Vitamin D is great for dogs and cats too
Vitamin D might increase dogs' healthspan and lifespan - no trials yet
TL;DR
- No interventional study has demonstrated that vitamin D supplementation extends lifespan or healthspan in dogs or cats. Every "longevity" claim in the pet space rests on cross-sectional and retrospective associations between low 25(OH)D and disease severity/short-term mortality; causal inference is unsupported and the only canine RCTs target surrogate endpoints (pruritus, FGF-23, PTH).
- The therapeutic window is narrower than in humans, and the iatrogenic risk is real and well-documented: rodenticide LD50 ~88 mg/kg in dogs but clinical hypercalcemia at ~0.1–0.5 mg/kg; the 2019 Hill's recall (~22 million cans, 33 SKUs, premix up to ~33× the SUL, alleged hundreds of deaths) and the 2017 Crossley/Wehner case series in cats illustrate how thin the margin is. AAFCO cut the canine dietary safe upper limit from 5,000 to 3,000 IU/kg DM in 2016 because of endochondral-ossification damage in Great Dane puppies fed 4,000 IU/kg.
- For an owner feeding an AAFCO-compliant commercial diet, the evidence does not support routine supplementation above label for healthspan/lifespan. Plausible disease-targeted indications (atopic dermatitis, CKD-MBD, chronic enteropathy, possibly DCM/CHF) exist, but should be guided by serum 25(OH)D and veterinary monitoring, not by extrapolation from human RCTs.
Key Findings
Evidence grade summary
| Question | Best evidence | Grade |
|---|---|---|
| Dogs/cats depend on dietary vitamin D | In vitro + in vivo (How 1994, Morris 1999) | A (definitive) |
| Low 25(OH)D associates with disease/mortality in dogs and cats | Multiple cross-sectional + retrospective cohorts | B (consistent associations) |
| Supplementation improves a clinical endpoint in pets | One small canine atopic-dermatitis RCT (Klinger 2018, n=23 cross-over); CKD surrogate-endpoint trials | C (preliminary) |
| Supplementation extends lifespan or healthspan in pets | None | F (no direct evidence) |
| Toxicity from over-supplementation is real and dose-quantified | Multiple recalls, case series, ASPCA/APCC data | A |
1. Species-specific metabolism
How et al. (1994, Gen Comp Endocrinol) directly compared dog, cat, and rat skin: total 7-DHC ~1,860 ng/cm² (dog) and ~1,960 ng/cm² (cat) versus ~17,620 ng/cm² (rat) — roughly 10-fold lower precursor. After 2.25 J UV-B, rat skin showed a ~40-fold rise in vitamin D₃; dog and cat skin showed no detectable change. Morris (1999, J Nutr) confirmed in vivo: kittens fed a vitamin D–free diet under 2 h/day UV maintained undetectable 25(OH)D unless given a 7-DHC-Δ7-reductase inhibitor (then plasma 25(OH)D rose to 91 ± 22 nmol/L). The mechanism is high cutaneous 7-dehydrocholesterol-Δ7-reductase activity diverting 7-DHC to cholesterol — an evolutionary trait of carnivores adapted to vitamin D–rich prey (How et al. 2015, broader carnivore survey). Implication: serum 25(OH)D is essentially a pure dietary biomarker in dogs and cats, free of the seasonal, latitude, and skin-pigmentation confounders that plague human epidemiology — making them, in principle, a clean model. Cats also discriminate strongly against ergocalciferol (D₂); D₃ is the operative vitamer (Morris 2002).
2. "Optimal" 25(OH)D ranges
- Dogs (Selting 2014/2016, Vet Comp Oncol; n=282 healthy German Shepherds and Goldens): iPTH plateaued and c-CRP variation flattened at 25(OH)D ~100–120 ng/mL (250–300 nmol/L). The authors proposed sufficiency ≥100 ng/mL; this is the de facto canine reference, although it is a single-study definition based on biomarker inflection, not a hard outcome. In Sharp/Selting 2015, 320 healthy dogs showed median commercial-diet 25(OH)D 47–100 ng/mL by manufacturer (overall median 67.9 ng/mL, range 9.5–249) — i.e., a majority of "healthy" pet dogs are below this threshold.
- Cats: No equivalent inflection-point study. Reported healthy ranges roughly 50–150 nmol/L; Ware 2020 reported median summation 25(OH)D 58.65 ng/mL in normal cats vs 47.1 in cardiomyopathy cats. Cats produce substantial 3-epi-25(OH)D (Sprinkle 2018), so summation 25(OH)D₃ + 3-epi is a more accurate clinical index than 25(OH)D₃ alone.
3. Observational/epidemiological associations
Dogs - Cancer (Selting 2014): 63 hemoabdomen dogs (43 malignant, 20 benign) had wide 25(OH)D range (19.4 to >150 ng/mL); relative splenic-neoplasia risk was inversely related to 25(OH)D. Wakshlag 2011: Labradors with cutaneous mast cell tumors had 25(OH)D 42 vs 48 ng/mL in controls. Husbands 2013 (n=335 cancer dogs) median 62.6 vs 67.4 ng/mL controls. Effects are real but small. - CKD (Parker 2017, JVIM; OSU cohort): 25(OH)D, 1,25(OH)₂D, and 24,25(OH)₂D fell with IRIS stage; FGF-23, hyperphosphatemia, hypoalbuminemia, and CaPP >70 predicted survival, but in their model 25(OH)D alone was NOT independently associated with survival (P=0.73). So the dog CKD survival signal travels primarily through phosphorus/FGF-23, not 25(OH)D. - Cardiac (Kraus 2014, JVIM; n=82): 25(OH)D 100 ± 44 nmol/L in CHF dogs vs 123 ± 42 nmol/L controls (P=0.023); 25(OH)D associated with time-to-CHF/sudden-death (P=0.02). Osuga 2015 confirmed graded reduction across ACVIM stages. - Critical illness (Jaffey 2018, PLOS One; n=99 ICU): low 25(OH)D independent predictor of in-hospital and 30-day mortality. - IBD/CE/PLE: Allenspach 2017 (43 PLE dogs) — non-survivors had median 25(OH)D 4.36 ng/mL vs much higher in survivors. Titmarsh 2015 — 25(OH)D inversely associated with neutrophils, monocytes, IL-2, IL-8. Multiple replications. - Immune-mediated disease (Mick 2019): MST 106 days if 25(OH)D₃ ≤ median, not reached if > median. - Infection: blastomycosis — Jacobs/Jaffey 2021 found association with mortality (n=19); O'Brien 2018 (n=22) did not — direct conflict in small samples. Coccidioidomycosis, leishmaniasis, spirocercosis, CIRDC all show low-25(OH)D associations.
Cats - Hospitalized 30-day mortality (Titmarsh 2015, PLOS One; n=99): cats in lowest 25(OH)D tertile had OR 8.27 (95% CI 2.54–31.52) for 30-day death, independent in 12-variable model. This is the strongest cat all-cause mortality signal. - Cardiomyopathy (Ware 2020, JVIM; n=44 CM, 56 normal): summation 25(OH)D positively associated with survival time and fractional shortening, negatively with left atrial enlargement. - Mycobacteriosis, CKD, hyperthyroidism: smaller studies all show low-25(OH)D associations.
Caveat applying to all of the above: these are cross-sectional or short-prospective designs in sick animals. Reverse causation (illness lowering 25(OH)D via inflammation, hypoalbuminemia, anorexia, vitamin D-binding protein loss) is not excluded. There is no prospective association of 25(OH)D status with all-cause longevity in healthy dogs or cats.
4. Interventional trials
- Atopic dermatitis (Klinger 2018, Vet Rec): randomized, double-blinded, placebo-controlled, 8-week cross-over, 23 client-owned dogs; cholecalciferol significantly reduced pruritus and CADESI lesion scores vs placebo, with rise in 25(OH)D₃ correlating with pruritus reduction. Half the paricalcitol arm developed hypercalcemia. This is the only properly controlled canine RCT showing a clinical benefit of D3 supplementation on a hard symptom. Small n; not replicated.
- CKD-MBD (Parker/Miller, OSU 2020–2022): 84-day calcifediol (extended-release 25(OH)D₃, ~2 µg/kg/d) in 10 dogs with IRIS stages 2–3 CKD raised 25(OH)D and reduced PTH and FGF-23; a follow-up subset showed reduced ACE activity. No survival or progression endpoint.
- Healthy-dog dose-response: Backus/Young 2016 — D₃ at 5× NRC RA produced only a marginal 25(OH)D rise after 9–10 weeks. Tryfondidou-style work and the Mellanby group later showed that oral D₃ in dogs is metabolized variably and slowly; oral 25(OH)D₃ (calcifediol) is ~3–5× more potent and ~2× faster (Young/Backus 2017; Cartiff 2023). A full dose-response (Hazel et al. 2023, Front Vet Sci) fed 795–9,992 IU/kg DM for 6 months: 25(OH)D rose linearly with dietary D, ≥5,510 IU/kg achieved >100 ng/mL in all dogs, no observable adverse effects up to 9,992 IU/kg DM in adult dogs (well above the AAFCO SUL but below puppy-toxicity thresholds).
- Cats: No published vitamin D supplementation RCTs with clinical endpoints.
Difference from human trials: Human RCTs (VITAL, ViDA, D-Health) of D₃ at 2,000–60,000 IU/wk show null effects on all-cause mortality, cancer incidence, and cardiovascular events at the population level, with possible benefit in cancer mortality and respiratory infection subgroups. Dog/cat data have nothing comparable; we are decades behind.
5. Toxicity
- Cholecalciferol in dogs: oral LD50 reported 10–88 mg/kg (rodenticide context, 0.075% bait); clinical signs at ~0.1 mg/kg; hypercalcemia and azotemia at ~0.5 mg/kg; fatalities at 2–13 mg/kg. Long half-life (weeks); persistent 25(OH)D elevations after intoxication (Gerhard/Jaffey 2020).
- Cats are particularly sensitive to D₃; the Crossley 2017 case series (3 kittens) and Vecchiato 2021 case series (5 cats, fish-based "complementary" food) showed hypercalcemia, soft-tissue mineralization, AKI, with two cats developing permanent IRIS-stage-2 CKD.
- Hill's 2019 recall: 33 SKUs of canned dog food, ~22 million cans; FDA tested lots at up to ~33× the safe upper limit (FDA cited some samples ~70× intended D content, ≈8,750 IU/1,000 kcal). Plaintiffs allege hundreds to thousands of dog deaths; class action consolidated in MDL 2887. December 2018 had a separate dry-food recall at a contract manufacturer. AAFCO consequently reaffirmed the 2016 reduction of the canine SUL to 3,000 IU/kg DM (750 IU/1,000 kcal).
- Therapeutic margin: in healthy dogs, clinical signs begin at ~10–50× the AAFCO minimum daily intake; in cats the margin is wider for D₃ (cats discriminate against D₂ but tolerate D₃ doses up to ~30,080 IU/kg DM by AAFCO max), but pediatric cats and homemade-diet cats have tipped over with single ingredient errors.
6. AAFCO and commercial diets
- Dog (2016 revision): minimum 500 IU/kg DM, maximum 3,000 IU/kg DM (down from 5,000) — based on Tryfonidou's Great Dane endochondral-ossification work at 4,000 IU/kg.
- Cat: minimum reduced to 280 IU/kg DM; maximum ~30,080 IU/kg DM (raised).
- Empirically (Sharp 2015): with commercial AAFCO-compliant diets, median canine 25(OH)D was 67.9 ng/mL (range 9.5–249 across 320 dogs and 40 manufacturers), with >50% below the proposed 100 ng/mL sufficiency cutoff. So commercial diets are sufficient for rickets prevention but probably suboptimal under the Selting framework. There is a real argument for raising AAFCO minimums (or for routine 25(OH)D monitoring), but no consensus.
7. Disease-specific summary
| Condition | Association (low 25OHD ↔ worse) | Interventional evidence |
|---|---|---|
| Canine cancer (lymphoma, MCT, OSA, hemangiosarcoma) | Yes, multiple cohorts | None on outcomes |
| Canine CKD-MBD | Yes; 25(OH)D itself NOT independently linked to survival in best study (Parker 2017) | Calcifediol improves surrogate markers (PTH, FGF-23); no survival data |
| Canine CHF/DCM | Yes (Kraus, Osuga) | None |
| Feline CM | Yes (Ware) | None |
| Canine atopic dermatitis | Yes | Klinger 2018 RCT positive (n=23) |
| Canine chronic enteropathy / PLE | Yes, strong | None |
| Canine parvovirus | One ZA cohort (Schoeman group) – low 25(OH)D associated with non-survival | None |
| Canine sepsis / critical illness | Yes (Jaffey) | None |
| Feline 30-day all-cause mortality | OR 8.27 lowest tertile | None |
8. Lifespan/healthspan endpoints
This is the central question, so let me be blunt:
- No published RCT or prospective cohort in dogs or cats has shown that vitamin D supplementation extends lifespan or healthspan. Period.
- The longest, best controlled lifespan intervention in dogs remains the Lawler/Kealy 14-year Labrador caloric-restriction study (n=48): 25% restriction extended median lifespan by 1.8 years (P=0.004) versus controls. The same cohort received identical (presumably AAFCO-compliant) vitamin D in their food — no vitamin D arm.
- The Dog Aging Project (>50,000 dogs enrolled, NIA-funded) has published demographic and frailty papers, biomarker work (e.g., protein-catabolite biomarkers, 2024), and runs the TRIAD rapamycin RCT. No DAP publication to date addresses vitamin D supplementation as an intervention or 25(OH)D as a longevity biomarker. The Golden Retriever Lifetime Study (Morris Animal Foundation, n≈3,000) has published serial biomarker data but no vitamin D longevity analyses I can identify.
- Cat-specific longitudinal lifespan data are even thinner; nothing equivalent exists.
The "vitamin D extends pet lifespan" claim therefore lives entirely in inference from cross-sectional disease associations + analogy to human meta-analyses. Rigorous grading: speculative, not "supported."
9. Practical takeaways
For an owner of a healthy adult dog or cat fed an AAFCO-compliant commercial diet:
- The probability that supplementation above AAFCO levels meaningfully extends lifespan is unknown and unsupported by direct evidence. Plausibility is moderate by analogy; effect size is unmeasured.
- Risk of harm from naive supplementation is non-trivial (Hill's, kitten-food, rodenticide cases). Dogs and cats lack the cutaneous "self-regulating" buffer humans have.
- For a dog with confirmed atopic dermatitis, CKD-MBD, CHF, or chronic enteropathy, supplementation guided by serum 25(OH)D — preferably calcifediol where the goal is rapid normalization — is supportable from current data, with monitoring of ionized calcium, phosphorus, and creatinine.
Recommendations
Stage 1 — Default position (healthy pet, commercial diet): Do not supplement vitamin D beyond AAFCO label. The lifespan/healthspan rationale is unsupported and the therapeutic margin is narrow. Verify the diet is AAFCO-compliant and avoid stacking products (multivitamin + omega-3 + treats — fat-soluble vitamins compound).
Stage 2 — Measure before treating: If interested in optimization (especially for senior dogs, large/giant breeds, or homemade-diet feeders), measure serum 25(OH)D (Heartland Assays via the Veterinary Diagnostics Institute "VDI" panel is the standard reference; LC-MS/MS preferred over chemiluminescent assays for dogs because of 3-epi cross-reactivity in cats and inter-method drift). Targets reasonable under current data: dogs 100–150 ng/mL (Selting); cats summation 25(OH)D + 3-epi >50–60 ng/mL (extrapolation, weaker basis).
Stage 3 — Targeted supplementation if 25(OH)D < ~60–80 ng/mL in a dog or in any pet with one of the disease-associated conditions above: Use D₃ at ~5–10 IU/kg/day, or calcifediol if rapid correction is needed (markedly more potent and predictable; evidence: Young/Backus 2017, Cartiff 2021). Recheck 25(OH)D, ionized calcium, phosphorus, creatinine at 4–8 weeks and at 12 weeks. Dietary cholecalciferol up to ~5,500–10,000 IU/kg DM diet has been shown safe over 6 months in adult dogs (Hazel 2023) but exceeds the AAFCO SUL and should not be done blind.
Stage 4 — Contraindications/red flags that should change this recommendation: any history of hypercalcemia, granulomatous disease, primary hyperparathyroidism, calcium-oxalate urolithiasis, growing puppies (especially giant breeds), pregnant queens, concurrent calcitriol therapy, or diet already including high-fish-oil/fortified treats.
Benchmarks that would change my answer: - A DAP, GRLS, or independent prospective interventional trial showing D-supplementation associated with reduced all-cause mortality or frailty progression — would move this from "speculative" to "supported." - A canine equivalent of VITAL (e.g., a placebo-controlled D supplementation trial in ≥1,000 senior dogs with hard endpoints) — none is announced. - Replication of the Klinger atopic-dermatitis RCT at larger n.
Caveats
- Assay heterogeneity is severe. Chemiluminescent immunoassays, RIA, and LC-MS/MS give materially different 25(OH)D values; the "100 ng/mL" Selting cutoff was set with a specific chemiluminescent platform and is not directly portable. Cats further require attention to 3-epi-25(OH)D.
- Reverse causation pervades the canine and feline observational literature. Inflammation (CRP-mediated), hypoalbuminemia, and PLE-driven loss of vitamin D–binding protein lower measured 25(OH)D independently of intake. The "low 25(OH)D = bad outcome" pattern in critical illness, CHF, IBD, and cancer is therefore partially confounded.
- Sample sizes are small. Most canine cohort studies have n=20–100; the largest (Selting/Sharp) is 282–320 healthy dogs. Cat studies are smaller still. Single-study results (e.g., Klinger atopic RCT n=23) are not yet replicated.
- Conflicting findings exist within infectious disease (blastomycosis: O'Brien 2018 negative for survival association vs Jacobs 2021 positive) and within the CKD literature (25(OH)D not independently associated with canine CKD survival in Parker 2017 despite consistent associations in human CKD).
- The Hill's recall mortality figure ("hundreds to thousands of pets") comes from plaintiff counsel in active litigation, not from FDA confirmed adverse-event counts; the FDA confirmed elevated D and renal injury but did not publish a death tally.
- AAFCO maxima are derived from puppy and pediatric toxicity data, not adult longevity optima. The truly "optimal" dietary D for adult dog or cat lifespan is genuinely unknown — and absent an interventional lifespan study, probably will remain so for another decade.
50 IU of Vitamin D per pound of dog (helps blood in this study) - May 2024
A study on the effects of vitamin D supplementation on hematological parameters and serum 25-hydroxy vitamin D in healthy dogs
BMC Vet Res . 2024 May 24;20(1):221. doi: 10.1186/s12917-024-04080-1.
Reza Gholipour Shahraki 1, Tahoora Shomali 2, Mahnaz Taherianfard 1, Nooshin Derakhshandeh 3, Saeed Nazifi 3, Ebrahim Abhaji 3
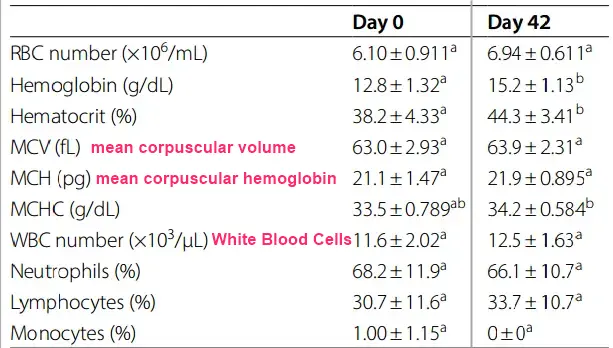
Background: Limited studies are available on vitamin D supplementation in dogs. This study evaluates the effect of a commercial vitamin D3 supplement on serum 25-hydroxy vitamin D as well as selected biochemical and hematological parameters in healthy dogs. Eight intact male adult dogs with a mean body weight of 20 kg from mixed breeds were included in the study. After adaptation period, dogs received vitamin D3 supplement at the dose of 50 IU/kg body weight per day. Blood samples were collected on days 0, 14, 28 and 42 of supplementation. Food was used for analysis of vitamin D3 content.
Results: Significant increase in serum level of 25-hydroxy vitamin D3 was detected since day 14 of supplementation.
Changes in serum 25-hydroxy vitamin D3 concentration during time showed an upward significance (p < 0.05).
Vitamin D3 content of the food was 2900 IU/kg dry matter.
Changes in serum phosphorus levels were upward significant.
No dog showed calcium or phosphorus levels above the highest reference level.
Liver and kidney parameters remained in the reference range during the experiment.
A gradual significant increase was observed in hemoglobin and hematocrit which was started from day 14. Vitamin D3 supplementation had no significant effect on neutrophils, monocytes and lymphocytes percent during the study.
Conclusions: Vitamin D3 supplementation at 50 IU/kg BW daily, increases serum levels of 25-hydroxy vitamin D in healthy dogs fed with a diet containing proper amount of this vitamin. It also increases hemoglobin and hematocrit levels in a time dependent manner without inducing adverse effects.
📄 Download the PDF from VitaminDWiki
References
McCollum EV, Simmonds N, Becker JE, Shipley PG. An experimental demonstration of the existence of a vitamin which promotes calcium deposition. J Biol Chem. 1922;53:293-8.
Chang SW, Lee HC. Vitamin D and health - the missing vitamin in humans . Pediatr Neonatol. 2019;60(3):237-44. https://doi.org/10.1016/j. pedneo.2019.04.007.
Hurst EA, Homer NZ, Mellanby RJ. Vitamin D metabolism and profiling in Veterinary Species. Metabolites. 2020;10(9):371. https://doi.org/10.3390/metabo10090371.
Zafalon RVA, Risolia LW, Pedrinelli V, Vendramini THA, Rodrigues RBA, Amaral AR, Kogika MM, Brunetto MA.
- Vitamin D metabolism in dogs and cats and its relation to diseases not associated with bone metabolism.
- J Anim Physiol Anim Nutr (Berl). 2020;104(1):322-42. https://doi.org/10.1111/jpn.13259.
Mellanby RJ, Mee AP, Berry JL, Herrtage ME. Hypercalcaemia in two dogs caused by excessive dietary supplementation of vitamin D. J Small Anim Pract. 2005;46(7):334-8. https://doi.org/10.111l7j.1748-5827. 2005.tb00329. x.
Backus RC, Foster LR. Investigation of the effects of dietary supplementation with 25-hydroxyvitamin D3 and vitamin D3 on indicators of vitamin D status in healthy dogs. Am J Vet Res. 2021;82(9):722-36. https://doi.org/10.2460/ajvr.82.9.722.
Jewell DE, Panickar KS. Increased dietary vitamin D was associated with increased circulating vitamin D with no observable adverse effects in adult dogs. Front Vet Sci. 2023;10:1242851. https://doi.org/10.3389/fvets.2023.1 242851.
National Research Council Ad Hoc Committee on Dog and Cat Nutrition. Vitamins in: nutrient requirements of dogs and cats. Washington, DC: The National Academies; 2006. pp. 1 93-245.
Medrano M, Carrillo-Cruz E, Montero I, Perez-Simon JA, Vitamin D. Effect on Haematopoiesis and Immune System and clinical applications. Int J Mol Sci. 2018;19(9):2663. https://doi.org/10.3390/ijms19092663.
Muller T, Lohse L, Blodau A, Frommholz K. Vitamin D and blood parameters. Biomolecules. 2021;11(7):1017. https://doi.org/10.3390/biom11071017.
Ochogwu OL, Salawu L, Owojuyigbe TO, Adedeji TA. Vitamin D Deficiency and its association with Anemia and blood transfusion requirements in Nigerian adults with Sickle Cell Anemia. Plasmatology. 2021;15. https://doi. org/10.1177/2634853521 1051690.
Alizadeh K, Ahmadi S, Sarchahi AA, Mohri M. The effects of age, sex, breed, diet, reproductive status and housing condition on the amounts of 25(OH) vitamin D in the serum of healthy dogs: reference values. Vet Med Sci. 2022;8(6):2360-6. https://doi.org/10.1002/vms3.943. Epub 2022 Sep 22.
Arifin WN, Zahiruddin WM. Sample size calculation in Animal studies using resource equation Approach. Malays J Med Sci. 2017;24(5):101-5. https://doi.org/10.21315/mjms2017.24.5.1 1. Epub 2017 Oct 26
Latimer KS, Robert J, editors. Duncan & Prasse's veterinary laboratory medicine: clinical pathology, 2011,5th ed. Wiley-Blackwell, Chichester, West Sussex, UK.
Official Methods of Analysis, AOAC INTERNATIONAL, USA, 18th Edition. 2005, Chap. 45, pp 28-31.
Kurzbard RA, Backus RC, Yu S. Rapid improvement in vitamin D status with dietary 25-hydroxycholecalciferol in vitamin D insufficient dogs. J Nutr Sci., Ahmad S, Ullah H, Khan MI, Gul M, Ahmed MS, Khalil M, Ahmad M, Khan AB. Effect of Vitamin D Supplementation on the Hemoglobin Level in Chronic Kidney Disease Patients on Hemodialysis: A Systematic Review and MetaAnalysis. Cureus. 2023;15(6):e40843. doi: 10.7759/cureus.40843.
Young LR, Backus RC. Oral vitamin D supplementation at five times the recommended allowance marginally affects serum 25-hydroxyvitamin D concentrations in dogs. J Nutr Sci. 2016;5:e31. https://doi.org/10.1017/jns.2016.23.
Liu T, Zhong S, Liu L, Liu S, Li X, Zhou T, Zhang J. Vitamin D deficiency and the risk of anemia: a meta-analysis of observational studies. Ren Fail. 2015;37(6):929-34. https://doi.org/10.3109/0886022X.2015.1052979.
Smith EM, Tangpricha V. Vitamin D and anemia: insights into an emerging association. Curr Opin Endocrinol Diabetes Obes. 2015;22(6):432-8. https:// doi.org/10.1097/MED.0000000000000199.
Ahmad S, Ullah H, Khan MI, Gul M, Ahmed MS, Khalil M, Ahmad M, Khan AB. Effect of vitamin D supplementation on the Hemoglobin Level in chronic kidney disease patients on Hemodialysis: a systematic review and Meta-analysis. Cureus. 2023;15(6):e40843. https://doi.org/10.7759/cureus.40843.
Titmarsh HF, Cartwright JA, Kilpatrick S, Gaylor D, Milne EM, Berry JL, Bommer NX, Gunn-Moore D, Reed N, Handel I, Mellanby RJ. Relationship between vitamin D status and leukocytes in hospitalised cats. J Feline Med Surg. 2017;19(4):364-9. https://doi.org/10.1177/1098612X15625454.
Keywanloo M, Ahmadi-Hamedani M, Jebelli Javan A, Rakhshani Zabol F Effects of parenteral vitamin D3 supplementation on Hematological Parameters of Healthy Holstein Bulls. Arch Razi Inst. 2021;76(5):1 509-1 7. https://doi. org/10.22092/ari.2020.352209.1 552.
Wall-Gremstrup G, Holt R, Yahyavi SK, Jorsal MJ, Juul A, Jorgensen N, Blomberg Jensen M. High-dose vitamin D3 supplementation shows no beneficial effects on white blood cell counts, acute phase reactants, or frequency of respiratory infections. Respir Res. 2024;25(1):11. https://doi.org/10.1186/s12931-023-02642-9.
Selting KA, Sharp CR, Ringold R, Thamm DH, Backus R. Serum 25-hydroxyvi- tamin D concentrations in dogs - correlation with health and cancer risk. Vet Comp Oncol. 2016;14(3):295-305. https://doi.org/10.1111/vco.12101.
Cats and dogs can generate Vitamin D in their fur, and then lick if off - 2012
The Offbeat Reason Why Cats and Dogs Lay In The Sun and Lick Their Fur
"Dogs and cats secrete the same oily cholesterol from their skin, just like us. When they lay in the sun, that oil is, again, converted to Vitamin D3. The difference, however, is that because they have so much hair, the vitamin ends up on their fur and doesn’t get absorbed through their skin."
" Oh and by the way, I mention cats and dogs, but this info applies to ALL furry mammals and also to birds."
Vitamin D in Health and Disease in Dogs and Cats Nov 2020
REVIEW ARTICLE| VOLUME 1, P265-277, NOVEMBER 2020 https://doi.org/10.1016/j.yasa.2020.07.017
Ronald Jan Corbee, DVM, PhD, Dipl ECVCN
Table of Contents

Key points
25-hydroxyvitamin D is not a sensitive indicator of vitamin D status in dogs and cats.
Food intake and food ingredient analysis are often absent in studies on vitamin D in dogs and cats.
1,25-dihydroxyvitamin D is the most potent vitamin D metabolite with the greatest binding affinity to the vitamin D receptor.
Determination of other vitamin D metabolites rather than 25-hydroxyvitamin D in vitamin D studies will provide better insight in cause-effect relationships.
Web
- https://www.vetinfo.com/vitamin-d-for-dogs.html 227 IU/pound of dry dog food
- A toxic dose > 2272 IU per pound of dry dog food
- Serum 25-hydroxyvitamin D concentrations in dogs – correlation with health and cancer risk July 2014
- 3.9X more likely for dog to get Cancer if < 40 ng (100 ng typical in dogs)
- 📄 Download the PDF from VitaminDWiki
- Fat Soluble Vitamins: A, D, E & K in Dogs
| Vitamin | Recommended Minimum Daily Dose for Dogs |
Toxic Dose (This dose must be given daily for months to create toxicity.) |
Sources | Signs of Deficiencies |
| A | 2272 IU/lb of food consumed on a dry matter basis | 113,600 IU/lb of food consumed on a dry matter basis | Liver, fish liver oil, vegetables, dairy products | Night blindness, retarded growth, poor quality skin and hair |
| D | 227 IU/lb of food consumed on a dry matter basis | 2272 IU/lb of food consumed on a dry matter basis | Sunshine, dairy products, fish liver oil | Rickets, poor eruption of permanent teeth |
| E | 23 IU/lb of food consumed on a dry matter basis | 455 IU/lb of food consumed on a dry matter basis | Cold pressed vegetable oils, meats, nuts, green leafy vegetables | Reproductive failure, brown bowel syndrome |
| K | Synthesized in the body | none | Kelp, alfalfa, egg yolk | Increased clotting time and hemorrhage |
Vitamin D overdose . . “is rare and we have never heard of it happening in real life situations ”
Examples of Vitamin D suppliers for dogs
- PetLife-400 New Zealand $23 US
- 300 squirts: 400 IU of Vitamin D, 3 IU Vitamin E, Aloe vera, water, and perhaps Magnesium L Threonate
- 2 squirts daily for dogs up to 5 kg
- Senior pets rejuvenated with vitamin D He had such success that he is now making PetLife
- Miracle Coat Miracle Mist Skin Treatment Spray
- $8 Amazon, Vitamins A, D2 & E
- Uses “Tea Tree oil. Far more effective than Aloe Vera”
MaxxiDog Amazon $22 Omegas and ingredients per ounce include Vitamin A 500 IU Vitamin D3 100 IU Vitamin E 50 IU
-
- Vitamin B Complex, Amino Acids, 63 Trace Minerals, Ester C, Glucosamine, Aloe Vera, Vitamin D, E , K, Iron, Calcium and Magnesium ,Zinc
Related in VitaminDWiki
- Vitamin D: A Predictor of Diseases in Dogs and Cats
- Dogs health increased by Vitamin D
- Cats 8.2X more likely to die in 30 days if low vitamin D when enter hospital
- Half of dogs now get cancer, it used to be just 1 percent (probably low Vitamin D)
- Dogs with Cancer have low vitamin D, same as humans
- Controversy cats and vitamin D
