5 Critical Analyses of the 2024 Endocrine Society Guideline on Vitamin D
Summary
Pilz et al. (Nutrients, May 2026) provide a comprehensive summary and critical appraisal of the 2024 Endocrine Society Clinical Practice Guideline on vitamin D (Demay et al., JCEM 2024). The Endocrine Society guideline — built on GRADE + Evidence-to-Decision methodology with RCT priority — recommends empiric vitamin D supplementation (above DRI, without 25(OH)D testing) for four populations only:
| Population | Indication | Trial weighted avg | Trial range |
|---|---|---|---|
| Ages 1–18 | Rickets, RTIs | ~1,200 IU/d | 300–2,000 IU |
| Ages ≥75 | Mortality | ~900 IU/d | 400–3,333 IU |
| Pregnancy | Preeclampsia, IUM, preterm, SGA, neonatal mortality | 2,500 IU/d | 600–5,000 IU |
| Prediabetes (high-risk) | T2DM progression | ~3,500 IU/d | 842–7,543 IU |
Key methodological positions of the guideline: against routine 25(OH)D screening (including in dark skin and obesity); 30 ng/mL threshold from 2011 abandoned; daily over intermittent dosing for ≥50; healthy 50–74 may rationally choose to supplement; and — per a 2025 correction — the 2024 guideline replaces the 2011 guideline.
Pilz et al. organize their critique into six domains: dose uncertainty, target-population gaps, implementation, laboratory testing, new evidence, and outlook (sun/UV-B).
Where Pilz et al. agree with other publications
- Holick 2024, 2025 (Endocr Pract): Both Pilz and Holick flag that retracting the 2011 guideline leaves populations (malabsorption, CKD, low measured 25(OH)D, established deficiency) without guidance. Pilz endorses Holick's suggestion to continue applying 2011 recommendations to those populations.
- Pludowski et al. 2024 (Nutrients): Pilz explicitly endorses 2,000 IU/day for adults as their expert-opinion default — directly aligning with Pludowski's review.
- Grant, Wimalawansa, Pludowski, Cheng 2025 (Nutrients) and Wimalawansa et al. 2024 (Nutrients): Shared view that Endocrine Society under-recognizes extra-skeletal benefits and that population-level recommendations need broader scope.
- Riedmann et al. 2025 / Alfredsson et al. 2020: Pilz frames sunlight/UV-B as the "elephant in the room" — agreeing with the growing literature that insufficient sun exposure is itself a public health problem and that UV confers non-vitamin-D-mediated benefits (mortality reductions in UK Biobank, Adventist Health Study 2).
- USPSTF / Choosing Wisely: Pilz accepts the broad anti-screening position but argues it's incomplete.
- Munns et al. 2016 (Global Consensus on Nutritional Rickets): Endorsed as the operative reference for infants <1 year.
Where Pilz et al. disagree or push back
- Against the Endocrine Society guideline itself — primary disagreements:
- 1. Dose vagueness. Stating "optimal doses are unknown" without providing a practical range is, per Pilz, an abdication. They explicitly endorse 2,000 IU/d as within the trial range for all four indications and safe.
- 2. No bridging for measured 25(OH)D. Real-world patients arrive with test results; the guideline gives no advice on interpreting them, including for values approaching the IOM 125 nmol/L "concern" threshold. Pilz proposes raising it to 150 nmol/L given 15+ years of new safety data (Billington 2020, Zittermann 2023, Cianferotti 2026, Malihi 2019).
3. Implementation void. The guideline implicitly recommends supplementation for ~50% of the US population but provides no implementation pathway.
Tension with Bolland 2021 / Breth-Petersen 2022: Those papers argue testing is wasteful; Pilz pushes for more nuanced guidance rather than blanket testing rejection.
Tension with newer evidence (which Pilz interprets generously toward the guideline):
- Jolliffe et al. 2025 (Lancet Diabetes Endocrinol) updated RTI meta-analysis lost overall statistical significance (OR 0.94, 95% CI 0.88–1.00); pediatric effect (1–15 y) preserved (OR 0.74, 0.60–0.92). Pilz judges this doesn't undermine the pediatric RTI recommendation but it weakens the all-ages story.
- Palacios et al. 2024 (Cochrane) pregnancy update removed 20 of 30 prior trials for trustworthiness concerns; significant outcomes contracted to severe postpartum hemorrhage and low birth weight. Pilz argues recommendations still hold; a stricter reader would say the evidence base has thinned.
- Thouvenot et al. 2025 (D-Lay MS, JAMA) — 100,000 IU q2wk in CIS reduced disease activity, also a safety signal at high intermittent doses, in some tension with the guideline's caution about ≥100,000 IU boluses.
Implicit disagreement with the more skeptical commentators (Pittas 2025 "In the Shadow of Enthusiasm"; the panel's own conservative posture): Pilz et al. are more pro-supplementation than the guideline authors. They cite Nwosu 2025 pediatric rebuttal but don't fully engage it.
Notable research gaps the authors flag
- No defined upper dose limit for empiric supplementation.
- No guidance for the ~10% (Switzerland data) to ~20% of populations who have been tested.
- Whether the IOM 125 nmol/L concern threshold should be revised upward.
- UV/sun exposure entirely absent from vitamin D guidelines globally.
- Implementation data — is the guideline actually changing practice?
The authors' AGREE II appraisal is in Supplementary Table S1 (not in the main text). Overall posture: methodologically rigorous guideline, but the conservative scope and silence on dose, testing, and replaced populations leave the practicing clinician without operational answers — a critique consistent with the broader European/Pludowski/Holick/Grant camp and at odds with the more minimalist US Endocrine Society/USPSTF position.
2024 Endocrine Society Guideline.pdf
Editorial of 5+ studies that disliked the 2024 guideline
Editorial: Impact of new vitamin D guidelines on pediatric and adult health: insights, evidence, and implications Editorial Front Endocrinol (Lausanne). 2026 Apr 29:17 doi: 10.3389/fendo.2026.1851754.
Clipped from 2-page PDF
"In a rebuttal to the 2024 Guideline (1), Nwosu rejects the sweeping one-size-fits-all recommendations, and the opaque recommendations for infants, children, adolescents, obese individuals,darkly pigmented individuals, pregnant women and fetuses. This rebuttal calls for routine screening, supplementation, and monitoring of serum 25(OH)D concentrations to ensure that everyone benefits from the skeletal and extra-skeletal actions of vitamin D."
"In conclusion, this Research Topic employed evidence-based and real-world research to examine the 2024 Endocrine Society Guideline on Vitamin D for Prevention of Disease by focusing on the role of vitamin D deficiency on childhood hypocalcemia; the health impact of disparate cut-off points for vitamin D status designation by different medical societies; the impact of high-dose vitamin D supplementation on T1D, T2D, and complete heart block; the negative impact of vitamin D deficiency in asthma, thyroid cancer, and diabetes mellitus. There is also the exciting possibility that transient overexpression of SDR42E1 gene can reverse these vitamin D deficiency states. These innovative works will spur further research in this field, and will shape future recommendations for vitamin D screening, monitoring, and supplementation."
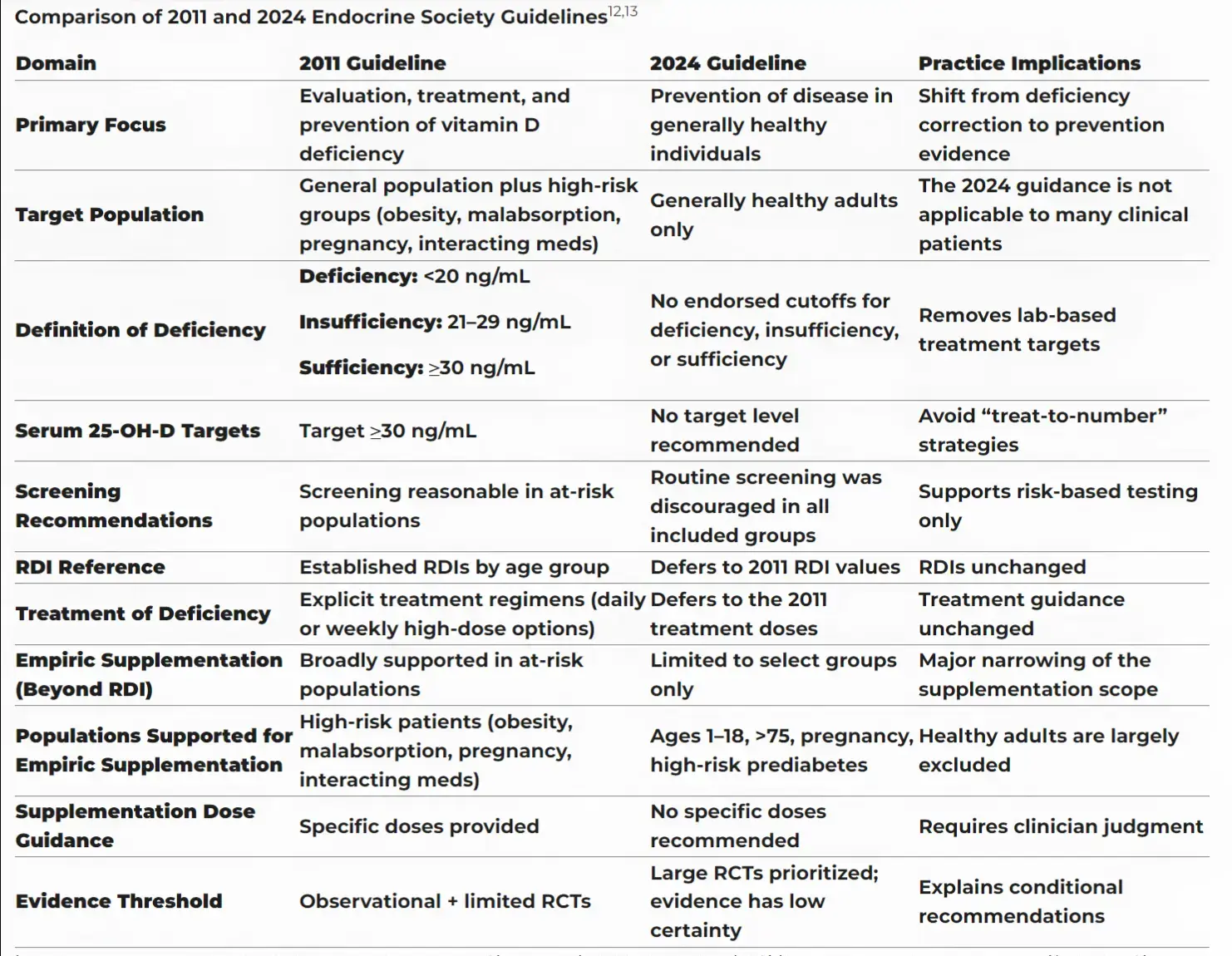
A Continuing Medical Education compares 2011 and 2024
Related in VitaminDWiki
- US admits math mistake was made in 2010 in estimating Vitamin D, but will not change recommendations
- Consensus Vitamin D
- Vitamin D supplementation guidelines (adults – 50,000 IU per week)
- One pill every two weeks gives you all the vitamin D most adults need
- Vitamin D: 40-70 ng is optimal, sometimes more is needed - Grant, Sunil, Pawel, Cheng
- Comparing High-dose vitamin D therapies - may succeesful trials use cofactors and personalized dosing
- Vitamin D Cofactors in a nutshell
Other critiques of 2024 Endocrine Society
