Air pollution associated with poor bone density (less vitamin D)
See also VitaminDWiki
Air pollution while pregnant reduced vitamin D level in infant – Aug 2012
Fewer pre-term births after pollution reduction (vitamin D not mentioned) April 2014
Higher air pollution associated with 7 ng lower vitamin D levels – Aug 2013
Diesel air pollution causes liver problems (and low vitamin D) if low Omega-3 (mice) – Jan 2018
Association of air particulate pollution with bone loss over time and bone fracture risk: analysis of data from two independent studies
The Lancet Planetary Health Vol 1, No. 8, e337–e347, Nov 2017 DOI: http://dx.doi.org/10.1016/S2542-5196(17)30136-5 |
Diddier Prada, PhD, Jia Zhong, ScD, Elena Colicino, PhD, Antonella Zanobetti, PhD, Prof Joel Schwartz, PhD, Nicholas Dagincourt, MSc, Shona C Fang, PhD, Itai Kloog, PhD, Joseph M Zmuda, PhD, Prof Michael Holick, PhD, Prof Luis A Herrera, PhD, Prof Lifang Hou, PhD, Prof Francesca Dominici, PhD, Prof Benedetta Bartali, PhD, Prof Benedetta Bartali, Prof Benedetta Bartali, Prof Andrea A Baccarelli, PhD, Prof Andrea A Baccarelli, Prof Andrea A Baccarelli
📄 Download the PDF from VitaminDWiki
Study compared Air Pollution and Bone Mineral Density for North East US Zip codes
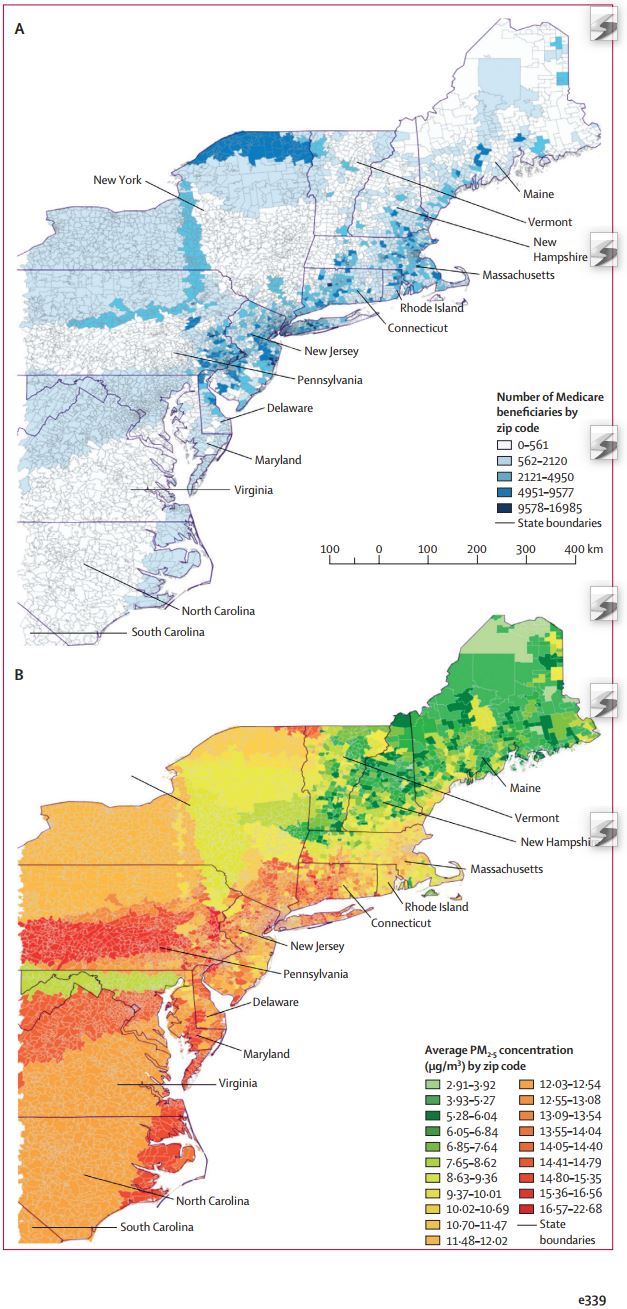
Background
Air particulate matter is a ubiquitous environmental exposure associated with oxidation, inflammation, and age-related chronic disease. Whether particulate matter is associated with loss of bone mineral density and risk of bone fractures is undetermined. We did two independent studies with complementary designs, objectives, and measures to determine the relationship between ambient concentrations of particulate matter and bone health.
Methods
In the first study, we examined the association of long-term concentrations of particulate matter less than 2·5 μm (PM2·5) and osteoporosis-related fracture hospital admissions among 9·2 million Medicare enrollees (aged ≥65 years) of the northeast-mid-Atlantic USA between January, 2003, and December, 2010. In the second study, we examined the association of long-term black carbon and PM2·5 concentrations with serum calcium homoeostasis biomarkers (parathyroid hormone, calcium, and 25-hydroxyvitamin [25(OH)D]) and annualised bone mineral density over 8 years (baseline, November, 2002–July, 2005; follow-up, June, 2010–October, 2012) of 692 middle-aged (46·7 years [SD12·3]), low-income men from the Boston Area Community Health/Bone Survey (BACH/Bone study) cohort. PM2·5 concentrations were estimated using spatiotemporal hybrid modelling including Aerosol Optical Depth data, spatial smoothing, and local predictors. Black carbon concentrations were estimated using spatiotemporal land-use regression models.
Findings
In the Medicare analysis, risk of bone fracture admissions at osteoporosis-related sites was greater in areas with higher PM2·5 concentrations (risk ratio [RR] 1·041, 95% CI 1·030 to 1·051). This risk was particularly high among low-income communities (RR 1·076, 95% CI 1·052 to 1·100). In the longitudinal BACH/Bone study, baseline black carbon and PM2·5 concentrations were associated with lower serum parathyroid hormone (β=–1·16, 95% CI −1·93 to −0·38, p=0·004, for 1 IQR increase [0·106 μg/m3] in the 1-year average of black carbon concentrations; β=–7·39, 95% CI −14·17 to −0·61, p=0·03, for 1 IQR increase [2·18 μg/m3] in the 1-year average of PM2·5 concentrations). Black carbon concentration was associated with higher bone mineral density loss over time at multiple anatomical sites, including femoral neck (−0·08% per year for 1 IQR increase, 95% CI −0·14 to −0·02) and ultradistal radius (−0·06% per year for 1 IQR increase, −0·12 to −0·01). Black carbon and PM2·5 concentrations were not associated with serum calcium or serum 25(OH)D concentrations.
Interpretation: Our results suggest that poor air quality is a modifiable risk factor for bone fractures and osteoporosis, especially in low-income communities.
Review of this study at Ney York Times
"The study in middle-aged men found that people living in locations with higher levels of air pollution had lower concentrations of parathyroid hormone and lower levels of bone mineral density."
Air pollution: a largely neglected risk factor for osteoporosis – comment
Lancet Planetary Health Vol 1, No. 8, e311–e312, Nov 2017 DOI: http://dx.doi.org/10.1016/S2542-5196(17)30143-2
Tuan V Nguyen, Tuan V Nguyen
In The Lancet Planetary Health, Diddier Prada and colleagues1 have identified an important, but heretofore largely overlooked, risk factor for osteoporosis: air pollution. The investigators showed that long-term exposure to black carbon was associated with an excess of longitudinal bone loss. They also showed that people living in areas with higher PM2·5 concentrations had a greater risk of osteoporotic fracture. Air pollution is considered the most important risk factor for mortality at the population level;2 now we know that it is also a risk factor for osteoporosis. These findings, together with a previous study3 and the development of the exposome concept,4 have pointed to a new and exciting area of research in the aetiology of osteoporosis.
To appreciate the significance of these findings, it is important to consider the complex aetiology of osteoporosis and its consequence of fragility fracture in the general population. Osteoporosis is a systemic disease of the bone that affects a large number of people worldwide. Approximately one-third of women and one-tenth of men aged 50 years and older have osteoporosis or osteopaenia.5 A traumatic fracture is a consequence of osteoporosis. Among those aged 50 years, about one in two women and one in three men will sustain a fracture during their remaining lifetime.6 It has not been well recognised that individuals with a fracture have a higher risk of risk of mortality, and the risk is greater in men than in women.7 Those who survive a fracture are more likely to sustain another fracture and develop one or more of chronic pain, increased dependence, and reduced quality of life. Taken together, osteoporosis and its consequence of fracture represent a common and serious skeletal disorder that is expected to increase in magnitude over the next few decades as populations are rapidly ageing. Globally, WHO considers osteoporosis to be the second (only to cardiovascular disease) leading health problem.
Research on osteoporosis has largely focused on two important outcomes: fracture and bone mineral density (BMD). A bone fracture occurs when a force exerted against a bone is greater than the bone can bear. Thus, reduced bone strength is the key determinant of fracture risk. Bone strength is, in turn, mainly determined by BMD and the microstructure of the bone. Low BMD is the strongest risk factor for fracture, with each SD lower BMD being associated with a 1·5–2·5-fold increase in fracture risk , 8 a magnitude of association equivalent to the association between blood pressure and stroke. Therefore, BMD is operationally used to diagnose osteoporosis. However, in postmenopausal women and older men, BMD progressively declines with advancing age. Although the average rate of decline is modest (<1% per year),9 the individual rates of bone loss are highly heterogeneous, with some individuals losing BMD excessively and some losing BMD in a gradual fashion. Moreover, excessive rate of bone loss is an important predictor of fracture risk independent of baseline BMD.10 Therefore, there are good reasons to study factors influencing the variation in bone loss and fracture risk in the population at large.
Over the past four decades, although considerable research has been devoted to biological and genetic risk factors, less attention has been paid to environmental factors. This is logical since studies of twins suggest that between 65% and 80% of the variation in BMD is determined by genetic factors.11 Intensive research over the past decade has identified more than 70 genetic variants associated with low BMD or fracture.12 However, these variants account for less than 10% of variance in BMD and fracture risk, suggesting that genetic variation is not the major determinant of osteoporosis. This fact implies that research on osteoporosis should be broadened to examine the impact of environmental factors.
In osteoporosis research, environmental factors are often understood as factors intrinsic to an individual such as lifestyle, diet, physical activity, and exposure to chronic stress. The influence of a bigger and more omnipresent factor—such as indoor and outdoor air pollution—on osteoporosis has received little attention. There is good reason to study the impact of environmental pollutants on bone, because bone is a lifetime reservoir for heavy metals ingested by diets and exposed to external environmental factors. Lead and other toxic metals, such as cadmium, mercury, and aluminium,13 are sequestered in the skeleton, binding with calcium in hydroxyapatite, as a biological ‘toxic waste dump’ throughout life. Indeed, more than 90% of the lead in the body resides in bones and teeth. Lead can exert its effect on bone directly and indirectly through decreased vitamin D concentrations, decreased bone-forming cells, and increased bone-resorbing cells. The increased production of bone-resorbing cells and reduced bone-forming cells lead to loss of bone and low bone mass (ie, osteoporosis).
Emerging empirical evidence is consistent with a relationship between environmental pollution and osteoporosis. A registry-based study in Taiwan found that greater concentrations of air pollutants (ie, CO and NO2) were associated with progressively lower BMD.3 Furthermore, exposure to lead and cadmium is adversely associated with BMD and fracture risk.14 Nevertheless, it appears from these studies1, 3 that the impact of air pollution on fracture risk is modest, with the average relative risk being less than 1·1. However, given that a large proportion of the population is exposed to air pollution, the impact of such exposure on disease is substantial. Still, the finding of Prada and colleagues adds to the growing body of literature on the influence of air pollution on bone health in the general population.
However, air pollution is only a constituent of endogenous and exogenous environmental factors that individuals are exposed to from the time of birth to the time of death. These exposures include, but are not limited to, pollutants, heavy metals, foods, drugs, and other toxic chemicals that affect tissues. The realisation of such lifetime exposures gives rise to the concept of the exposome, which is defined as the cumulative measure of all non-genetic exposures in an individual's lifetime.4 The demarcation of the effect of the exposome on diseases, including osteoporosis, is the next frontier of medical research. Fortunately, recent advances in mass spectrometry make it possible to profile the serum exposome by using metabolomics and proteomics. This unbiased quantification of environmental exposure can help discover non-genetic determinants of fracture.
In summary, osteoporosis and its consequence of fragility fracture represent one of the most important public health problems worldwide, because fracture is associated with increased mortality. It is now clear that genetic factors account for a modest proportion of fracture cases and bone density variance, suggesting that an environmental profile in the form of the exposome is likely the main driver of the disease. Conceptually, an individual's risk of fracture is grounded by the individual's genome and modified by the individual's exposome. The delineation of the interaction between genome and exposome has the potential to transform our thinking about the aetiology of osteoporosis.
I declare no competing interests.
References for comment
Prada, D, Zhong, J, Colicino, E et al. Association of air particulate pollution with bone loss over time and bone fracture risk: analysis of data from two independent studies. Lancet Planet Health. 2017; 1: e337–e347
Landrigan, PJ. Air pollution and health. Lancet Public Health. 2017; 2: e4–e5
Chang, KH, Chang, MY, Muo, CH et al. Exposure to air pollution increases the risk of osteoporosis: a nationwide longitudinal study. Medicine (Baltimore). 2015; 94: e733
Wild, CP. Complementing the genome with an “exposome”: the outstanding challenge of environmental exposure measurement in molecular epidemiology. Cancer Epidemiol Biomarkers Prev. 2005; 14: 1847–1850
Looker, AC, Melton, LJ 3rd, Harris, TB, Borrud, LG, and Shepherd, JA. Prevalence and trends in low femur bone density among older US adults: NHANES 2005–2006 compared with NHANES III. J Bone Miner Res. 2010; 25: 64–71
Nguyen, ND, Ahlborg, HG, Center, JR, Eisman, JA, and Nguyen, TV. Residual lifetime risk of fractures in women and men. J Bone Miner Res. 2007; 22: 781–788
Bliuc, D, Nguyen, ND, Milch, VE, Nguyen, TV, Eisman, JA, and Center, JR. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA. 2009; 301: 513–521
Marshall, D, Johnell, O, and Wedel, H. Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. BMJ. 1996; 312: 1254–1259
Jones, G, Nguyen, T, Sambrook, P, Kelly, PJ, and Eisman, JA. Progressive loss of bone in the femoral neck in elderly people: longitudinal findings from the Dubbo osteoporosis epidemiology study. BMJ. 1994; 309: 691–695
Nguyen, TV, Center, JR, and Eisman, JA. Femoral neck bone loss predicts fracture risk independent of baseline BMD. J Bone Miner Res. 2005; 20: 1195–1201
Pocock, NA, Eisman, JA, Hopper, JL, Yeates, MG, Sambrook, PN, and Eberl, S. Genetic determinants of bone mass in adults. A twin study. J Clin Invest. 1987; 80: 706–710
Estrada, K, Styrkarsdottir, U, Evangelou, E et al. Genome-wide meta-analysis identifies 56 bone mineral density loci and reveals 14 loci associated with risk of fracture. Nat Genet. 2012; 44: 491–501
Bellinger, DC. The protean toxicities of lead: new chapters in a familiar story. Int J Environ Res Pub Health. 2011; 8: 2593–2628
Campbell, JR and Auinger, P. The association between blood lead levels and osteoporosis among adults—results from the third national health and nutrition examination survey (NHANES III). Environ Health Perspect. 2007; 115: 1018–1022
