Better sleep somewhat associated with higher vitamin D
Association of serum 25-hydroxyvitamin D concentrations with sleep phenotypes in a German community sample
PLOS x July 5, 2019https://doi.org/10.1371/journal.pone.0219318
Ezgi Dogan-Sander , Anja Willenberg, İnci Batmaz, Cornelia Enzenbach, Kerstin Wirkner, Elisabeth Kohls, Roland Mergl, Joachim Thiery, Jürgen Kratzsch, Ulrich Hegerl, Christian Sander
📄 Download the PDF from VitaminDWiki
Background
Sleep disorders and vitamin D deficiency are among the most common health problems. Few studies investigated the effect of vitamin D on objectively recorded sleep with sound methodological quality and reasonable temporal proximity.
Objective
To investigate the relationship between serum 25-hydroxyvitamin D (25(OH)D) concentrations and objective sleep parameters assessed within close temporal proximity in a population-based sample. It is expected that higher serum 25(OH)D concentrations are associated with 1) better objective sleep outcomes (longer sleep duration, higher sleep efficiency, earlier mid-sleep time) and 2) more positive subjective sleep evaluations.
Methods
A subset of participants (n = 1045) from the LIFE-Adult-Study was analysed. Measurement of serum 25(OH)D vitamin was performed using an electrochemiluminescence immunoassay. Actigraphic assessments were performed using SenseWear Pro 3 devices. The following objective sleep parameters were calculated: total sleep duration, night sleep duration, night sleep efficiency, midsleep time and wake after sleep onset (WASO). Subjective sleep evaluations were assessed via questionnaire (sleep quality (PSQI), daytime sleepiness (ESS)). Data were analysed applying a multiple linear regression model with a stepwise approach.
Results
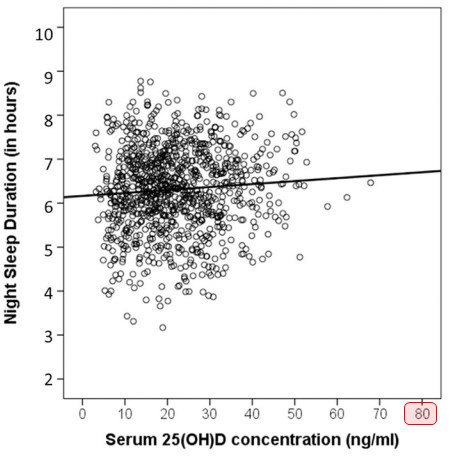
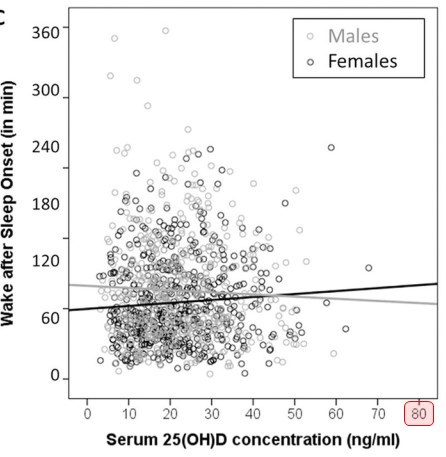
The regression models revealed significant associations of serum 25(OH)D concentration with night sleep duration and midsleep time. No association was found for total sleep duration and night sleep efficiency. Higher serum 25(OH)D concentration was further associated with shorter WASO in males but longer WASO in females. Moreover, serum 25(OH)D concentration did not show any significant association with subjective sleep quality and daytime sleepiness.
Conclusion
The results indicate that a higher concentration of serum 25(OH)D is associated with longer and earlier night sleep. Although the present study was able to demonstrate an association between serum 25(OH)D concentration and objective sleep parameters, no conclusion about underlying mechanisms or causal inferences can be drawn.
First portion of the PDF
Vitamin D is being considered more than just a steroid hormone which plays a central role in calcium-phosphorus and bone homeostasis [1,2]. Recently, other effects of vitamin D on different medical conditions such as cardiovascular diseases, autoimmune diseases, depression, diabetes mellitus and obesity have been demonstrated in several studies [3,4,1,5,6]. Locating several extra-skeletal vitamin D receptors in most tissues and cells (e.g., brain, skin) also supports the hypotheses regarding these effects [7,8]. Additionally, vitamin D is found to take a part in brain development and function [9]. Against the background of the wide spread vitamin D deficiency across Europe [10] investigations on the associations between serum vitamin D concentration as well as vitamin D intake and medical conditions are rapidly emerging.
It is hypothesized that vitamin D has a potential role in the regulation of sleep [11,12]. Sleep disorders are also widely common [13]: The prevalence of sleep disorders ranges from 25% to 32.1% depending on study population and assessment methods [14–16]. Sleep disorders were also considered to play a role in the aetiology of many other disorders such as hypertension [17], heart diseases [18] and diabetes [19]. Furthermore, it has been shown that adults with shorter sleep duration exhibit more health risk factors, e.g. obesity, physical inactivity, smoking and alcohol consumption [20]. Since 2009 there has been an increase regarding studies focusing on the relationship between serum vitamin D concentration (as well as vitamin D intake) and different sleep parameters [21]. For instance, a positive association between serum 25-hydroxyvitamin D (25(OH)D) concentrations and self-reported daily sleep duration was demonstrated among elderly adults [22]. Contrary, Shiue (2013) found no relationship between sleep duration and serum 25(OH)D concentrations; but reported a significant inverse association between sleep onset latency (minutes to fall asleep) and serum 25(OH)D concentrations [23]. Additionally, an inverse correlation between Epworth Sleepiness Scale (ESS) scores, indicating daytime sleepiness, and serum 25(OH)D concentration was reported for patients without vitamin D deficiency (25(OH)D >20 ng/mL) but no correlation between ESS scores and serum 25(OH)D concentrations was found in patients with actual vitamin D deficiency [24]. Surprisingly, a direct correlation between ESS scores and serum 25(OH)D levels was reported in black patients with vitamin D deficiency. Beydoun et al. (2014) [25] also reported a significant inverse relationship between serum 25(OH)D concentrations and a factor analytically derived sub-scale of a sleep questionnaire reflecting sleepiness. However, only a marginal negative association was reported between serum 25(OH)D concentrations and very short sleep, when a sub-group with very short sleep duration (<5 hours per night) was compared to a sub-group with normal sleep duration (7–8 hours per night). Among Korean workers an odds ratio of 1.43 for having poor sleep quality (PSQI (Pittsburgh Sleep Quality Index) score<5) was found in case of vitamin D deficiency (<10 ng/mL) [26].
In the current literature there are only few studies using objective sleep parameters (measured by actigraphy or polysomnography) to investigate the relationship between serum 25(OH)D concentrations as well as vitamin D intake and sleep parameters. In a study by Grandner et al. [27] the association between vitamin D intake and subjective as well as objective sleep parameters in postmenopausal women was investigated, resulting in positive correlations between vitamin D intake and sleep acrophase (“the peak of a fitted 24-hour cosine”) as well as self-reported number of naps. No significant correlations were reported between vitamin D intake and total sleep time, sleep efficiency and duration of naps.
Some of the References
1. Caprio M, Infante M, Calanchini M, Mammi C, Fabbri A. Vitamin D: not just the bone. Evidence for beneficial pleiotropic extraskeletal effects. Eat Weight Disord. 2017;22(1): 27–41. pmid:27553017
2. Eyles DW, Burne THJ, McGrath JJ. Vitamin D, effects on brain development, adult brain function and the links between low levels of vitamin D and neuropsychiatric disease. Front Neuroendocrinol. 2013;34(1): 47–64. pmid:22796576
3. Imai CM, Halldorsson TI, Eiriksdottir G, Cotch MF, Steingrimsdottir L, Thorsdottir I, et al. Depression and serum 25-hydroxyvitamin D in older adults living at northern latitudes–AGES-Reykjavik Study. J Nutr Sci 2015;4: e37. pmid:26688723
4. Papandreou D, Hamid Z. The Role of Vitamin D in Diabetes and Cardiovascular Disease: An Updated Review of the Literature. Dis Markers 2015: 580474. pmid:26576069
5. Zhang Y, Zhang X, Wang F, Zhang W, Wang C, Yu C et al. The relationship between obesity indices and serum vitamin D levels in Chinese adults from urban settings. Asia Pac J Clin Nutr. 2016;25(2): 333–339. pmid:27222417
6. Milaneschi Y, Hoogendijk W, Lips P, Heijboer AC, Schoevers R, van Hemert AM, et al. The association between low vitamin D and depressive disorders. Mol Psychiatry. 2014;19(4): 444–451. pmid:23568194
7. Eyles DW, Smith S, Kinobe R, Hewison M, McGrath JJ. Distribution of the Vitamin D receptor and 1α-hydroxylase in human brain. J Chem Neuroanat. 2005;29(1): 21–30. pmid:15589699
8. Christakos S, Dhawan P, Verstuyf A, Verlinden L, Carmeliet G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol Rev, 2016;96(1): 365–408. pmid:26681795
9. Cui X, Gooch H, Petty A, McGrath JJ, Eyles D. Vitamin D and the brain: Genomic and non-genomic actions. Mol Cell Endocrinol. 2017;453: 131–143. pmid:28579120
10. Cashman KD, Dowling KG, Skrabakova Z, Gonzalez-Gross M, Valtuena J, De Henauw S, et al. Vitamin D deficiency in Europe: pandemic?. Am J Clin Nutr. 2016;103(4): 1033–1044. pmid:26864360
11. Gominak SC, Stumpf WE. The world epidemic of sleep disorders is linked to vitamin D deficiency. Med Hypotheses. 2012;79(2): 132–135. pmid:22583560
12. McCarty DE, Chesson AL, Jain SK, Marino AA. The link between vitamin D metabolism and sleep medicine. Sleep Med Rev. 2014;18(4): 311–319. pmid:24075129
13. Colten HR, Altevogt BM. Sleep disorders and sleep deprivation: An unmet public health problem. Wachington, D.C.: National Academies Press; 2006.
14. Schmeiser-Rieder A, Kapfhammer G, Bolitschek J, Holzinger B, Skrobal A, Kunze M, et al. Self reported prevalence and treatment of sleep disorders in Austria. J Epidemiol Community Health. 1995;49(6): 645–646. pmid:8596103
15. WHO Regional Office for Europe. WHO technical meeting on sleep and health. 2004. Available from: http://www.euro.who.int/_data/assets/pdffile/0008/114101/E84683.pdf.
16. Kerkhof GA (2017) Epidemiology of sleep and sleep disorders in The Netherlands. Sleep Med. 2017;30: 229–239. pmid:28215254
17. Bansil P, Kuklina EV, Merritt RK, Yoon PW. Associations between sleep disorders, sleep duration, quality of sleep, and hypertension: results from the National Health and Nutrition Examination Survey, 2005 to 2008. J Clin Hypertens (Greenwich). 2011;13(10): 739–743.
18. Budhiraja R, Roth T, Hudgel DW, Budhiraja P, Drake CL. Prevalence and polysomnographic correlates of insomnia comorbid with medical disorders. Sleep. 2011;34(7): 859–867. pmid:21731135
19. Knutson KL, van Cauter E. Associations between sleep loss and increased risk of obesity and diabetes. Ann N Y Acad Sci. 2008;1129: 287–304. pmid:18591489
