Vitamin D: Bolus is Bogus (if more than 17 days between oral doses)
Vitamin D: Bolus is Bogus - a narrative review
JBMR Plus https://doi.org/10.1002/jbm4.10567
1Richard B. Mazess, 2,3Heike A. Bischoff-Ferrari, 4Bess Dawson-Hughes Affiliations:
1Dept. of Medical Physics, University of Wisconsin, Madison, Wisconsin USA
2Dept. of Aging Medicine and Aging Research, University of Zurich, Zurich, Switzerland
3University Clinic for Aging Medicine, City Hospital Zurich, Waid, Zurich, Switzerland
4Jean Mayer USDA Human Nutrition Research Center on Aging, Tufts University, Boston,
Massachusetts, USA
Abstract:
In this review we summarize the impact of bolus versus daily dosing of vitamin D on 25(OH)D and 1,25(OH)2D levels, as well as on key countervailing factors that block vitamin D functions at the cellular level. Further, we discuss the role of bolus versus daily dosing of vitamin D for several health outcomes, including respiratory infections and COVID-19, rickets, falls and fractures, any cancer, and cancer-related mortality. This discussion appears timely as bolus doses continue to be tested for various disease outcomes despite a growing amount of evidence suggesting lack of efficacy or even detrimental effects of bolus dosing of vitamin D for outcomes where daily dosing at modest levels was effective in the vitamin D deficient. As a result, these discordant results may bias health recommendations for vitamin D if the recommendations are based on meta-analyses combining both daily and bolus dosing trials.
Introduction:
Fifty years ago, the metabolism of vitamin D became elucidated in detail allowing clarification of its role in the skeletal system and stimulating examination of its extra-skeletal effects[1]. It was not until 30 years later that its role in systemic immunomodulation was elaborated and only 10 years ago did its role in cellular immune responses, autocrine and paracrine, unfold[2, 3], That intracellular process, mediated by both cholecalciferol, or D3, and the small free portion of calcifediol (25(OH)D) rather than circulating calcitriol (1,25(OH)2D), accounts for over 90% of the innate and adaptive immune response of vitamin D [4, 5], This tightly regulated relationship of zymatic transformation depends on CYP2R1 for initial hydroxylation and then on CYP27B1 for further hydroxylation to 1,25(OH)2D, versus enzymatic destruction of 1,25(OH)2D by CYP24A1 24-hydroxylation[3],
Over the past 20 years, there have been many clinical trials examining the supplementation of vitamin D in disease prevention. The low cost and safety of vitamin D often has led to its evaluation in the general public rather than in a cohort of deficient subjects with specific risk of a disease. As a consequence, much of the supplementation research is compromised, hence the value of correcting deficiencies remains uncertain based on the results of meta-analyses that include these trials. There are also other reasons why trials may not have shown clear effects of supplementation on disease outcomes [6, 7], as shown in Table 1.
In this review, we explore whether bolus dosing may be a design feature that leads to variable results of trials with several different outcomes. Our focus on bolus dosing arose because its lack of success was recently shown in several areas, including meta-analyses of acute respiratory infection trials[8, 9], in contrast to some studies in the same areas with positive results using daily or weekly dosing[8, 9]. It has long been known that high vitamin D dosing turns off the tightly regulated hormonal activation process of vitamin D, in both renal-skeletal and extra-renal (intracrine) metabolism, thereby inhibiting its modulatory function for weeks or longer. In fact, large bolus doses trigger countervailing factors, such as 24-hydroxylase (CYP24A1), that results in down-regulation of 1,25(OH)2D[3]. In contrast, moderate dosing at short intervals (daily or every- other day) is preferred because it does not trigger countervailing factors. Additionally, cholecalciferol itself, with a 20-hour half-life, is active intracellularly[3, 10]. Moreover, as suggested by Hollis and Wagner[11], most cells have 25-hydroxylase activity and therefore can utilize cholecalciferol directly whereas 99% of 25(OH)D is bound (or deposited in fat) and not available to cellular use. Further, cholecalciferol itself has been found to have greater cellular effects than either 25(OH)D or 1,25(OH)2D[12].
Effects of bolus versus daily dosing on countervailing factors
Several studies tested the effect of vitamin D dose on the resulting change in 25(OH)D concentrations[13]. The circulating 25(OH)D level has been chosen as the usual marker of vitamin D status because of its relatively long half-life of 20 days and bolus dosing with vitamin D dramatically increases circulating 25(OH)D levels. In fact, a meta-analysis of 30 studies using bolus dosing found that doses of >100,000 III increased 25(OH)D levels significantly, with levels peaking between 7 and 30 days [14], However, this is a limited short-term effect and the longer- term systemic effects on 1,25(OH)2D and the upregulation of countervailing factors, such as 24- hydroxylase (CYP24A1), that results in down-regulation of 1,25(OH)2D, were not studied. Also, the effects of bolus dosing on anti-microbial proteins (cathelicidin or LL-37), defensins, or regulatory Tcells were not examined. Notably, unlike daily doses of D3, large bolus doses have failed to increase anti-inflammatory cytokines nor decrease C-reactive protein response[14]. However, bolus doses of vitamin D almost double the risk of hypercalcemia[15].
Regarding the direct treatment with calcifediol, it has been shown to increase the blood level of 25(OH)D faster than cholecalciferol by a factor of 3.2[16]. This, however, may not indicate greater efficacy in 1,25(OH)2D production and immunomodulation. Notably, in a direct comparison of daily calcifediol dosing (5ug, 10ug, or 15 ug) versus 20ug of D3 daily over 24 weeks in 4 groups of women (n=59) over 65 years of age, 20ug of D3 produced a blood level of 25(OH)D that was 20% lower than that produced by 10ug of calcifediol (2.5X less potent)[17]. However, the 1,25(OH)2D produced with 20ug of D3 was equal to that of 5ug of calcifediol, and the production of the untervailing factor 24,25(OH)2D3 with D3 was only about half that of 5ug of calcifediol. The increases in 25(OH)D correlated highly (r=0.91) with those of 24,25(OH)2D3 but not with 1,25(OH)2D3. Another comparison of calcifediol and cholecalciferol, each at 20 ug/day, over 14 weeks in postmenopausal women (mean 61.5 years) showed that the doubled 25(OH)D response of calcifediol was not accompanied by a difference of 1,25(OH)2D, and 5 of 7 markers of innate immune function declined with both[18]. One comparative study over 7 months in older adults (>75years with 90% deficient) using equal doses (150 Dg/week showed calcifediol produced only a 20% higher serum level of 25(OH)D[19], Both were equal in doubling 1,25(OH)2D and reducing PTH and CRP. Thus, increases of 25(OH)D with bolus doses of vitamin D or with calcifediol can be deceptive as they do not increase 1,25(OH)2D as much as supposed.
Also, there is a concern that markedly elevated 25(OH)D levels disrupt the tightly regulated production of 1,25(OH)2D by stimulating fibroblast growth factor 23 (FGF23) which in turn suppresses 1 ?-hydroxylase (CYP2R1)[20]. Additionally, markedly elevated 25(OH)D levels activate 24-hydroxylase (CYP24A1) which converts 1,25(OH)2D into an inactive form (see Figure 1). These factors affect both renal and extra-renal (intracrine) metabolism. In contrast, a meta-analysis of 23 studies, of which 14 were in end-stage renal disease, concluded that FGF23 was not increased with daily vitamin D supplementation <2,000 IU, and if the 25(OH)D level was under <40 ng/ml[21]. A note of caution is appropriate when comparing FGF23 levels in trials that used different FgF23 assays, as some assays measure intact FGF23 and others measure a combination of the intact molecule and an inactive C-terminal fragment.
(Weekly Bolus)
The trigger of countervailing factors as a response to bolus dosing of D3 is apparent even at weekly intervals. Owens et al. tested 35,000 IU and 70,000 IU of D3 as a weekly bolus to athletes for l2 weeks and then followed them for an additional 6 weeks[22]. At week 12, the increase with the higher dose was only 10 to 15% higher than that with the lower dose but at week 12, the higher dose caused a 2.4X increase of 24,25(OFI)2D compared to a 0.4X increase at the lower dose[22]. Consequently, there was a similar (about 20-30%) increase of 1,25(OFI)2D with both doses, and serum 1,25(OFI)2D declined to baselines in both groups at week 18. Notably, the elevated 24,25(OFI)2D persisted for 6 weeks after dosing ended reflecting the longer-term induction of 24- hydroxylase blocking the formation of 1,25(OFI)2D [22], Fassio et al gave healthy adults under age 60 three different treatment regimens of vitamin D including daily and bolus dosing concepts (group A: 10,000 lU/day for eight weeks followed by 1,000 lU/day for four weeks; group B: 50,000 lU/week for 12 weeks, group C: 100,000 IU every other week for 12 weeks), amounting to the same cumulative dose of 600,000 IU D3, over 12 weeks[23]. They reported that daily dosing was superior in elevating 25(OFI)D with a 20% higher area under the curve. In another study, a single bolus of 150,000 IU compared to the same amount by daily dosing (5,000 IU) over 30 days, produced somewhat higher 25(OFI)D concentrations over the first 15 days in non-deficient women [24], however, the countervailing factor 24,25(OFI)2D3 increased about 50% with the bolus and 30% with daily dosing.
Effects of vitamin D dosing on acute respiratory infections and COVID-19 Mechanistically, vitamin D is relevant to healthy lungs because the alveolar epithelium has been found to convert it locally into active 1,25(OH)2D [10], This is further supported by the well- established effects of vitamin D on the innate and adaptive immune response [4, 25], including reduction of inflammation and the inhibition of cytokine excess by Tregs[26], Also, the cathelicidin LL-37 produced by vitamin D causes apoptosis of infected airway epithelium[27],
In a cohort of 9,548 patients followed over 15 years, mortality from respiratory disease was 2-fold higher in those with 25(OFI)D levels <20ng/ml and 3-fold higher with levels <12ng/ml [28], u?wever, prospective studies of supplementation have yielded conflicting results. The New Zealand ViDA study (n=5,111) that used a monthly bolus dose of 100,000 IU found no benefit for respiratory infections over 3 years even for those with baseline deficiency <20ng/ml [29], An expanded study (n=21,315) providing a monthly bolus of 60,000IU to adults aged 60-79 years over 5 years found no reduction of infections compared to placebo although the duration and severity of symptoms was decreased (34). A clarification of those findings was provided by Martineau et al [8] who evaluated individual participant data on about 11,000 participants from a previously reported meta-analysis of 25 vitamin D trials for acute respiratory infection. Bolus dosing was used in 10 of the trials (n= 5,595) while 15 trials (n=5,133) used daily or weekly doses. A 20% reduction of risk was shown for daily or weekly D3, but not in those receiving one or more bolus doses. That protective value of daily or weekly dosing was greatest (70%) in those with initial deficiency (25(OFI)D <10 ng/ml) versus those with insufficiency (25%). Heterogeneity among trials was due in large part to bolus dosing and the degree of initial deficiency. An expanded meta- analysis ? identified 46 RCTs including 75,541 participants aged 0-95 years but unfortunately the researchers did not obtain individual participant data that would have allowed accurate assessment of factors affecting responses. Vitamin D compared with placebo slightly (8%) reduced respiratory infections; bolus doses had no benefit while daily dosing reduced infections by 22%[9]. A similar discordance due to a preponderance of bolus dosing has been observed in meta-analyses of D3 in COPD patients [30].
(COVID)
The beginning of the COVID-19 pandemic in 2020 led to observations that the lungs are the primary infection site of COVID with secondary vascular manifestations[31]. Early observations of increased COVID exacerbation in adults with vitamin D deficiency, soon led to suggestions that supplementation with vitamin D needs investigation[32-34]. This was further supported by an association between 25(OH)D level and the degree of regional and total lung involvement observed in COVID patients [35, 36], as well as an association between the hyperinflammatory response or cytokine storm, characteristic in COVID-19 infection, produced by lung macrophages[25, 37],
As the pandemic continued several observational studies suggested that 25(OH)D insufficiency (<20 ng/ml) slightly increased risk of COVID-19 infection in adults[38-40]; one aberrant study using decade old 25(OH)D results and inappropriate statistical adjustments found no association[41]. However, the most recent meta-analysis of cohort studies on the topic showed an 80% increased risk of infection with vitamin D deficiency in 14 studies[42]. Other observational studies suggested that hospitalization rates are doubled, and critical care and mortality are tripled in adults with vitamin D deficiency [34, 43, 44], Further Akbar et al [51] reviewed 14 studies with almost one million adults in 2021 and concluded that 25(OH)D deficiency was associated with both severity of COVID and 3-fold higher odds of mortality. This association was confirmed by another recent review supporting a 2-fold increased risk of mortality and a 4-fold increased risk of critical care admission, although the authors pointed out the weak quality of observational studies [45],
RCTs of vitamin D treatment in COVID patients are ongoing; only four trials have been reported. A Brazilian trial that gave a single bolus of 200,000 III to 120 patients hospitalized for COVID-19 found it of no benefit on length of stay or mortality compared to 120 controls[46]. This is consistent with the results of the meta-analyses showing that bolus dosing did not reduce acute respiratory infections, as well as length of stay, severity and mortality in critically ill patients [8, 9], A different approach was taken in three Spanish trials. Entrenas Castillo et al treated 50 of 76 newly hospitalized COVID-19 patients with oral calcifediol (532 Dg) or no calcifediol upon admission, next to the same standard of care[47]. Patients in the calcifediol treatment group continued with oral calcifediol (266 Dg) on day 3 and 7, and then weekly until discharge or ICU admission. Only nns? treated patient required ICU admission (2%), while 13 of 26 (50%; p<0.001) patients on standard non-calcifediol treatment required ICU admission[47], A second trial tested the impact of 532 pg of calcifediol initially followed by 266 pg on days 3, 7, 15, and 30 in newly hospitalized COVID patients [48], Of the 447 patients treated with calcifediol at admission, 20 (4.5%) required ICU, while 82 (21%) of 391 non-treated COVID patients required ICU (p-value<0.0001). There was also a difference in mortality, 21 (4.7%) out of 447 patients treated with calcifediol compared to 62 patients (15.9%) out of 391 non-treated dying (p=0.0001). The ongoing nationwide US VIVID trial is a pragmatic, cluster randomized, double-blinded trial enrolling 1500 newly diagnosed individuals with COVID-19 infection and will test 3,200 IU vitamin D/day versus placebo[49].
Effects of bolus dosing on rickets (every 3 months does not work)
Treatment with daily low-dose vitamin D (400 IU), and/or calcium, has been the standard for prevention of rickets in nutritionally deprived children[1]. While several small, short-term studies have shown some success with bolus doses, both intramuscular and oral, for rickets the evidence for their efficacy in a recent Cochrane review was inconclusive[50]. A vitamin D supplementation study of 3,060 stunted children under age 1 year was done in Afghanistan. Half were on placebo and half received 100,000 IU of vitamin D every 3 months for 18 months[51]. There was no effect of the bolus vitamin D treatment on growth, and in a subgroup of 20% of children who received radiographic evaluation, rickets frequency was the same (5.4%) between bolus vitamin D and placebo groups. Clearly bolus dosing of vitamin D also contrasts sharply with the historical precedent of daily dosing benefits in the prevention and treatment of rickets [1]. This was an issue leading several experts to advocate medical use of daily dosing rather than boluses[52].
Effects of bolus dosing on falls and fractures (both daily and bolus have inconsistent results)
Observational studies show that vitamin D deficiency is common in the sarcopenia and muscular atrophy of the elderly as well as in osteoporosis and osteomalacia (1). The consequences of deficiency when combined result in the high rate of falls and fractures in the elderly. Despite this, supplementation trials with vitamin D, and its active analogs, have produced widely discordant findings. For example, a review of observational studies concluded that each 10ng/ml increase in 25(OH)D concentration was associated with a 20% reduction of femoral fracture but no benefit was shown in the accompanying review of RCTs[53], Some reasons for this anomaly are that the trials have employed a wide variation of subjects, not just frail elderly fallers with both low baseline 25(OH)D levels and low bone density. Researchers at times are confusing vitamin D supplementation for public health with trials to resolve a clinical issue. Trials also have shown wide variation of:
(a) daily supplementation doses,
(b) dose and interval of boluses,
(c) exclusion of even modest calcium co-administration,
(d) observation length, as well as
(e) disregard for levels of achieved 25(OH)D.
Moreover, the underlying mechanisms involved in falls (muscle strength, neuromuscular control, and balance) differ from those fundamental in fracture (bone density). There obviously will be subsets of a population in which each of these factors dominate, for example aging bone loss in males versus females, in addition to the usual factors affecting vitamin D deficiency (diet, obesity, aging, insolation, renal compromise). Cohesive analysis from multiple studies therefore requires pooling of individual participant data. Meta-analysis, with its usual weighting approach and forest plots, are deceptive, as shown by several recent meta-analyses that concluded there was no effect of vitamin D supplementation on fracture risk[54-56]. Some fundamental defects of such specious meta-analyses were outlined by Heaney a decade ago[57].
(Sarcopenia 3 month interval does not help)
Studies of sarcopenia, muscle strength and falls have demonstrated a complex response to supplementation. Trials in the elderly with deficiency have shown that low doses have little effect in muscle strength or lower extremity function but modest daily dosing (1,000 to 3,000 IU) increased strength[58, 59], Several meta-analyses of trials in older adults with deficiency indicate a reduction in falls with modest daily dosing of D3 supplementation, although conclusions have varied by dose and target population [60-68], Overall, low dose D3 (<700 lU/day) did not reduce fall risk significantly, a reduction was seen with daily D3 of 700 to 1,000 IU vitamin D, but large bolus dose (monthly 60,000 to 100,000 III of vitamin D or annual dosing of 300,000 III to 500,000 III) increased fall risk among frail older adults. For example, long-term care residents receiving 100,000 lU/month for one year doubled falls, but not fractures, even though 25(OH)D increased only from 25 to 32 ng/ml[69]. On the other hand, a lower bolus overload (150,000 III every 3 months ) produced a similar increase of 25(OH)D concentrations without affecting falls or muscular function[70]. Daily dosing at an equivalent level in replete older subjects does not increase falls. The VITAL study found that supplementation with 2,000 lU/day in 25,871 healthy and replete (mean 31 ng/ml) adults over age 50 resulted in a fall rate over 5 years that was identical to the fall rate of controls[71]. Similarly, the DO-HEALTH study showed that 2,000 IU/day over 3 years, producing a 25(OH)D increase from 24 to 38 ng/ml, did not affect leg function or non-vertebral fractures in mostly replete subjects[72].
(Falls and Fractures – monthly, 4 months, and annual do not help)
Falls and fractures are more common in older women than men, the latter being associated with the greater aging bone loss of females. This discordant pattern in falls complicates the evaluation of vitamin D on resultant fractures, but the adverse effect of bolus dosing on falls does seem to be reflected in fracture risk. For example, Sanders et al tested a D3 bolus of 500,000 IU annually compared with placebo for up to 5 years among 2,256 women aged 70 years and older who were at risk of fracture[73]. Their initial mean 25(OH)D level was 20 ng/ml[73][72]. and fracture and fall rates were, respectively, 26% and 15% higher in the treated group. A study using 300,000 IU intramuscular vitamin D2 (ergocalciferol) injection versus matching placebo every autumn over 3 years in 9,440 men and women aged over 75 years found that femoral fractures were 49% higher and wrist fractures were 22% higher in the bolus-treated compared to placebo-treated participants[74]. Another trial by Khaw of 5,108 largely replete older men and women (25(OH)D =24ng/ml) found that a starting D3 bolus of 200,000 IU followed by a monthly bolus doses of 100,000 IU did not prevent fractures or falls over 3 to 4 years[75]. However, a study in older UK subjects, where median 25(OH)D averaged about 16 ng/ml, found that a less aggressive D3 bolus of 100,000 IU given every 4 months for 5 years reduced fractures by 22%[76],
Some studies of daily dosing in relatively vitamin D replete groups of older adults have not demonstrated a major impact on non-vertebral fractures[71,72], However, daily dosing has been more effective in older adults who are deficient. Most meta-analyses have not utilized individual participant data that would allow control for age and sex, baseline and achieved levels of 25(OH)D, as well as daily versus bolus dosing that are critical to the issue[57]. Most recent meta-analyses simply rejected the influence of vitamin D on fracture risk, but two conceded that vitamin D plus calcium reduced the risk of femoral fractures by 15%[77] and up to 30%[78], Only one meta-analysis has used participant data (31,022 from 11 trials of which 90% were female) from double-blind RCTs only that are so essential to analyses[79]. It showed that with a median dose of 800 IU vitamin D per day, older adults at risk of vitamin D deficiency, had a 30% lower risk for hip fractures. The sensitivity analysis showed that the inclusion of studies with bolus dosing attenuated that benefit[79]. The detrimental effect of bolus dosing with regard to fracture risk is evident in the recent meta-analyses where trials using bolus dosing constituted two-thirds of the weighting[55, 56],
EFFects of bolus dosing on any cancer and cancer related mortality
An association between vitamin D deficiency and cancer has been observed for several decades and there are numerous mechanistic studies detailing how vitamin D and its analogs can influence cancer cells and reduce proliferation[80-90]. However, results from clinical trials testing supplemental vitamin D were mixed, with an overall suggestion that vitamin D may have no benefit on the prevention of any cancer [91-93], but may reduce the risk of advanced cancer and fatal cancer[91-93]. Consistently, a recent umbrella meta-analysis of clinical trials of vitamin D supplementation found no benefit on cancer incidence, but a reduction of total cancer mortality risk, with five out of six meta-analyses reporting a relative risk (RR) reduction of up to 16%[94], The large VITAL trial examined the effects of daily vitamin D supplements (2000 IU) on cancer and cardiovascular disease in 25,871 subjects over 5 years. While the study did not demonstrate an effect on the incidence of any cancer[95], a detailed secondary analysis showed that nonobese subjects had a 38% reduction of fatal and metastatic cancer[96]. Also, VITAL found a 20-25% reduction in cancer mortality if the first or also second year of latency were excluded[95]. Notably, in a cost-benefit analysis, Niedermeier et al calculated that a 13% lower cancer mortality through D3 supplementation of adults over age 50 years would prevent 30,000 deaths per year and result in net savings of $300 million in Germany alone[97].
(Cancer not helped by monthly dosing)
With regard to a comparison of daily versus bolus dosing, a 2019 meta-analysis identified 10 trials that tested vitamin D for cancer incidence and mortality, including the VITAL trial[98]. The meta-analysis had 6,537 cases, follow-ups were between 3 and 10 years, and achieved 25(OH)D levels were between 21 and 54 ng/ml in the intervention groups. While there was no benefit of D3 on cancer incidence, the meta-analysis documented a 13% reduction in cancer mortality with daily dosing of D3 but there was no benefit with bolus dosing [98]. A case example is the large ViDA study of 5,108 subjects that used a monthly bolus of 100,000 IU for up to 4 years and found no benefit for either cancer incidence or cancer mortality[99].
Summary
In this review we summarized and discussed growing evidence that large bolus dosing of vitamin D may have minimal benefit, or even be counterproductive, while small to moderate daily dosing in individuals at risk of deficiency is beneficial. This applied to outcomes of
rickets,
musculoskeletal health (falls and fractures) as well as
respiratory infections and
cancer mortality, and
possibly weekly dosing for calcifediol with regard to COVID-19.
However, as discussed above, the benefits of daily dosing have been absent in several studies among vitamin D replete adults and those not at risk for the outcomes of interest (i.e. falls and factures [71,721), and although this needs further study, we cannot exclude that also higher daily doses may trigger countervailing factors[241.
The increased use of bolus dosing in a growing number of trials may in part be motivated by convenience and purported benefits on adherence compared to daily dosing [100], In addition, the frequent use of bolus dosing for trials of vitamin D may be due to a misinterpretation of its short-term increase in 25(OH)D levels, while longer-term countervailing factors triggered by such doses that turn off its tightly regulated hormonal activation process have been overlooked[3]. The obvious countervailing increase of 24-hydroxylase leads to down-regulation of 1,25(OH)2D and inhibits immune-modulation for weeks or even months[3]. In other words, while a large bolus produces a quick increase in 25(OH)D levels, it does so at the cost of down-regulation of cellular activation and of factors of immunity [101], In contrast, a small to moderate dose of daily D3 has superior intracellular effects and needs frequent dosing due to its 20-hour half-life [3, 11, 24], If a rapid increase of 25(OH)D is needed, administration of an initial low calcifediol dose may be an alternative[47, 48], And once the 25(OH)D level is up, calcifediol would no longer be needed.
It has been difficult to isolate the influence of bolus dosing in meta-analyses because many others have merged these trials with those using daily dosing. Further, the low-cost and safety of D3 has led to studies in the broad public, not just in deficient persons or those at clinical risk, so the usual criteria of clinical trials may not be appropriate. Therefore, public health agencies are challenged to make a comprehensive risk-benefit assessment of the potential role of a daily small to moderate dose of vitamin D in the current COVID pandemic, particularly for those with known deficiency (older adults and darker-skinned ethnic groups)[8, 9] . Notably, even a decade ago it became apparent that bolus dosing was “too much of a good thing” [102], while correction of existing deficiencies with daily dosing of vitamin D is a low-cost and safe public health strategy in ameliorating a host of affected disorders.
Literature
Bouillon, R., et al., Skeletal and Extraskeletal Actions of Vitamin D: Current Evidence and Outstanding Questions. Endocr Rev, 2019. 40(4): p. 1109-1151.
Hewison, M., et al., Extra-renal 25-hydroxyvitamin D3-1alpha-hydroxylase in human health and disease. J Steroid Biochem Mol Biol, 2007. 103(3-5): p. 316-21.
Hewison, M., Vitamin D and the immune system: new perspectives on an old theme. Endocrinol Metab Clin North Am, 2010. 39(2): p. 365-79, table of contents.
Xu, Y., et al., The importance of vitamin d metabolism as a potential prophylactic, immunoregulatory and neuroprotective treatment for COVID-19. J Transl Med, 2020. 18(1): p. 322.
Wimalawansa, S.J., Associations of vitamin D with insulin resistance, obesity, type 2 diabetes, and metabolic syndrome. J Steroid Biochem Mol Biol, 2018.175: p. 177-189.
Grant, W.B. and B.J. Boucher, Why Secondary Analyses in Vitamin D Clinical Trials Are Important and How to Improve Vitamin D Clinical Trial Outcome Analyses-A Comment on "Extra-Skeletal Effects of Vitamin D, Nutrients 2019,11,1460". Nutrients, 2019.11(9).
Zgaga, L., Heterogeneity of the Effect of Vitamin D Supplementation in Randomized Controlled Trials on Cancer Prevention. JAMA Netw Open, 2020. 3(11): p. e2027176.
Martineau, A.R., et al., Vitamin D supplementation to prevent acute respiratory infections: individual participant data meta-analysis. Health Technol Assess, 2019. 23(2): p. 1-44.
Jolliffe, D.A., et al., Vitamin D supplementation to prevent acute respiratory infections: a systematic review and meta-analysis of aggregate data from randomised controlled trials. Lancet Diabetes Endocrinol, 2021. 9(5): p. 276-292.
Hansdottir, S., et al., Respiratory epithelial cells convert inactive vitamin D to its active form: potential effects on host defense. J Immunol, 2008.181(10): p. 7090-9.
Hollis, B.W. and C.L. Wagner, Clinical review: The role of the parent compound vitamin D with respect to metabolism and function: Why clinical dose intervals can affect clinical outcomes. J Clin Endocrinol Metab, 2013. 98(12): p. 4619-28.
Gibson, C.C., et al., Dietary Vitamin D and Its Metabolites Non-Genomically Stabilize the Endothelium. PLoS One, 2015.10(10): p. e0140370.
Bischoff-Ferrari, H.A., et al., Benefit-risk assessment of vitamin D supplementation. Osteoporos Int, 2010. 21(7): p. 1121-32.
Kearns, M.D., J.A. Alvarez, and V. Tangpricha, Large, single-dose, oral vitamin D supplementation in adult populations: a systematic review. Endocr Pract, 2014. 20(4): p. 341-51.
Malihi, Z., et al., Adverse events from large dose vitamin D supplementation taken for one year or longer. J Steroid Biochem Mol Biol, 2019.188: p. 29-37.
Quesada-Gomez, J.M. and R. Bouillon, Is calcifediol better than cholecalciferol for vitamin D supplementation? Osteoporos Int, 2018. 29(8): p. 1697-1711.
Vaes, A.M.M., et al., Dose-response effects of supplementation with calcifediol on serum 25- hydroxyvitamin D status and its metabolites: A randomized controlled trial in older adults. Clin Nutr, 2018. 37(3): p. 808-814.
Bischoff-Ferrari, H.A., et al., Oral supplementation with 25(OH)D3 versus vitamin D3: effects on 25(OH)D levels, lower extremity function, blood pressure, and markers of innate immunity. J Bone
Miner Res, 2012. 27(1): p. 160-9.
Ruggiero, C., et al., Effects of Weekly Supplementation of Cholecalciferol and Calcifediol Among the Oldest-Old People: Findings From a Randomized Pragmatic Clinical Trial. Nutrients, 2019. 11(11).
Bacchetta, J., et al., Fibroblast growth factor 23 inhibits extrarenal synthesis of 1,25- dihydroxyvitamin D in human monocytes. J Bone Miner Res, 2013. 28(1): p. 46-55.
Zittermann, A., H.K. Berthold, and S. Pilz, The effect of vitamin D on fibroblast growth factor 23: a systematic review and meta-analysis of randomized controlled trials. Eur J Clin Nutr, 2021. 75(6): p. 980-987.
Owens, D.J., et al., Efficacy of High-Dose Vitamin D Supplements for Elite Athletes. Med Sci Sports Exerc, 2017. 49(2): p. 349-356.
Fassio, A., et al., Pharmacokinetics of Oral Cholecalciferol in Healthy Subjects with Vitamin D Deficiency: A Randomized Open-Label Study. Nutrients, 2020.12(6).
Ketha, H., et al., Comparison of the effect of daily versus bolus dose maternal vitamin D3 supplementation on the 24,25-dihydroxyvitamin D3 to 25-hydroxyvitamin D3 ratio. Bone, 2018. 110: p. 321-325.
Kumar, R., et al., Putative roles of vitamin D in modulating immune response and immunopathology associated with COVID-19. Virus Res, 2021. 292: p. 198235.
Chung, C., et al., Vitamin D-Cathelicidin Axis: at the Crossroads between Protective Immunity and Pathological Inflammation during Infection. Immune Netw, 2020. 20(2): p. el2.
Barlow, P.G., et al., The human cathelicidin LL-37 preferentially promotes apoptosis of infected airway epithelium. Am J Respir Cell Mol Biol, 2010.43(6): p. 692-702.
Brenner, H., B. Holleczek, and B. Schottker, Vitamin D Insufficiency and Deficiency and Mortality from Respiratory Diseases in a Cohort of Older Adults: Potential for Limiting the Death Toll during and beyond the COVID-19 Pandemic? Nutrients, 2020.12(8).
Camargo, C.A., et al., Effect of Monthly High-Dose Vitamin D Supplementation on Acute Respiratory Infections in Older Adults: A Randomized Controlled Trial. Clin Infect Dis, 2020. 71(2): p.311-317.
Chen, F.Y., et al., Vitamin D does not improve lung function decline in COPD: a meta-analysis. Eur Rev Med Pharmacol Sci, 2019. 23(19): p. 8637-8644.
Azer, S.A., COVID-19: pathophysiology, diagnosis, complications and investigational therapeutics. New Microbes New Infect, 2020. 37: p. 100738.
Grant, W.B., et al., Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients, 2020.12(4).
Benskin, L.L., A Basic Review of the Preliminary Evidence That COVID-19 Risk and Severity Is Increased in Vitamin D Deficiency. Front Public Health, 2020. 8: p. 513.
Charoenngam, N., et al., Association of Vitamin D Status With Hospital Morbidity and Mortality in Adult Hospitalized Patients With COVID-19. Endocr Pract, 2021. 27(4): p. 271-278.
Abrishami, A., et al., Possible association of vitamin D status with lung involvement and outcome in patients with COVID-19: a retrospective study. Eur J Nutr, 2021. 60(4): p. 2249-2257.
Ricci, A., et al., Circulating Vitamin D levels status and clinical prognostic indices in COVID-19 patients. Respir Res, 2021. 22(1): p. 76.
Kloc, M., et al., Effects of vitamin D on macrophages and myeloid-derived suppressor cells (MDSCs) hyperinflammatory response in the lungs of COVID-19 patients. Cell Immunol, 2021. 360: p. 104259.
Kaufman, H.W., et al., SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels. PLoS One, 2020.15(9): p. e0239252.
Merzon, E., et al., Low plasma 25(OH) vitamin D level is associated with increased risk of COVID- 19 infection: an Israeli population-based study. FEBS J, 2020. 287(17): p. 3693-3702.
Brenner, H., Vitamin D Supplementation to Prevent COVID-19 Infections and Deaths- Accumulating Evidence from Epidemiological and Intervention Studies Calls for Immediate Action. Nutrients, 2021. 13(2).
Hastie, C.E., et al., Vitamin D concentrations and COVID-19 infection in UK Biobank. Diabetes Metab Syndr, 2020. 14(4): p. 561-565.
Teshome, A., et al., The Impact of Vitamin D Level on COVID-19 Infection: Systematic Review and Meta-Analysis. Front Public Health, 2021. 9: p. 624559.
Angelidi, A.M., et al., Vitamin D Status Is Associated With In-Hospital Mortality and Mechanical Ventilation: A Cohort of COVID-19 Hospitalized Patients. Mayo Clin Proc, 2021. 96(4): p. 875886.
Petrelli, F., et al., Therapeutic and prognostic role of vitamin D for COVID-19 infection: A systematic review and meta-analysis of 43 observational studies. J Steroid Biochem Mol Biol, 2021. 211: p. 105883.
Bassatne, A., et al., The link between COVID-19 and Vitamin D (VIVID): A systematic review and meta-analysis. Metabolism, 2021.119: p. 154753.
Murai, I.H., et al., Effect of a Single High Dose of Vitamin D3 on Hospital Length of Stay in Patients With Moderate to Severe COVID-19: A Randomized Clinical Trial. JAMA, 2021. 325(11): p. 10531060.
Entrenas Castillo, M., etal., "Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study". J Steroid Biochem Mol Biol, 2020. 203: p. 105751.
Nogues, X., et al., Calcifediol treatment and COVID-19-related outcomes. J Clin Endocrinol Metab, 2021.
Wang, R., et al., The vitamin D for COVID-19 (VIVID) trial: A pragmatic cluster-randomized design. Contemp Clin Trials, 2021.100: p. 106176.
Chibuzor, M.T., et al., Vitamin D, calcium or a combination of vitamin D and calcium for the treatment of nutritional rickets in children. Cochrane Database Syst Rev, 2020.4: p. CD012581.
Crowe, F.L., et al., Vitamin D for Growth and Rickets in Stunted Children: A Randomized Trial. Pediatrics, 2021.147(1).
Griffin, G., et al., Perspective: Vitamin D supplementation prevents rickets and acute respiratory infections when given as daily maintenance but not as intermittent bolus: implications for COVID- 19. Clin Med (Lond), 2021. 21(2): p. el44-el49.
Yao, P., et al., Vitamin D and Calcium for the Prevention of Fracture: A Systematic Review and Meta-analysis. JAMA Netw Open, 2019. 2(12): p. el917789.
Kahwati, L.C., et al., Vitamin D, Calcium, or Combined Supplementation for the Primary Prevention of Fractures in Community-Dwelling Adults: Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA, 2018. 319(15): p. 1600-1612.
Zhao, J.G., et al., Association Between Calcium or Vitamin D Supplementation and Fracture Incidence in Community-Dwelling Older Adults: A Systematic Review and Meta-analysis. JAMA, 2017. 318(24): p. 2466-2482.
Bolland, M.J., A. Grey, and A. Avenell, Effects of vitamin D supplementation on musculoskeletal health: a systematic review, meta-analysis, and trial sequential analysis. Lancet Diabetes Endocrinol, 2018. 6(11): p. 847-858.
Heaney, R.P., Vitamin D-baseline status and effective dose. N Engl J Med, 2012. 367(1): p. 77-8.
Halfon, M., O. Phan, and D. Teta, Vitamin D: a review on its effects on muscle strength, the risk of fall, and frailty. Biomed Res Int, 2015. 2015: p. 953241.
Kupisz-Urbanska, M., P. Pludowski, and E. Marcinowska-Suchowierska, Vitamin D Deficiency in Older Patients-Problems of Sarcopenia, Drug Interactions, Management in Deficiency. Nutrients, 2021. 13(4).
Bischoff-Ferrari, H.A., et al., Effect of Vitamin D on falls: a meta-analysis. Jama, 2004. 291(16): p. 1999-2006.
Bischoff-Ferrari, H.A., et al., Fall prevention with supplemental and active forms of vitamin D: a meta-analysis of randomised controlled trials. BMJ, 2009. 339(1): p. 339:b3692.
Jackson, C., et al., The effect of cholecalciferol (vitamin D3) on the risk of fall and fracture: a metaanalysis. Qjm, 2007. 100(4): p. 185-92.
Kalyani, R.R., et al., Vitamin D treatment for the prevention of falls in older adults: systematic review and meta-analysis. J Am Geriatr Soc, 2010. 58(7): p. 1299-310.
O'Donnell, S., et al., Systematic review of the benefits and harms of calcitriol and alfacalcidol for fractures and falls. J Bone Miner Metab, 2008. 26(6): p. 531-42.
Richy, F., L. Dukas, and E. Schacht, Differential effects ofD-hormone analogs and native vitamin D on the risk of falls: a comparative meta-analysis. Calcif Tissue Int, 2008. 82(2): p. 102-7.
Michael, Y.L., etal., Interventions to Prevent Falls in Older Adults: An Updated Systematic Review. U.S. Preventive Services Task Force Evidence Syntheses, formerly Systematic Evidence Reviews., 2011.
Michael, Y.L., et al., Primary care-relevant interventions to prevent falling in older adults: a systematic evidence review for the u.s. Preventive services task force. Ann Intern Med, 2011. 153(12): p.815-25.
Cameron, I.D., et al., Interventions for preventing falls in older people in nursing care facilities and hospitals. Cochrane Database Syst Rev, 2010(1): p. CD005465.
Ginde, A.A., et al., High-Dose Monthly Vitamin D for Prevention of Acute Respiratory Infection in Older Long-Term Care Residents: A Randomized Clinical Trial. J Am Geriatr Soc, 2017. 65(3): p. 496-503.
Glendenning, P., et al., Effects of three-monthly oral 150,000 IU cholecalciferol supplementation onfalls, mobility, and muscle strength in older postmenopausal women: a randomized controlled trial. J Bone Miner Res, 2012. 27(1): p. 170-6.
LeBoff, M.S., et al., VITamin D and OmegA-3 TriaL (VITAL): Effects of Vitamin D Supplements on Risk of Falls in the US Population. J Clin Endocrinol Metab, 2020.105(9).
Bischoff-Ferrari, H.A., et al., Effect of Vitamin D Supplementation, Omega-3 Fatty Acid Supplementation, or a Strength-Training Exercise Program on Clinical Outcomes in Older Adults: The DO-HEALTH Randomized Clinical Trial. JAMA, 2020. 324(18): p. 1855-1868.
Sanders, K.M., et al., Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. JAMA, 2010. 303(18): p. 1815-22.
Smith, H., et al., Effect of annual intramuscular vitamin D on fracture risk in elderly men and women-a population-based, randomized, double-blind, placebo-controlled trial. Rheumatology (Oxford), 2007. 46(12): p. 1852-7.
Khaw, K.T., et al., Effect of monthly high-dose vitamin D supplementation on falls and nonvertebral fractures: secondary and post-hoc outcomes from the randomised, double-blind, placebo-controlled ViDA trial. Lancet Diabetes Endocrinol, 2017. 5(6): p. 438-447.
Trivedi, D.P., R. Doll, and K.T. Khaw, Effect of four monthly oral vitamin D3 (cholecalciferol) supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. BMJ, 2003. 326(7387): p. 469.
Avenell, A., J.C. Mak, and D. O'Connell, Vitamin D and vitamin D analogues for preventing fractures in post-menopausal women and older men. Cochrane Database Syst Rev, 2014(4): p. CD000227.
Weaver, C.M., et al., Calcium plus vitamin D supplementation and risk of fractures: an updated meta-analysis from the National Osteoporosis Foundation. Osteoporos Int, 2016. 27(1): p. 36776.
Bischoff-Ferrari, H.A., et al., A pooled analysis of vitamin D dose requirements for fracture prevention. N Engl J Med, 2012. 367(1): p. 40-9.
Gaschott, T. and J. Stein, Short-chain fatty acids and colon cancer cells: the vitamin D receptor- butyrate connection. Recent Results Cancer Res, 2003. 164: p. 247-57.
Sundaram, S., et al., The vitamin D3 analog EB1089 enhances the antiproliferative and apoptotic effects of adriamycin in MCF-7 breast tumor cells. Breast Cancer Res Treat, 2000. 63(1): p. 110.
Campbell, M.J., et al., Inhibition of proliferation of prostate cancer cells by a 19-nor-hexafluoride vitamin D3 analogue involves the induction of p21waf1, p27kip1 and E-cadherin. J Mol Endocrinol, 1997.19(1): p. 15-27.
Li, P., et al., p27(Kipl) stabilization and G(l) arrest by 1,25-dihydroxyvitamin D(3) in ovarian cancer cells mediated through down-regulation ofcyclin E/cyclin-dependent kinase 2 and Skpl- CuIIin-F-boxprotein/Skp2 ubiquitin ligase. J Biol Chem, 2004. 279(24): p. 25260-7.
Kawa, S., et al., Inhibitory effect of 220-oxa-l,25-dihydroxyvitamin D3 on the proliferation of pancreatic cancer cell lines. Gastroenterology, 1996.110(5): p. 1605-13.
Yanagisawa, J., et al., Convergence of transforming growth factor-beta and vitamin D signaling pathways on SMAD transcriptional coactivators. Science, 1999. 283(5406): p. 1317-21.
Moreno, J., et al., Regulation of prostaglandin metabolism by calcitriol attenuates growth stimulation in prostate cancer cells. Cancer Res, 2005. 65(17): p. 7917-25.
James, S.Y., A.G. Mackay, and K.W. Colston, Effects of 1,25 dihydroxyvitamin D3 and its analogues on induction of apoptosis in breast cancer cells. J Steroid Biochem Mol Biol, 1996. 58(4): p. 395401.
Diaz, G.D., et al., Apoptosis is induced by the active metabolite of vitamin D3 and its analogue EB1089 in colorectal adenoma and carcinoma cells: possible implications for prevention and therapy. Cancer Res, 2000. 60(8): p. 2304-12.
Kumagai, T., et al., 19-Nor-l,25(OH)2D2 (a novel, noncalcemic vitamin D analogue), combined with arsenic trioxide, has potent antitumor activity against myeloid leukemia. Cancer Res, 2005. 65(6): p.2488-97.
Jiang, F., et al., Induction of ovarian cancer cell apoptosis by 1,25-dihydroxyvitamin D3 through the down-regulation of telomerase. J Biol Chem, 2004. 279(51): p. 53213-21.
Chandler, P.D., et al., Effect of Vitamin D3 Supplements on Development of Advanced Cancer: A Secondary Analysis of the VITAL Randomized Clinical Trial. JAMA network open, 2020. 3(11): p. e2025850-e2025850.
Haykal, T., et al., The role of vitamin D supplementation for primary prevention of cancer: metaanalysis of randomized controlled trials. Journal of community hospital internal medicine perspectives, 2019. 9(6): p. 480-488.
Keum, N., et al., Vitamin D supplementation and total cancer incidence and mortality: a metaanalysis of randomized controlled trials. Annals of Oncology, 2019. 30(5): p. 733-743.
Sluyter, J.D., J.E. Manson, and R. Scragg, Vitamin D and Clinical Cancer Outcomes: A Review of Meta-Analyses. JBMR Plus, 2021. 5(1): p. el0420.
Manson, J.E., et al., Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N Engl J Med, 2019. 380(1): p. 33-44.
Chandler, P.D., et al., Effect of Vitamin D3 Supplements on Development of Advanced Cancer: A Secondary Analysis of the VITAL Randomized Clinical Trial. JAMA Netw Open, 2020. 3(11): p. e2025850.
Niedermaier, T., et al., Vitamin D supplementation to the older adult population in Germany has the cost-saving potential of preventing almost 30 000 cancer deaths per year. Mol Oncol, 2021.
Keum, N., et al., Vitamin D supplementation and total cancer incidence and mortality: a metaanalysis of randomized controlled trials. Ann Oncol, 2019. 30(5): p. 733-743.
Scragg, R., et al., Monthly High-Dose Vitamin D Supplementation and Cancer Risk: A Post Hoc Analysis of the Vitamin D Assessment Randomized Clinical Trial. JAMA Oncol, 2018. 4(11): p. e182178.
Kearns, M.D., et al., The effect of a single, large bolus of vitamin D in healthy adults over the winter and following year: a randomized, double-blind, placebo-controlled trial. Eur J Clin Nutr, 2015. 69(2): p. 193-7.
Arabi, S.M., et al., Efficacy of high-dose versus low-dose vitamin D supplementation on serum levels of inflammatory factors and mortality rate in severe traumatic brain injury patients: study protocol for a randomized placebo-controlled trial. Trials, 2020. 21(1): p. 685.
Dawson-Hughes, B. and S.S. Harris, High-dose vitamin D supplementation: too much of a good thing? JAMA, 2010. 303(18): p. 1861-2.
Table 1. Factors potentially affecting vitamin D supplement trial outcomes.
subjects with mixed or replete baseline 25(OH)D levels rather than deficiency or insufficiency
results affected by obesity and aging
discordant increases of blood levels
short-term focus when longer-term intervention is needed
failure to consider baseline level and achieved level of 25(OH)D
use of large bolus doses rather than daily or weekly dosing at modest levels
Table 2 - RCT Data regarding acute respiratory infections and COVID-19
VitaminDWiki
Non-daily (Bolus) is virtually always better:
Better than Daily has the following
{include}
Examples of non-daily vitamin D
44 percent of successful RCTs in VitaminDWiki used non-daily dosing - Nov 2020 217
Vitamin D restoration then monthly was the most popular dosing by trials – Nov 2018
Bi-weekly would be better, but people can easily rememberi the first day of the month
50,000 IU of vitamin D every two weeks – Jordan conclusion - RCT July 2017
22 diseases proven to be prevented/treated by biweekly Vitamin D
10 additional diseases with weekly Vitamin D
Vitamin D for infants – 100,000 IU monthly is better than 400 IU daily – RCT Oct 2020
Optimal vitamin D supplementation strategies (D3 - weekly or monthly) – Feb 2017
Editorial by Bouillon with comments by VitaminDWiki
Study on this page ignores the fact that bolus dosing gets past VDR barriers
Infrequent dosing provides higher Vitamin D gradient
Approximately 30% of people have VDR barriers, some of which were caused by the health problem
Study on this page ignores that fact that all animals store Vitamin D to survive during winter, rainy times
Pregnancies and breast feeding do not stop during the winter or rainy seasons
injections release Vitamin D so slowly that they can be done less often (every 2 months)
Injection contains the following
{include}
Intervention - non daily AND Diabetes
{category}
Intervention - non daily AND Pain
{category}
Intervention - non daily AND Pregnancy
{category}
Intervention - non daily AND Senors
{category}
Intervention - non daily AND Infant-Child
{category}
Intervention - non daily ALL
Charts of dosing interval: Vitamin D <30 ng and > 50 ng
Take vitamin D3 daily, weekly, or bi-weekly
Notional chart for Vitamin D supplementation for levels <30 nanograms
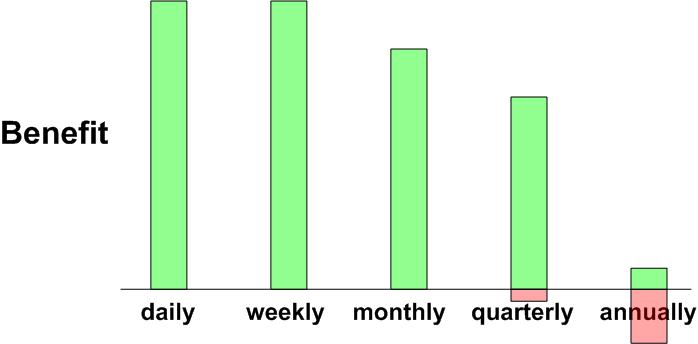
Notional chart for Vitamin D supplementation for levels >50 nanograms

