Hospital-acquired pneumonia treated by vitamin D if person was deficient – RCT
Effect of adjunctive single high-dose vitamin D3 on outcome of community-acquired pneumonia in hospitalised adults: The VIDCAPS randomised controlled trial.
Sci Rep. 2018 Sep 14;8(1):13829. doi: 10.1038/s41598-018-32162-2.
Slow S1, Epton M2, Storer M2, Thiessen R3, Lim S3, Wong J4,5, Chin P4,6, Tovaranonte P4,7, Pearson J8, Chambers ST8, Murdoch DR8; VIDCAPS Group.
 CAP Death with low Vitamin D
* Pneumonia acquired in hospital – 3X more likely to die if low vitamin D – June 2015
* Pneumonia patients 3 X more likely to die if low vitamin D – meta-analysis Sept 2017
* Severe Pneumonia (CAP) results in death 2 times more often if very low vitamin D – July 2016
CAP Children
* Pneumonia is increasing (now 1 in 6 child deaths), more vitamin D studies needed – June 2018
* Pneumonia in Egyptian Children 3.6 X more likely if poor Vitamin D Receptor – Aug 2018
---
1. Breathing category starts with the following
{include}
CAP Death with low Vitamin D
* Pneumonia acquired in hospital – 3X more likely to die if low vitamin D – June 2015
* Pneumonia patients 3 X more likely to die if low vitamin D – meta-analysis Sept 2017
* Severe Pneumonia (CAP) results in death 2 times more often if very low vitamin D – July 2016
CAP Children
* Pneumonia is increasing (now 1 in 6 child deaths), more vitamin D studies needed – June 2018
* Pneumonia in Egyptian Children 3.6 X more likely if poor Vitamin D Receptor – Aug 2018
---
1. Breathing category starts with the following
{include}
📄 Download the PDF from VitaminDWiki
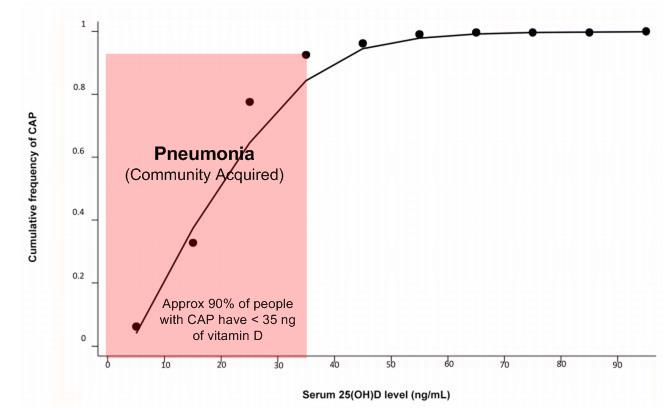
Note: (XX) XX =%
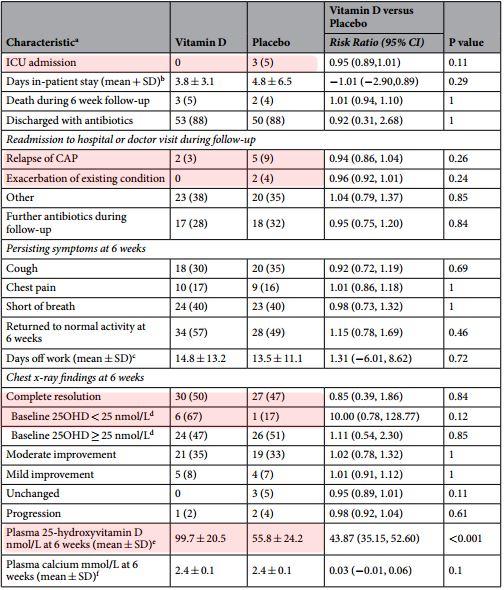
Low vitamin D status is associated with increased risk of pneumonia, greater disease severity and poorer outcome. However, no trials have examined the effect of adjunctive vitamin D therapy on outcomes in adults with community-acquired pneumonia (CAP). We conducted a randomised, double-blind, placebo-controlled trial examining the effects of adjunctive vitamin D in adults hospitalised with CAP. Participants were randomised to either a single oral dose of 200,000 IU vitamin D3 or placebo. The primary outcome was the complete resolution of chest radiograph infiltrate at 6 weeks post-study treatment. Secondary outcomes included length of hospital stay, intensive care admission and return to normal activity. Only participants who completed the study or died within the 6 week period were included in the analysis (n = 60 vitamin D, n = 57 placebo). Adjunctive vitamin D did not have any effect on the primary outcome (OR 0.78, 95% CI 0.31 to 1.86, p = 0.548).
However, there was evidence it increased the complete resolution of pneumonia in participants with baseline vitamin D levels <25 nmol/L (OR 17.0, 95% CI 1.40-549.45, P = 0.043), but this did not reach statistical significance using exact methods (OR 13.0, 95%CI 0.7-960.4, P = 0.083). There were no significant effects for any secondary outcome.
