Rheumatoid Arthritis synovial fluid has lower levels Vitamin D binding protein, 3-epi
Serum and synovial fluid vitamin D metabolites and rheumatoid arthritis
Journal of Steroid Biochemistry and Molecular Biology https://doi.org/10.1016/j.jsbmb.2018.10.008
 Meta-analysis of Rheumatoid Arthritis and Vitamin D in VitaminDWiki
{category}
Meta-analysis of Rheumatoid Arthritis and Vitamin D in VitaminDWiki
{category}
📄 Download the PDF from VitaminDWiki
Synovial Fluid (SF) has lower levels of 3-epi metabolite
In those with Rheumatoid Arthritis (not necessarily the joint which was measured)
Synovial Fluid (SF) has lower levels of 3-epi metabolite

Synovial Fluid (SF) has lower levels of Vitamin D Binding Protein
Danyang Lia, Louisa E. Jeffery*5, Carl Jenkinsorf, Stephanie R. Harrison3/ Rene F. Chund, John S. Adamsd, Karim Razac,e, Martin Hewisona,f’m. hewison@bham.ac.uk
a Institute of Metabolism and Systems Research, The University of Birmingham, Birmingham, B15 2TT, UK
b Institute of Translational Medicine, The University of Birmingham, Birmingham, B15 2TT, UK
c Department of Rheumatology, Sandwell and West Birmingham Hospitals NHS Trust, Birmingham, B18 7QH, UK
d Department of Orthopaedic Surgery, University of California, Los Angeles, Los Angeles, CA 90095, USA
e Institute of Inflammation and Ageing, Arthritis Research UK Rheumatoid Arthritis Pathogenesis Centre of Excellence and MRC Arthritis Research UK
Vitamin D-deficiency has been linked to inflammatory diseases including rheumatoid arthritis (RA). Studies to date have focused on the impact of serum 25-hydroxyvitamin D3 (25(OH)D3), an inactive form of vitamin D, on RA disease activity and progression. However, anti-inflammatory actions of vitamin D are likely to be mediated at sites of RA disease, namely the inflamedjoint, and may involve other vitamin D metabolites notably the active form of vitamin D, 1,25-dihydroxyvitamin D3 (1,25(OH)2D3). In the current study serum and synovial fluid samples from n = 20 patients with persistent RA and n = 7 patients with reactive arthritis (ReA) were analysed for multiple vitamin D metabolites. Serum data for RA and ReA patients were compared to healthy controls (HC). There was no significant difference between RA or ReA patients relative to HC for 25(OH)D3, 24,25(OH)2D3, 1,25(OH)2D3 or 25(OH)D2. However, 3-epi-25(OH)D3 was significantly lower in RA and ReA patients compared to HC (p < 0.05). All vitamin D metabolites, apart from 25(OH)D2, were lower in SF compared to serum, and SF 1,25(OH)2D3 was unquantifiable in 13/20 RA and 4/7 ReA samples. SF 25(OH)D3, 3-epi-25(OH)D3 and DBP correlated inversely with swollen joint score, and serum 25(OH)D2 and SF DBP correlated directly with C- reactive protein levels. These data indicate that serum 25(OH)D3 provides only limited insight into the role of vitamin D in RA. Alternative serum metabolites such as 3-epi-25(OH)2D3, and SF metabolites, notably lack of SF 1,25(OH)2D3, may be more closely linked to RA disease severity and progress.
Introduction
In addition to its established actions as a regulator of mineral homeostasis and bone metabolism, vitamin D has been reported to promote potent immunomodulatory responses including the induction of innate antibacterial [1], and adaptive anti-inflammatory, tolerogenic adaptive [2] immunity. Studies ex vivo suggest that the proposed immunomodulatory actions of vitamin D are mediated via metabolism of precursor 25-hydroxyvitamin D3 (25(OH)D3) to active 1,25-dihydroxyvitamin D3 (1,25(OH)2D3) by antigen-presenting cells (APC) such as macrophages [3] and dendritic cells [4]. The resulting localized synthesis of 1,25(OH)2D3 may then fulfill endogenous, intracrine, functions such as enhanced antibacterial responses [5,6] or suppression of antigen-presentation [4,7,8]. However, APC-derived 1,25(OH)2D3 may also function in a paracrine fashion by acting on activated T lymphocytes (T cells) that express the nuclear vitamin receptor (VDR), and which show anti-inflammatory, tolerogenic responses to 1,25(OH)2D3 [9-12]. The over-arching role of vitamin D within the immune system therefore appears to be to combat infection, whilst suppressing potentially detrimental inflammation that may be associated with infection or inappropriate activation by self-antigens.
The role of vitamin D as a regulator of immune function has also been studied in vivo. Mice with knockout of the gene for VDR (Vdr KO) or the vitamin D-activating enzyme 1a-hydroxylase (Cyp27b1) show increased severity of colitis in mouse models of inflammatory bowel disease (IBD) colitis. Vdr KOs also shows aggravated inflammatory disease in murine models of rheumatoid arthritis (RA).
Likewise, mice that are vitamin D-deficient (low serum 25(OH) D3) show increased murine colitis [16] and experimental autoimmune encephalomyelitis [17]. In humans, vitamin D-deficiency has been linked to increased risk and disease severity for autoimmune diseases such as multiple sclerosis [18], type 1 diabetes [19], IBD [20], and RA [21-23].
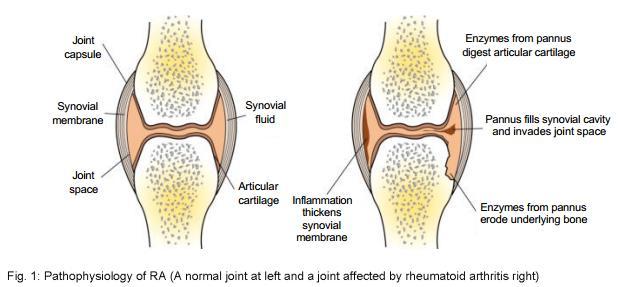
RA is a systemic inflammatory disease characterised by the symmetrical onset of multiple painful swollen joints, eventually leading to permanent joint destruction, loss of mobility and increased risk of mortality [24]. Clinically the diagnosis of RA is defined by the points- based EULAR Classification Criteria; these include measurements of c- reactive protein (CRP), erythrocyte sedimentation rate (ESR) and the tender/swollen joint count (TJC/SJC). This cumulative scoring system defines a score of > 6 as a definitive diagnosis for RA. In contrast to RA, Reactive Arthritis (ReA) is an acute form of inflammatory joint arthritis often triggered as a secondary response to gram-negative bacterial infection [25,26]. Unlike RA the diagnosis of ReA is based on clinical features such as swollen joints, as opposed to measurements of inflammatory markers in blood. Elimination of bacterial infection requires the activation of T-helper (Th) 1 cells and the release of associated pro-inflammatory cytokines IFN-y and TNF-a, triggering secondary inflammation as seen in ReA [27].
Despite the observed links between vitamin D-deficiency and autoimmune disease, a causative role for vitamin D-deficiency in the onset and severity of inflammatory disease is still not clear. In RA, some studies have demonstrated a link between low vitamin D in early stage RA and subsequent disease progression [23], whilst others have not [28]. In common with other inflammatory diseases, the impact of vitamin D on RA has been assessed primarily via the measurement of serum levels of 25(OH)D3, although this is not an active metabolite. The aim of the current study was therefore to firstly determine whether other vitamin D metabolites, including 1,25(OH)2D3, may be more informative in defining RA disease. The study also involved analysis of paired synovial fluid samples to assess the relationship between tissue- specific concentrations of vitamin D metabolites and associated serum levels. The third aim of the study was to assess the impact of the serum carrier protein, vitamin D binding protein (DBP), on serum and SF vitamin D function with respect to RA disease activity.
Materials and methods
Patients
Matched serum and synovial fluid samples from n = 20 patients with active rheumatoid arthritis (RA) and n = 7 patients with resolving reactive arthritis (ReA) were obtained with patient consent according to existing ethical agreements. Both RA and ReA patients had arthritis and active inflammation at the time of presentation and sample collection in clinic. RA patients continued to have ongoing inflammatory disease whereas ReA patients had a form of reactive arthritis that spontaneously resolved. Patients were assessed for various disease activity markers including erythrocyte sedimentation rate (ESR), c-reactive protein (CRP), and the 28 joint tender joint count (TJC) and swollen joint count (SJC) [29]. Disease-free age-matched subjects were used as healthy control (HC) group for serum analyses. RA and ReA patient demographics are shown in full in Table 1, and Supplemental Table 1 for healthy controls.
Quantification of serum and SF vitamin D metabolites
Liquid-chromatography tandem mass-spectrometry (LC-MS/MS) was used to quantify the following vitamin D metabolites in serum and SF; 25(OH)D3, 3-Epi-25(OH)D3, 25(OH)D2, 24,25-dihydroxyvitamin D3 (24,25(OH)2D3) and 1,25(OH)2D3 as described previously [30-32] with slight modifications. In brief, samples were prepared for analysis by protein precipitation and supported liquid-liquid extraction, followed by derivatization with 4-(2-(6,7-dimethoxy-4-methyl-3-oxo-3,4- dihydroquinoxalinyl)ethyl)-1,2,4-triazoline-3,5-dione (DMEQ-TAD) as previously described [33]. Analysis of extracted sera and SF vitamin D metabolites was performed on a Waters ACQUITY ultra performance liquid chromatography (UPLC) coupled to a Waters Xevo TQ-XS mass spectrometer. The LC-MS/MS method has been validated on US Food and Drug Administration guidelines for analysis of these metabolites.
Quantification of vitamin D binding protein and albumin, and estimation of free and bioavailable 25(OH)D3
Vitamin D binding protein (DBP) and albumin were quantified in RA and ReA serum and SF using VDBP (Immunodiagnostik, Bensheim, Germany) and Human Serum Albumin ELISA kits (Thermo Fisher, Runcorn, UK) respectively. All reagents supplied were diluted to the appropriate concentrations as stated in the manufacturers’ protocols and all steps were carried out at room temperature. Serum and SF concentrations of ‘free’ (not bound to any serum protein) and ‘bioa- vailable’ (not bound to DBP) 25(OH)D were estimated using values for total 25(OH)D, DBP and albumin, and mathematical algorithms adapted from the original calculations [34,35].
Statistics
All statistical analyses were carried out using GraphPad PRISM Version 6.07 software (San Diego, CA, USA). Normality was assessed using D’Agostino-Pearson omnibus normality test, with multifactorial data compared using either one-way ANOVA (parametric) or Kruskal-Wallis test (non-parametric) based on ranks, with Tukey or Dunn’s method used as for post hoc multiple-comparison procedures respectively.
Results
Serum vitamin D metabolites in RA and healthy controls
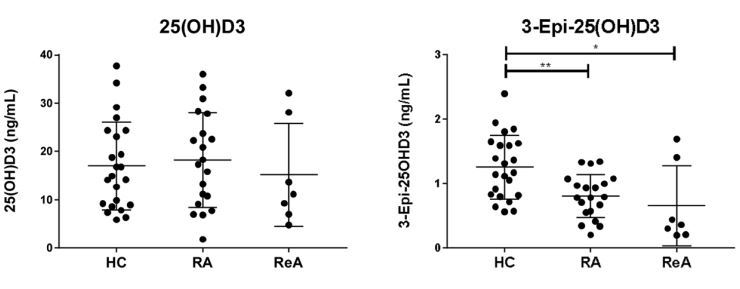
Data in Fig. 1 showed that of the vitamin D metabolites analysed, only 3-epi-25(OH)D3 was significantly different in serum from RA (median 0.788 ng/ml, 96% confidence limits 0.573-0.997, p < 0.01) and ReA (median 0.361 ng/ml, 98% confidence limits 0.197-1.69, p < 0.05) patients relative to healthy controls (HC) (median 1.17 ng/ ml, 97% confidence limits 0.832-1.59). Data for serum vitamin D metabolites in RA, ReA and HC were unaffected by donor gender, age, or duration of disease (data not shown). Serum concentrations of 25(OH) D3 for RA patients showed strong correlation with other serum vitamin D metabolites, apart from 25(OH)D2 which showed a trend to inverse correlation with 25(OH)D3 (Supplemental Fig. 1).
Serum versus SF vitamin D metabolites in RA and ReA
With the exception of 25(OH)D2, all vitamin D metabolites showed significantly lower concentrations in SF vs paired serum from RA patients (Fig. 2A). However, only SF 25(OH)D3 and 3-epi-25(OH)D3 showed significant correlation with serum levels of the same metabolites (Fig. 2B). Furthermore, in contrast to RA serum vitamin D metabolite data, only 3-Epi-25(OH)D3 showed correlation with 25(OH)D3 in SF (Supplemental Fig. 1). In patients with ReA, 25(OH)D3, 25,25(OH)2D3 and 1,25(OH)2D3 were statistically lower in SF vs paired serum (Supplemental Fig. 2A), while only 25(OH)D3 and 24,25(OH)2D3 in ReA SF correlated significantly with the same metabolites in serum (Supplemental Fig. 2B).
DBP, albumin and free vs bioavailable serum and SF vitamin D
Concentrations of DBP were significantly lower in RA SF (median 33 pg/ml, 95% confidence limits 24.37-43.19) and ReA SF (median 35.82, 95% confidence limits 19.25-77.98) relative paired serum for RA (median 117 pg/ml, 95% confidence limits 95.38-149.8, p < 0.001) and ReA (median 85.66, 95% confidence limits 68.48-103.1, p = 0.0156) patients (Fig. 4). Albumin concentrations were also significantly lower in RA SF (median 1.423 g/dL, 95% confidence limits 1.398-1.712) vs paired serum (median 2.22 g/dL, 95% confidence limits 2.054-2.602, p = 0.0005).
Table 1 RA and ReA patient demographics and disease scores.
Data for patients with active rheumatoid arthritis (RA) (n = 20) and resolving reactive arthritis (ReA) (n = 7), including age of patient, disease duration, and disease severity and progression markers tender joint count (TJC); swollen joint count (SJC); erythrocyte sedimentation rate (ESR); c-reactive protein (CRP); diagnosis and medication. DMARDs (disease-modifying anti-rheumatic drugs), NSAIDs (non-steroidal anti-inflammatory drugs), steroids and biological drugs taken by each patient prior to study entry are also listed. N/A refers to missing data.
Based on DBP and albumin levels and total 25(OH)D3 values it was possible to calculate free 25(OH)D3 (not bound to DBP or albumin) and bioavailable 25(OH)D3 (not bound to DBP) for serum and SF samples. These values showed no significant difference between RA and ReA serum. Furthermore, in contrast to the decreased total serum 25(OH)D3 observed for SF versus serum (see Fig. 1), free 25(OH)D3 levels were significantly higher in RA SF compared to RA serum (p = 0.0046). Bioavailable 25(OH)D3 showed no significant variations in RA vs ReA for either serum or SF (Fig. 3).
Fig. 1. Rheumatoid arthritis and the serum vitamin D metabolome.
Serum from n = 20 persistent RA (RA) patients, n = 7 reactive arthritis (ReA) patients and n = 23 healthy controls (HC) were analysed by LC-MS/MS for: 25(OH)D3 (ng/mL); 3-epi-25(OH)D3 (ng/mL); 25(OH)D2 (ng/mL); 24,25(OH)2D3 (ng/mL); 1,25(OH)2D3 (pg/mL). Data shown are mean ± SD. * = significantly different HC vs RA, p < 0.05, ** p < 0.01. Mann-Whitney T-test for unpaired samples was performed for statistical analysis.
Fig. 2. Serum vs SF vitamin D metabolites in patients with persistent RA.
Comparison of serum and synovial fluid [2] concentrations for: 25(OH)D3; 3-epi-25(OH)D3; 25(OH)D2; 24,25(OH)2D3 (ng/mL); 1,25(OH)2D3 (pg/mL). B) Correlation of serum and SF concentrations for: 25(OH)D3; 3-epi-25(OH)D3; 25(OH)D2; 24,25(OH)2D3 (all ng/mL); 1,25(OH)2D3 (pg/mL). n = 20 matched for serum and SF (One-way ANOVA). Linear regression was performed by best-fit values ± SD. Coefficient of determination (r2) values are shown for correlations that were statistically significant, p < 0.05.
Fig. 3. DBP, albumin and free versus bioavailable 25(OH)D in serum and SF.
Serum and SF from n = 20 RA and n = 7 ReA patients were used to measure concentrations of DBP and albumin, and calculate free (not bound to DBP or albumin) and bioavailable (not bound to DBP) 25(OH)D. Data shown are mean ± SD. Statistical significance = p < 0.05; p < 0.01; * p < 0.001; ***p < 0.0001. Wilcoxon matched-pairs signed rank test was performed for matched serum vs SF samples. Mann-Whitney unpaired t-test was performed for non-matched samples.
Vitamin D metabolites, DBP, albumin and disease activity in RA
Specific vitamin D metabolite concentrations were studied in relation to measures of RA disease activity. Significant correlations are shown in Fig. 4. In SF, 25(OH)D3 and 3-epi-25(OH)D3 correlated inversely with swollen joint score (SJC), which also correlated inversely with SF DBP levels. However, serum 25(OH)D2 correlated directly with serum levels of c-reactive protein (CRP), and serum 25(OH)D2 also correlated with SJC. The relationship between serum or SF 25(OH)D3, 25(OH)D2, 3-epi-25(OH)D3, 24,25(OH)2D3 and 1,25(OH)2D3 and markers of RA disease (CRP, ESR, SJC and TJC) in RA patients is shown in full in Supplemental Figures 3-6.
Serum DBP was positively correlated with CRP and inversely correlated with SJC, only in the SF of RA patients (Fig. 4). None of the other disease markers showed significant correlation with DBP concentrations in either serum or SF (Supplemental Figure 7). Albumin was not significantly correlated with any disease markers (Supplemental Figure 8).
Discussion
Low serum concentrations of 25(OH)D3 have been reported to be a feature of inflammatory diseases such as RA [36-39], although some studies have indicated that the vitamin D status of RA patients is not necessarily lower than in healthy controls [38]. Consistent with the latter report, in the current study serum concentrations of 25(OH)D3 were not significantly different between RA and ReA patients and healthy controls, although all groups showed mean values that were lower than 50 nmol/L, the vitamin D-sufficiency level proposed by the Institute of Medicine [40].
Notably the one vitamin D metabolite that was significantly lower in serum from RA and ReA patients relative to healthy controls was 3-epi-25(OH)D3 , but the potential impact of this is unclear. Although 3-epi-25(OH)D3 was initially observed in serum from neonates, recent studies have shown that this metabolite is present in adult serum at levels which vary between 0 and 25% of 25(OH)D3 in the same sample [41]. The precise function of 3-epi-25(OH)D3 in vitamin D physiology is still unclear, and interpretation of data is complicated by the fact that levels of this metabolite are closely linked to serum 25(OH)D3. It is also unclear what contribution serum or SF vitamin D metabolites may make to the resolution of inflammatory disease in ReA patients, although it is important to recognize that the current study was limited by the low numbers of patients with this form of inflammatory arthritis.
A key observation in the current study is the lower vitamin D metabolite concentrations in SF versus paired serum samples.
For 25(OH) D3 the mean ratio of serum/SF was 1.74,
whilst for 1,25(OH)2D3 this was 6.07.
Similar observations have been reported previously for 25(OH)D3 [42,43], although levels of 25(OH)D3 in serum and SF in these studies varied considerably. By contrast, previous analysis of 1,25(OH)2D3 in RA patients reported similar [44], or modestly suppressed [43] concentrations in SF relative to serum. One possible explanation for significantly lower levels of SF 1,25(OH)2D3 in the current study is the use of more reliable LC-MS/MS technology for quantification of 1,25(OH)2D3, compared to the relatively non-specific radioreceptor [44] or radioimmunoassays [43] used to measure 1,25(OH)2D3 in the previous studies. Previous studies also reported strong correlation between serum and SF 1,25(OH)2D3, and between SF 25(OH)D3 and SF 1,25(OH)2D3 [43], which we did not observe in the current study.
In common with previous studies we were unable to analyse SF from control HC subjects, and thus it is unclear whether decreased 1,25(OH)2D3 is a feature of SF that is unaffected by inflammatory disease.
It is also unclear whether SF 1,25(OH)2D3 is a more informative marker of vitamin D function than more conventionally measured 25(OH)D3.
SF 1,25(OH)2D3 did not appear to correlate with RA disease activity, whereas
SF 25(OH)D3,
3-epi-25(OH)D3 and
DBP
were all inversely related to inflammation markers. One challenge with the interpretation of these data is that measures of inflammation where at the ‘whole patient’ level (i.e. systemic inflammatory markers or total joint counts) rather than at the level of the individual joint from which synovial fluid was studied and in which levels of vitamin D metabolites were measured. However, our data linking 25(OH)D3 rather than 1,25(OH)2D3 with RA inflammation scores would endorse the proposed intracrine model for SF metabolism of 25(OH)D3 [45], with locally synthesized 1,25(OH)2D3 acting to promote anti-inflammatory responses and inhibit disease markers independent of serum 1,25(OH)2D3.
The inference from previous studies is that inflammatory factors promote enhanced extra-renal synthesis of 1,25(OH)2D3 via immune cell activity of 1a-hydroxylase [45]. Based on this model, enhanced vitamin D status (higher serum levels of 25(OH)D3), leads to increased SF 25(OH)D3, and this in turn promotes SF production of 1,25(OH)2D3. Data in the current study do not appear to support this mechanism, with SF (OH)2D3 being below the lower limit of quantification in 13/20 RA samples, and 4/7 ReA samples. This may reflect the efficacy of the antiinflammatory therapies currently used to manage RA, with several of the treatments used in the current study such as hydroxychloroquine being potential inhibitors of the 1a-hydroxylase enzyme involved in SF synthesis of 1,25(OH)2D3 [45,46]. However, analysis of treated versus non-treated RA samples, including steroidal versus non-steroidal therapies, did not reveal any significant differences in serum or synovial fluid 25(OH)D3 or 1,25(OH)2D3 (data not shown). It is also possible that synthesis of 1,25(OH)2D3 within SF is tightly regulated and limited to the immediate immune microenvironment. This would maximize anti-inflammatory responses to 1,25(OH)2D3, whilst limiting the potential for spill-over of 1,25(OH)2D3 into the general circulation.
As well as studying the impact of vitamin D metabolites on RA disease, serum and SF levels of DBP and albumin were also measured. Interestingly SF DBP correlated inversely with SJC. This may simply reflect the association between DBP and SF concentrations of 25(OH) D3, but 25(OH)D3-independent DBP activities have also been described, including effects as a macrophage-activation factor [47], and actin-binding capacity [48], that have the potential to influence RA disease-severity and progression. In common with most vitamin D metabolites, and consistent with previous publications [42], concentrations of DBP in RA and ReA were lower in patient SF relative to serum. We also observed lower concentrations of albumin in SF as previously reported [49]. The net effect of this is that calculated levels of 25(OH)D3 not bound to DBP or albumin (free 25(OH)D3) and 25(OH)D3 not bound to DBP (bioavailable 25(OH)D3) were the same for RA and ReA SF as paired serum values.
This suggests that there is rheostatic regulation of vitamin D binding proteins in SF to maximize free or bioavailable 25(OH)D3, but it is important to recognise that the values presented in the current study are calculated estimates of unbound 25(OH)D3. We were unable to directly measure free 25(OH)D3 as recently described in other studies [50], and it is possible that other factors present in SF may significantly enhance or impede protein binding of 25(OH)D3. Further studies are required to determine more accurately the proportions of bound and unbound 25(OH)D3 in SF from RA patients.
Although 25(OH)D3 has been shown to promote anti-bacterial and anti-inflammatory activity via intracrine or paracrine pathways involving immune cell synthesis of 1,25(OH)2D3, the precise mechanism by which 25(OH)D3 gains access to immune cells expressing 1a-hydro- xylase is far from clear. Cells such as macrophages [5] and dendritic cells [6] express 1a-hydroxylase, but do not appear to express the DBP receptor megalin [51]. In this setting, synthesis of 1,25(OH)2D3, and associated immune function, may be dependent on macrophage uptake of 25(OH)D3 that is not bound to DBP (free or bioavailable 25(OH)D3) [51]. Due to the lipophilic nature of 25(OH)D3 it is thought that its biological actions are exerted through the unbound 25(OH)D that is able to diffuse across cell membranes - the so-called free hormone hypothesis [52,53]. In the current study we were unable to demonstrate that free or bioavailable 25(OH)D3 are better correlates of RA disease than the more conventionally measured total 25(OH)D3 (data not shown). However, the fact that the limited inverse correlations with RA disease that we identified involved SF rather than serum further supports the view that circulating concentrations of vitamin D provide only a limited perspective on the extra-skeletal actions of vitamin D. This appears to be particularly true in diseases such as RA where tissue- specific levels of 25(OH)D3 may be crucial in defining the anti-inflammatory potential of vitamin D.
Appendix A. Supplementary data
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.jsbmb.2018.10.008.
References
M. Hewison, Antibacterial effects of vitamin D, Nat. Rev. Endocrinol. 7 (2011) 436.
M. Hewison, An update on vitamin D and human immunity, Clin. Endocrinol. (Oxf) 76 (2012) 315-325.
M. Kreutz, R. Andreesen, S.W. Krause, A. Szabo, E. Ritz, H. Reichel, 1,25-dihy- droxyvitamin D3 production and vitamin D3 receptor expression are developmentally regulated during differentiation of human monocytes into macrophages, Blood 82 (1993) 1300-1307.
M. Hewison, L. Freeman, S.V. Hughes, K.N. Evans, R. Bland, A.G. Eliopoulos, M.D. Kilby, P.A. Moss, R. Chakraverty, Differential regulation of vitamin D receptor and its ligand in human monocyte-derived dendritic cells, J. Immunol. 170 (2003) 5382-5390.
P.T. Liu, S. Stenger, H. Li, L. Wenzel, B.H. Tan, S.R. Krutzik, M.T. Ochoa, J. Schauber, K. Wu, C. Meinken, D.L. Kamen, M. Wagner, R. Bals, A. Steinmeyer, U. Zugel, R.L. Gallo, D. Eisenberg, M. Hewison, B.W. Hollis, J.S. Adams, B.R. Bloom, R.L. Modlin, Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response, Sci. (New York, N.Y.) 311 (2006) 1770-1773.
J. Bacchetta, J.J. Zaritsky, J.L. Sea, R.F. Chun, T.S. Lisse, K. Zavala, A. Nayak, K. Wesseling-Perry, M. Westerman, B.W. Hollis, I.B. Salusky, M. Hewison, Suppression of iron-regulatory hepcidin by vitamin d, J. Am. Soc. Nephrol. 25 (2014) 564-572.
G. Penna, S. Amuchastegui, N. Giarratana, K.C. Daniel, M. Vulcano, S. Sozzani, Adorini, 1,25-Dihydroxyvitamin D3 selectively modulates tolerogenic properties in myeloid but not plasmacytoid dendritic cells, J. Immunol. 178 (2007) 145-153.
L.E. Jeffery, A.M. Wood, O.S. Qureshi, T.Z. Hou, D. Gardner, Z. Briggs, S. Kaur, K. Raza, D.M. Sansom, Availability of 25-Hydroxyvitamin D3 to APCs Controls the Balance between Regulatory and Inflammatory T Cell Responses, J. Immunol. 189 (2012) 5155-5164.
E.M. Colin, P.S. Asmawidjaja, J.P. van Hamburg, A.M. Mus, M. van Driel, J. M. Hazes, J.P. van Leeuwen, E. Lubberts, 1,25-dihydroxyvitamin D3 modulates Th17 polarization and interleukin-22 expression by memory T cells from patients with early rheumatoid arthritis, Arthritis Rheum. 62 (2010) 132-142.
S. Joshi, L.C. Pantalena, X.K. Liu, S.L. Gaffen, H. Liu, C. Rohowsky-Kochan, K. Ichiyama, A. Yoshimura, L. Steinman, S. Christakos, S. Youssef, 1,25-dihydrox- yvitamin D(3) ameliorates Th17 autoimmunity via transcriptional modulation of interleukin-17A, Mol. Cell. Biol. 31 (2011) 3653-3669.
L.E. Jeffery, O.S. Qureshi, D. Gardner, T.Z. Hou, Z. Briggs, B. Soskic, J. Baker, K. Raza, D.M. Sansom, Vitamin D antagonises the suppressive effect of inflammatory cytokines on CTLA-4 expression and regulatory function, PLoS One 10 (2015) e0131539.
L.E. Jeffery, F. Burke, M. Mura, Y. Zheng, O.S. Qureshi, M. Hewison, L.S. Walker, D.A. Lammas, K. Raza, D.M. Sansom, 1,25-Dihydroxyvitamin D(3) and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing CTLA-4 and FoxP3, J. Immunol. 183 (2009) 5458-5467.
M. Froicu, V. Weaver, T.A. Wynn, M.A. McDowell, J.E. Welsh, M.T. Cantorna, A crucial role for the vitamin D receptor in experimental inflammatory bowel diseases, Mol. Endocrinol. 17 (2003) 2386-2392.
N. Liu, L. Nguyen, R.F. Chun, V. Lagishetty, S. Ren, S. Wu, B. Hollis, H.F. DeLuca, J.S. Adams, M. Hewison, Altered endocrine and autocrine metabolism of vitamin D in a mouse model of gastrointestinal inflammation, Endocrinology 149 (2008) 4799-4808.
K. Zwerina, W. Baum, R. Axmann, G.R. Heiland, J.H. Distler, J. Smolen, S. Hayer, J. Zwerina, G. Schett, Vitamin D receptor regulates TNF-mediated arthritis, Ann. Rheum. Dis. 70 (2011) 1122-1129.
V. Lagishetty, A.V. Misharin, N.Q. Liu, T.S. Lisse, R.F. Chun, Y. Ouyang, S.M. McLachlan, J.S. Adams, M. Hewison, Vitamin D deficiency in mice impairs colonic antibacterial activity and predisposes to colitis, Endocrinology 151 (2010) 2423-2432.
M.T. Cantorna, C.E. Hayes, H.F. DeLuca, 1,25-Dihydroxyvitamin D3 reversibly blocks the progression of relapsing encephalomyelitis, a model of multiple sclerosis, Proc. Natl. Acad. Sci. U.S.A. 93 (1996) 7861-7864.
A. Raghuwanshi, S.S. Joshi, S. Christakos, Vitamin D and multiple sclerosis, J. Cell. Biochem. 105 (2008) 338-343.
C. Mathieu, K. Badenhoop, Vitamin D and type 1 diabetes mellitus: state of the art, Trends Endocrinol. Metab. 16 (2005) 261-266.
M.T. Cantorna, Vitamin D, multiple sclerosis and inflammatory bowel disease, Arch. Biochem. Biophys. 523 (2012) 103-106.
Q. Hong, J. Xu, S. Xu, L. Lian, M. Zhang, C. Ding, Associations between serum 25- hydroxyvitamin D and disease activity, inflammatory cytokines and bone loss in patients with rheumatoid arthritis, Rheumatology 53 (2014) 1994-2001.
Y.E. Park, B.H. Kim, S.G. Lee, E.K. Park, J.H. Park, S.H. Lee, G.T. Kim, Vitamin D status of patients with early inflammatory arthritis, Clin. Rheumatol. 34 (2015) 239-246.
M. Di Franco, I. Barchetta, C. Iannuccelli, M.C. Gerardi, S. Frisenda, F. Ceccarelli, Valesini, M.G. Cavallo, Hypovitaminosis D in recent onset rheumatoid arthritis is predictive of reduced response to treatment and increased disease activity: a 12 month follow-up study, BMC Musculoskelet. Disord. 16 (2015) 53.
J.T. Sharp, F. Wolfe, D.M. Mitchell, D.A. Bloch, The progression of erosion andjoint space narrowing scores in rheumatoid arthritis during the first twenty-five years of disease, Arthritis Rheum. 34 (1991) 660-668.
P. Ahvonen, K. Sievers, K. Aho, Arthritis associated with Yersinia enterocolitica infection, Acta Rheumatol. Scand. 15 (1969) 232-253.
R.D. Inman, Classification criteria for reactive arthritis, J. Rheumatol. 26 (1999) 1219-1221.
J. Sieper, Pathogenesis of reactive arthritis, Curr. Rheumatol. Rep. 3 (2001) 412-418.
F.A. Cooles, A.G. Pratt, D.W. Lendrem, W.F. Ng, T.J. Aspray, J.D. Isaacs, Retrospective analysis of the role of serum vitamin D in early rheumatic disease, Rheumatology 54 (2015) 374-375.
T. Pincus, B. Richardson, V. Strand, M.J. Bergman, Relative efficiencies of the 7 rheumatoid arthritis core data set measures to distinguish active from control treatments in 9 comparisons from clinical trials of 5 agents, Clin. Exp. Rheumatol. 32 (2014) S-47-54.
C. Jenkinson, A.E. Taylor, Z.K. Hassan-Smith, J.S. Adams, P.M. Stewart, M. Hewison, B.G. Keevil, High throughput LC-MS/MS method for the simultaneous analysis of multiple vitamin D analytes in serum, J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 1014 (2016) 56-63.
J.A. Tamblyn, R. Susarla, C. Jenkinson, L.E. Jeffery, O. Ohizua, R.F. Chun, S.Y. Chan, M.D. Kilby, M. Hewison, Dysregulation of maternal and placental vitamin D metabolism in preeclampsia, Placenta 50 (2017) 70-77.
C. Jenkinson, A.E. Taylor, Z.K. Hassan-Smith, J.S. Adams, P.M. Stewart, M. Hewison, B.G. Keevil, High throughput LC-MS/MS method for the simultaneous analysis of multiple vitamin D analytes in serum, J. Chromatogr. B 1014 (2016) 56-63.
M. Kaufmann, J.C. Gallagher, M. Peacock, K.P. Schlingmann, M. Konrad, F. DeLuca, R. Sigueiro, B. Lopez, A. Mourino, M. Maestro, R. St-Arnaud, J. S. Finkelstein, D.P. Cooper, G. Jones, Clinical utility of simultaneous quantitation of 25-hydroxyvitamin D and 24,25-dihydroxyvitamin D by LC-MS/MS involving derivatization with DMEQ-TAD, J. Clin. Endocrinol. Metab. 99 (2014) 2567-2574.
J. Arnaud, J. Constans, Affinity differences for vitamin D metabolites associated with the genetic isoforms of the human serum carrier protein (DBP), Hum. Genet. 92 (1993) 183-188.
R.F. Chun, B.E. Peercy, J.S. Adams, M. Hewison, Vitamin D binding protein and monocyte response to 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D: analysis by mathematical modeling, PLoS One 7 (2012) e30773.
Y.H. Lee, S.C. Bae, Vitamin D level in rheumatoid arthritis and its correlation with the disease activity: a meta-analysis, Clin. Exp. Rheumatol. 34 (2016) 827-833.
J. Lin, J. Liu, M.L. Davies, W. Chen, Serum vitamin D level and rheumatoid arthritis disease activity: review and meta-analysis, PLoS One 11 (2016) e0146351.
S. Cecchetti, Z. Tatar, P. Galan, B. Pereira, C. Lambert, G. Mouterde, A. Sutton, M. Soubrier, M. Dougados, Prevalence of vitamin D deficiency in rheumatoid arthritis and association with disease activity and cardiovascular risk factors: data from the COMEDRA study, Clin. Exp. Rheumatol. 34 (2016) 984-990.
J. Vojinovic, A. Tincani, A. Sulli, S. Soldano, L. Andreoli, F. Dall’Ara, R. Ionescu, K. S. Pasalic, I. Balcune, I. Ferraz-Amaro, M. Tlustochowicz, I. Butrimiene, E. Punceviciene, N. Toroptsova, S. Grazio, J. Morovic-Vergles, P. Masaryk, K. Otsa, M. Bernardes, V. Boyadzhieva, F. Salaffi, M. Cutolo, European multicentre pilot survey to assess vitamin D status in rheumatoid arthritis patients and early development of a new Patient Reported Outcome questionnaire (D-PRO), Autoimmun. Rev. 16 (2017) 548-554.
A.C. Ross, J.E. Manson, S.A. Abrams, J.F. Aloia, P.M. Brannon, S.K. Clinton, R.A. Durazo-Arvizu, J.C. Gallagher, R.L. Gallo, G. Jones, C.S. Kovacs, S.T. Mayne, C.J. Rosen, S.A. Shapses, The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know, J. Clin. Endocrinol. Metab. 96 (2011) 53-58.
G. Lensmeyer, M. Poquette, D. Wiebe, N. Binkley, The C-3 epimer of 25-hydro- xyvitamin D(3) is present in adult serum, J. Clin. Endocrinol. Metab. 97 (2012) 163-168.
A. Fairney, A.M. Straffen, C. May, M.H. Seifert, Vitamin D metabolites in synovial fluid, Ann. Rheum. Dis. 46 (1987) 370-374.
E.B. Mawer, M.E. Hayes, P.E. Still, M. Davies, G.A. Lumb, J. Palit, P.J. Holt, Evidence for nonrenal synthesis of 1,25-dihydroxyvitamin D in patients with inflammatory arthritis, J. Bone Miner. Res. 6 (1991) 733-739.
M. Inaba, K. Yukioka, Y. Furumitsu, M. Murano, H. Goto, Y. Nishizawa, H. Morii, Positive correlation between levels of IL-1 or IL-2 and 1,25(OH)2D/25-OH-D ratio in synovial fluid of patients with rheumatoid arthritis, Life Sci. 61 (1997) 977-985.
M.E. Hayes, J. Denton, A.J. Freemont, E.B. Mawer, Synthesis of the active metabolite of vitamin D, 1,25(OH)2D3, by synovial fluid macrophages in arthritic diseases, Ann. Rheum. Dis. 48 (1989) 723-729.
J.S. Adams, M.M. Diz, O.P. Sharma, Effective reduction in the serum 1,25-dihy- droxyvitamin D and calcium concentration in sarcoidosis-associated hypercalcemia with short-course chloroquine therapy, Ann. Intern. Med. 111 (1989) 437-438.
N. Yamamoto, V.R. Naraparaju, Role of vitamin D3-binding protein in activation of mouse macrophages, J. Immunol. 157 (1996) 1744-1749.
P.T. Gomme, J. Bertolini, Therapeutic potential of vitamin D-binding protein, Trends Biotechnol. 22 (2004) 340-345.
J.P. Binette, K. Schmid, The proteins of synovial fluid: a study of the alpha 1-alpha 2 globulin ratio, Arthritis Rheum. 8 (1965) 14-28.
C.M. Nielson, K.S. Jones, R.F. Chun, J.M. Jacobs, Y. Wang, M. Hewison, J.S. Adams, C.M. Swanson, C.G. Lee, D. Vanderschueren, S. Pauwels, A. Prentice, R.D. Smith, T. Shi, Y. Gao, A.A. Schepmoes, J.M. Zmuda, J. Lapidus, J.A. Cauley, R. Bouillon, Schoenmakers, E.S. Orwoll, Free 25-Hydroxyvitamin D: impact of vitamin D binding protein assays on racial-genotypic associations, J. Clin. Endocrinol. Metab. 101 (2016) 2226-2234.
R.F. Chun, A.L. Lauridsen, L. Suon, L.A. Zella, J.W. Pike, R.L. Modlin, A.R. Martineau, R.J. Wilkinson, J. Adams, M. Hewison, Vitamin D-binding protein directs monocyte responses to 25-hydroxy- and 1,25-dihydroxyvitamin D, J. Clin. Endocrinol. Metab. 95 (2010) 3368-3376.
D.D. Bikle, E. Gee, B. Halloran, J.G. Haddad, Free 1,25-dihydroxyvitamin D levels in serum from normal subjects, pregnant subjects, and subjects with liver disease, J. Clin. Invest. 74 (1984) 1966-1971.
R. Bouillon, F.A. Van Assche, H. Van Baelen, W. Heyns, P. De Moor, Influence of the vitamin D-binding protein on the serum concentration of 1,25-dihydroxyvitamin D3. Significance of the free 1,25-dihydroxyvitamin D3 concentration, J. Clin. Invest. 67 (1981) 589-596.
