Indian Pediatr 2010;47: 581-586

|
Hypovitaminosis D and Hypocalcemic Seizures in Infancy |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
From the Department of Pediatrics, Kalawati Saran Hospital, New Delhi; *Department of Endocrinology and Thyroid Research Centre, Institute of Nuclear Medicine and Allied Sciences, Delhi; and #Department of Endocrinology and Metabolism, All India Institute of Medical Sciences, New Delhi, India.
Correspondence to: Dr Raman K Marwaha, Department of Endocrinology and Thyroid Research Centre,
Received: May 9, 2008;
PII: S097475590800286-1 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Abstract
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Reports have indicated that there is a high prevalence of hypovitaminosis D in India, parti-cularly amongst pregnant and lactating women(8-10). We conducted this study to evaluate the role of vitamin D deficiency in the etiology of symptomatic hypocalcemia during infancy. Methods A total of 60 consecutive infants, 15 days to 6 months of age, presenting with hypocalcemic seizures (Study infants) to the pediatric emergency ward of a tertiary level children’s hospital, on two days a week, were recruited for the study along with their mothers (Study mothers), between April 2006 and March 2007. Hypocalcemia was considered to be the cause of seizures if total serum calcium level was <8mg/dL or ionized calcium level was <4mg/dL, with normal levels of serum albumin. A similar number of age and socioeconomically matched breastfed infants (Con-trol infants) and their mothers (Control mothers), attending an immunization clinic were taken as controls. The study was approved by the Ethics Committee of the Hospital. Written informed consent was obtained from the mothers enrolled in the study. Only infants from full term singleton deliveries, without congenital malformations were chosen as subjects for the study. Babies were considered full term based on a review of history obtained from the mother, corroborated by hospital records when available. Exclusion criteria included birthweight £2 kg and history of intake of drugs or supplements known to affect bone mineral metabolism. Mothers known to have hepatic, renal or bone disorders, malabsorption or intake of any drugs/supplements known to affect the calcium -vitamin D - PTH axis were also excluded from the study. The selected mother-infant pairs underwent concurrent clinical, radiological (only for study infants), biochemical and hormonal assessment on the first visit as described below. Dietary assessment of total calories, protein, carbohydrate, fat, calcium, fiber and phytate was done in study and control mothers by a 24 h recall method as per published guidelines(11). Sun exposure was quantified by calculating the UV score for the mothers. This was done by assessing the mean body surface area (m2) (using rule of 9) exposed to the sun and the mean duration (min/day) of sunlight exposure in a day between 9AM to 4PM. UV score (min.m2/day) was calculated by multiplying the above two parameters(12). Blood was drawn by venepuncture for total and ionic calcium (Ca), inorganic phosphate (P), alkaline phosphatase (ALP), magnesium (Mg), vitamin D (25(OH)D) and intact parathormone (iPTH) from both mothers and infants. Routine investigations to exclude other causes of seizures in infants were also performed. Serum was separated in a cold centrifuge and supernatant removed in three aliquots. While serum Ca, P, ALP were estimated on the same day, the remaining aliquots were stored at –80º C until 25(OH) D and iPTH were estimated. Radiographs of the wrist joint were done to look for radiological evidence of rickets in hypocalcaemic infants. Hormone assays were performed at the Institute of Nuclear Medicine and Allied Sciences (INMAS). The serum concentration of 25(OH) D (reference range 9.0-37.6 ng/mL) was measured by RIA (Diasorin, Stillwater, MN). Analytical sensitivity of the kit was 1.5ng/mL. We classified hypovitaminosis D based on the measurement of serum 25 (OH)D concentration(13), as follows: mild hypovitaminosis D: 10-20 ng/mL; moderate hypovitaminosis D: 5-10 ng/mL; and severe hypovitaminosis D: <5 ng/mL. However, in view of many earlier studies taking 10ng/mL as the cut-off for vitamin D deficiency, we have also discussed our results based on this value. Serum iPTH (reference range 13-54 ng/L) was measured using IRMA (Diasorin, Stillwater, MN). Analytical sensitivity of the kit was 0.7 ng/L. Serum ionized calcium was estimated by ion-exchange method (Electrolyte Analyzer, Roche, Mannheim, Germany). The reference range for ionized calcium was 4-5 mg/dL (infants 10 days-2 yrs) and 4.7-5.2 mg/dL (adult women). Total serum calcium was measured by colorimetric method and inorganic phosphate and alkaline phosphatase were measured by photometric method (Randox Lab Ltd, UK). The reference range for total calcium was 8.4-10.8 mg/dL (infants 10 days-2 yrs) and 8.8-10.2 mg/dL (adult women). Serum albumin was measured using bromo-cresol green (BCG) dye method. The normal range of inorganic phosphate was 3.0 to 7.0mg/dL (infants) and 2.7 to 4.5 mg/dL (adults). The upper limit of normal for ALP in infants was 1076 IU/L while that in non-pregnant women was 240 IU/L. Serum magnesium was measured by colorimetric method (Bio-chemistry Analyzer, Roche, Mannheim, Germany) and the normal range was 1.5-2.7 mg/dL. Statistical analysis was carried out using SPSS 11.0. Independent t test was used to compare difference of means between the study and control groups. Spearman correlation analysis was perfor-med to examine the relation of variables between infants and their respective mothers. Logistic and linear regression analysis was used wherever necessary. Results The mean age of study and control infants was 3.0 ± 0.16 and 3.0 ± 0.14 months, respectively, with male to female ratio of 1.14:1 and 0.66: 1. Among study infants, 41 (68.3%) were exclusively breastfed, 9 (15%) were supplemented with animal milk and 10 (16.7%) were exclusively on animal milk. The control infants were predominantly breastfed with some of them receiving water or 1-2 animal milk feeds along with breast feeds. Associated infections were demonstrable in 29 (48.3%) study infants lower respiratory tract infection in 15 (25%), acute gastroenteritis in 11 (18.3%) and sepsis in 3 (5%). Subtle signs of vitamin D deficiency in the form of wide anterior fontanel (>2.5 × 2.5 cm) and craniotabes were present in 8 (13%), while radiological evidence (metaphyseal fraying and osteopenia) was observed in 18 (30%) of study infants. There was no clinical evidence of rickets in any of the control infants. TABLE I Anthropometry and Dietary Characteristics of Study and Control Mothers
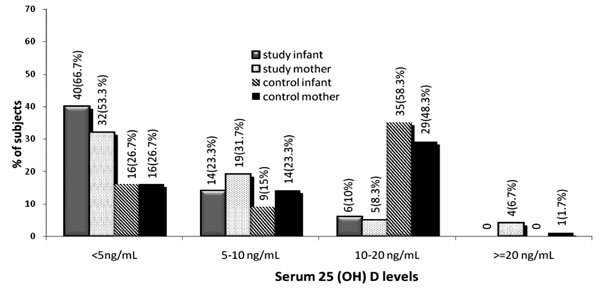
Dietary evaluation revealed gross differences in the nutritional status and sun exposure of study and control mothers (Table I).The mean/median serum values of total and ionic Ca, inorganic phosphate, ALP, 25(OH) D and iPTH in mother – infant pairs are shown in Table II. The distribution of serum 25(OH)D levels in mother-infant pairs is shown in Fig.1. Elevated serum ALP was observed in a significantly higher proportion of study infants (72%) as compared to study mothers (6.1%) (P <0.001). A significant inverse correlation between 25 (OH) D and PTH was noted in both study (r = –0.22, P<0.04) and control (r = –0.72, P<0.0001) mothers. An inverse correlation between 25 (OH) D and ALP was seen only in study mothers (r = –0.042, P = 0.0008). No significant correlation was noted between 25 (OH) D and calcium in either group. TABLE II Biochemical Parameters in Infant-Mother Pairs
Among study infants with 25 (OH) D <10ng/mL, 75% had raised PTH in contrast to only 3.1% infants with 25(OH) D>10ng/mL. The mean PTH level in infants with serum 25(OH)D level <10 ng/mL was significantly higher than those with levels >10ng/mL (94.2 ± 78.3 pg/mL vs 28.9 ± 11.6 pg/mL, P=0.001). No significant correlation was found between serum 25(OH) D and PTH, calcium, or ALP in study infants. In control infants, a significant inverse correlation was observed between 25(OH)D and PTH (r = –0.65, P <0.0001). Hypomagnesemia was noted in 4 infants with seizures and none of the controls. Seizures in these infants responded only when magnesium therapy was instituted. A strong positive correlation was noted between serum 25(OH)D levels of mother-infant pairs in both study (r = 0.64; P<0.0001) and control populations (r = 0.39; P<0.002). However, no such correlation was seen for serum calcium. Of the 54 study infants who had 25(OH)D levels <10ng/mL, 48 (89%) were born to mothers who also had 25(OH) D <10ng/mL. Using logistic regression, infants born to mothers with 25 (OH) D <10 ng/mL had a 40 times increased risk of hypovitaminosis D when compared to those born to mothers with 25(OH) D levels ³10 ng/mL. Discussion Vitamin D deficiency continues to be a public health problem prevalent in many Asian countries, including India, despite abundant sunlight (8,9,14). Results of the present study demonstrate that majority of mother-infant pairs from our population were vitamin D deficient with mean levels of 25(OH)D being <10ng/mL. Using Lips criteria(13), all study and control infants were having vitamin D deficiency. However, even if a 25(OH)D level of <10ng/mL was taken as the cut off value, as reported in some earlier studies(4,6), 90% of the study and 41.7% of control infants were still affected. The seasonal impact on 25(OH)D levels in children was not evaluated because, during early infancy, most infants are kept indoors and covered due to prevailing sociocultural beliefs. Balasubramanian, et al.(3), have reported that all 13 exclusively breast-fed infants with hypo-calcemic seizures had low serum 25(OH)D (mean 3.8 ± 2.08 ng/mL) and elevated PTH (106 ± 25.4 pg/mL). In a similar study conducted in Pakistan, in 65 infants presenting with hypocalcemic seizures, all the 15 mother-infant pairs in whom 25 (OH)D was estimated had levels <10ng/mL(3). Evaluating medical records of infants with a diagnosis of vitamin D deficiency and/or nutritional rickets, Hatun, et al.(15) reported that 79% had presented with seizures. A large number of reports are available in the literature highlighting the high prevalence of 25(OH) D deficiency in women of childbearing age(16-21), during pregnancy(10,22-24), and lactation(15,25, 26), resulting in adverse effects in women, fetus, infants and children(22). Two earlier studies from India have also highlighted the high prevalence of hypovitaminosis D in women during preg-nancy(8,10). Our study demonstrates a continuing high prevalence of vitamin D deficiency in lactating mothers, with 85% of the study mothers and 50% of control mothers having serum 25(OH)D levels <10ng/mL. Using similar cut offs for 25(OH)D, Dawodu, et al.(25) showed 61% of lactating mothers in Saudi Arabia to be vitamin D deficient. The strong correlation between maternal and infant serum 25(OH)D levels noted in the present study support the fact that infant serum 25 (OH)D levels are dependent not only on the maternal serum 25(OH)D levels at birth, but also on the breastmilk vitamin D content and on sunlight exposure of the mother and the child, which are probably very similar as they share a common environment(27-29). A positive relationship between maternal and fetal (cord blood) circulating 25(OH)D levels has been reported in recent studies(10,30,31). A similar relationship in vitamin D status has also been reported between mothers and infants suffering from rickets(2,32,33). The only study where no correlation between maternal-infant 25(OH)D levels was reported was from Turkey(15). We conclude that hypovitaminosis D in lactating mothers is strongly correlated with hypovitaminosis D in neonates and infants. Given this correlation, infants born to vitamin D deficient mothers are prone to develop hypocalcemic seizures. There is a need to assess the vitamin D status of all pregnant and lactating women and to consider routine vitamin D supplementation to breastfed infants, and pregnant and lactating women. Acknowledgments Kuntal Bhadra, Satvir Singh, Sushma Tripathy, Abhishek Kaushik and Madan Prasad for technical assistance. Contributors: RKM was involved with planning, and data analysis and will serve as guarantor; SA and AS were involved with planning and execution of the study; PM, BS And BS were involved with the execution of the study; MAG was involved with planning and data analysis; AS was involved with biochemical assays and NT was involved with planning, and data analysis. All authors have contributed to preparing the manuscript. Funding: Local grant (Project No INM 305) Institute of Nuclear Medicine and Allied Sciences, DRDO, Ministry of Defence, Government of India. Competing interests: None stated.
References 1. Chopra N. Study of etiology of seizures in young infants with specific reference to hypocalcemia. MD Thesis; University of Delhi: 2004. 2. Ahmed I, Atiq M, Iqbal J, Khurshid M, Whittaker P. Vitamin D deficiency rickets in breast-fed infants presenting with hypocalcaemic seizures. Acta Pediatr 1995; 84: 941-942. 3. Balasubramanian S, Shivbalan S, Kumar PS. Hypocalcemia due to vitamin D deficiency in exclusively breast fed infants. Indian Pediatr 2006; 43: 247-251. 4. Heckmatt JZ, Peacock M, Davies AEJ, McMurrray J, Isherwood DM. Plasma 25-hydroxyvitamin D in pregnant Asian women and their babies. Lancet 1979; 2: 546-548. 5. Cockburn F, Belton NR, Purvis RJ, Giles MM, Brown JK, Turner TL, et al. Maternal vitamin D intake and mineral metabolism in mothers and their new born infants. Br Med J 1980; 281: 11-14. 6. Edidin DV, Levitsky LL, Schey W, Dumbovic N, Campos A. Resurgence of nutritional rickets associated with breast feeding and special dietary practices. Pediatrics 1980; 65: 232-235. 7. Dawodu A, Agarwal M, Sankarankutty M, Hardy D. Kochiyil J, Badrinath P. Higher prevalence of vitamin D deficiency in mothers of rachitic than nonrachitic children. J Pediatr 2005; 147: 109-111. 8. Goswami R, Gupta N, Goswami D, Marwaha RK, Tandon N, Kochupillai N. Prevalence and significance of low 25-hydoxyvitamin D concentrations in healthy subjects in Delhi. Am J Clin Nutr 2000; 72: 472-475. 9. Marwaha RK, Tandon N, Reddy DR, Aggarwal R, Singh R, Sawhney RC, et al. Vitamin D and bone mineral density status of healthy school children in northern India. Am J Clin Nutr 2005; 82: 477-482. 10. Sachan A, Gupta R, Das V, Agarwal A, Awasthi PK, Bhatia V. High prevalence of vitamin D deficiency among pregnant women and their newborns in northern India. Am J Clin Nutr 2005; 81: 1060-1064. 11. ICMR. Recommended Dietary Intakes for Indians. Indian Council of Medical Research, New Delhi; 1990. 12. Balasubramanian K, Rajeswari J, Gulab, Govil YC, Agarwal AK, Kumar A, et al. Varying role of vitamin D deficiency in the etiology of rickets in young children vs adolescents in northern India. J Trop Pediatr 2003; 49: 201–206. 13. Lips P. Vitamin D deficiency and secondary hyperparathyroidism in the elderly: consequences for bone loss and fractures and therapeutic implications. Endocrinol Rev 2001; 22: 477-501. 14. Harinarayan CV, Ramalakshmi T, Prasad UV, Sudhakar D, Srinivasarao PV, Sarma KV, et al. High prevalence of low dietary calcium, high phytate consumption and vitamin D deficiency in healthy South Indians. Am J Clin Nutr 2007; 85: 1062-1067. 15. Hatun S, Ozkan B, Orbak Z, Doneray H, Cizmecioglu F, Toprak D, et al. Vitamin D deficiency in early infancy. J Nutr 2005; 135: 279-282. 16. Nesby-O’Dell S, Scanlon KS, Cogswell ME, Gillespie C, Hollis BW, Looker AC, et al. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: third National Health and Nutrition Examination survey: 1988-1994. Am J Clin Nutr 2002; 76: 187-192. 17. Rucker D, Allan JA, Fick GH, Hanley DA. Vitamin D insufficiency in a population of healthy western Canadians. CMAJ 2002; 166: 1517-1524. 18. Dawodu A. Absood G, Patel M, Agarwal M, Ezimokhai M, Abdulrazzaq Y, et al. Biosocial factors affecting vitamin D status of women of childbearing age in the United Arab Emirates. J Biosoc Sci 1998: 30: 431-437. 19. Gannage-Yared MH, Chemali R. Yaacoub N, Halaby G. Hypovitaminosis D in a sunny country: relation to lifestyle and bone markers. J Bone Miner Res 2000; 15: 1856-1862. 20. Ghannam NN, Hammami MM, Bakheet SM, Khan BA. Bone mineral density of the spine and femur in healthy Saudi females: relation to vitamin D status, pregnancy, and lactation. Calcif Tissue Int 1999; 65: 23-28. 21. Mishal AA. Effects of different dress styles of vitamin D levels in healthy young Jordanian women. Osteoporos Int 2001: 12: 931-935. 22. Hollis BW, Wagner CL. Assessment of dietary vitamin D requirements during pregnancy and lactation. Am J Clin Nutr 2004; 79: 717-726. 23. Van der Meer I, Karamali N, Boeke A, Lips P, Middelkoop BJ, Verhoeven I, et al. High prevalence of vitamin D deficiency in pregnant non-Western women in The Hague, Netherlands. Am J Clin Nutr 2006; 84; 350-353. 24. Grover SR, Morley R. Vitamin D deficiency in veiled or dark-skinned pregnant women. Med J Aust 2001: 175: 251-252. 25. Dawodu A, Agarwal M, Hossain M, Kochiyil J, Zayed R. Hypovitaminosis D and vitamin D deficiency in exclusively breast-feeding infants and their mothers in summer: a justification for vitamin D supplementation of breast-feeding infants. J Pediatr 2003: 142: 169-173. 26. Challa A, Ntourntoufi A, Cholevas V, Bitsori M, Galanakis E, Andronikou S. Breastfeeding and vitamin D status in Greece during the first 6 months of life. Eur J Pediatr 2003; 164: 724-729. 27. Rothberg DA, Pettifor JM, Cohen DF, Sonnendecker EWW, Ross FP. Maternal infant Vitamin D relationships during breast-feeding. J Pediatr 1982;101: 500-503. 28. Specker BL, Valanis B, Hetrzberg V, Edwards N, Tsang RC. Sunshine exposure and serum 25-hydroxyvitamin D concentrations in exclusively breast-fed infants. J Pediatr 1985; 107: 372-376. 29. Andiran N, Yordam N, Ozon A. Risk factors for vitamin D deficiency in breast-fed newborns and their mothers. Nutrition 2002; 18: 47-50. 30. Bassir M, Laborie S, Lapillonne A, Claris O, Chappuis MC, Salle BL. Vitamin D deficiency in Iranian mothers and their neonates: a pilot study. Acta Paediatr 2001; 90: 577-579. 31. Nicolaidou P, Hatzistamatiou Z, Papadopoulou A, Kaleyias J, Floropoulu E, Lagona E, et al. Low vitamin D status in mother-newborn pairs in Greece. Calcif Tissue Int 2006; 78: 337-342. 32. Daaboul J, Sanderson S, Kristensen K, Kitson H. Vitamin D deficiency in pregnant and breast-feeding women and their infants. J Perinatol 1997; 17: 10-14.
33. Nozza JM, Rodda CP. Vitamin D deficiency in mothers of infants with rickets. Med J Aust 2001; 175: 253-255. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||