Autoimmune system, Dendric cells and active Vitamin D (technical)
1,25-Dihydroxyvitamin D3 Restrains CD4+ T Cell Priming Ability of CD11c+ Dendritic Cells by Upregulating Expression of CD31
Front. Immunol., 28 March 2019 | https://doi.org/10.3389/fimmu.2019.00600
Louise Saul1†, Iris Mair1†, Alasdair Ivens2, Pamela Brown3, Kay Samuel4, John D. M. Campbell4, Daniel Y. Soong5, Nadine Kamenjarin1 and Richard J. Mellanby1,6*
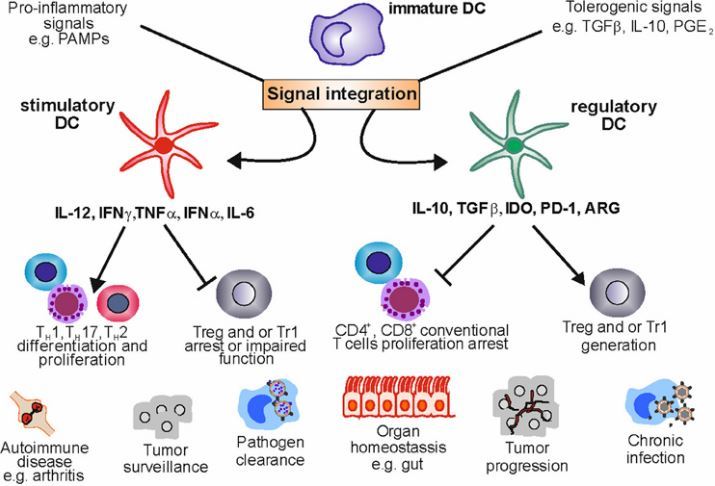
 1. Regulatory dendritic cells: there is more than just immune activation
Sept 2012, free PDF, https://doi.org/10.3389/fimmu.2012.00274
1. Regulatory dendritic cells: there is more than just immune activation
Sept 2012, free PDF, https://doi.org/10.3389/fimmu.2012.00274
 ---
1. Study described - based on a press release which highlighted MS, while MS was only mentioned once in the study
* Immune study sheds light on vitamin D effects Medical Press
* Vitamin D deficiency linked to increased susceptibility to MS Press Release for this study - Irish Times
* "Low vitamin D is implicated as risk factor for development of autoimmune diseases"
* " Vitamin D can dramatically affect the immune system and make people less susceptible to diseases such as multiple sclerosis, scientists have discovered."
* "Researchers at the University of Edinburgh say their study has shed light on how vitamin D deficiency may influence the risk of autoimmune diseases."
* Vitamin D helps the body fight off diseases such as MS, study finds Metro UK
* Vitamin D deficiency may make people more vulnerable to Multiple Sclerosis
---
1. Study described - based on a press release which highlighted MS, while MS was only mentioned once in the study
* Immune study sheds light on vitamin D effects Medical Press
* Vitamin D deficiency linked to increased susceptibility to MS Press Release for this study - Irish Times
* "Low vitamin D is implicated as risk factor for development of autoimmune diseases"
* " Vitamin D can dramatically affect the immune system and make people less susceptible to diseases such as multiple sclerosis, scientists have discovered."
* "Researchers at the University of Edinburgh say their study has shed light on how vitamin D deficiency may influence the risk of autoimmune diseases."
* Vitamin D helps the body fight off diseases such as MS, study finds Metro UK
* Vitamin D deficiency may make people more vulnerable to Multiple Sclerosis
📄 Download the PDF from VitaminDWiki
Dendritic cells (DC) are specialized sentinel cells that bridge the innate and adaptive immune response and play a crucial role in shaping the adaptive immune response. Vitamin D, a known epidemiological risk factor for the development of several autoimmune diseases, influences the development of dendritic cells. Consequently, vitamin D metabolites are frequently used in protocols to develop therapeutic dendritic cell therapies for autoimmune diseases. However, the mechanisms by which vitamin D modulates DC function remain poorly understood. We investigated the effects of vitamin D on murine CD11c+ bone marrow derived DC (BMDC) function by analyzing global gene expression in CD11c+ BMDC generated in the presence (VitD-CD11c+BMDC) or absence (Veh-CD11c+BMDC) of the active vitamin D metabolite, 1,25-dihydroxyvitamin D3 (1,25(OH)2D3). Seven genes were significantly increased in expression in both immature and LPS-matured VitD-CD11c+BMDC, one of which was CD31, a member of the immunoglobulin superfamily. Gene knockdown of CD31 enhanced the ability of VitD-CD11c+BMDC to prime naïve CD4+ T cells in vitro; conversely, increased expression of CD31 on vehicle treated CD11c+BMDC restrained their T cell priming abilities. Time-lapse imaging of BMDC and CD4+ T cells during in vitro priming revealed that CD31 reduced the BMDC–T cell interaction time. Finally, we confirmed a similar effect of 1,25(OH)2D3 on human CD34+ cell-derived CD11c+DC, whereby DC generated in the presence of 1,25(OH)2D3 had increased CD31 expression. In summary, we show that both mouse and human DC generated in the presence of 1,25(OH)2D3 upregulate CD31 expression, resulting in a reduced ability to prime CD4+ T cells by impairing a stable cell-cell contact.
