 over-expression represses CRC cells proliferation and migration in vivo and in vitro, while MEG3 knockdown leads to the enhanced proliferation and metastasis of CRC cells. In CRC cells, MEG3 over-expression is related to decreased Clusterin mRNA and the corresponding protein levels, and it also directly binds to Clusterin protein through its 732–1174 region. In further, Clusterin over-expression rescues the compromised abilities of proliferation and metastasis induced by MEG3 over-expression, suggesting that MEG3 inhibits the CRC progression through regulating the Clusterin activities.
Additionally, we found that 1α,25-(OH)2D and vitamin D receptor (VDR) stimulate MEG3 expression in CRC cells through directly binding to its promoter. These results suggested that MEG3 functions as a tumor suppressor in CRC via regulating the Clusterin activities and may underlie the anticancer activities of vitamin D on CRC cells. The VDR/MEG3/Clusterin signaling pathway may serve as potential therapeutic targets and prognosis biomarkers for CRC patients in future.
