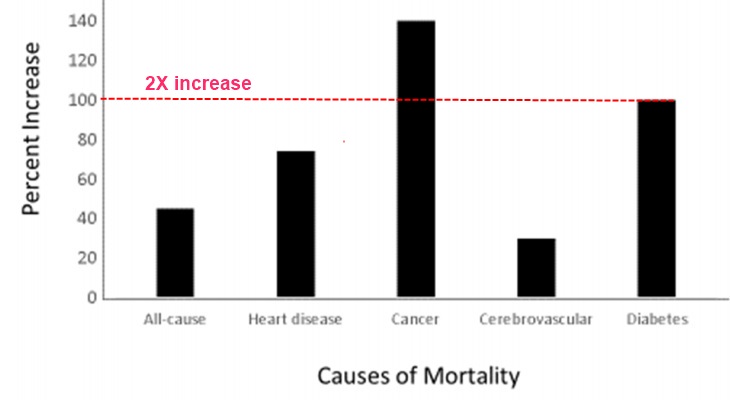
Diabetes (T2) 2X to 6X higher prevalence in some dark-skinned or poor people
Claude AI May 2026
Question: What is the higher incidence/prevalence of diabetes in the following groups? Native Americans, African Americans, Africans, Aborigines, Mongolians, Americans living below the poverty line
Bottom line
Type 2 diabetes prevalence in the six target groups ranges from about 0.5× (rural sub-Saharan Africa) to ~7× (Pima Indians of Arizona) the non-Hispanic-white U.S. baseline of ~6.9–7.5%. The most striking gradients are not racial per se — they tracked
- Westernization,
- urbanization and
- poverty almost everywhere studied
Pima vs. Mexican Pima, urban vs. rural Africa, remote vs. non-remote Aboriginal Australia, U.S. <100% FPL vs. ≥500% FPL FPL = Federal Poverty Level).
TL;DR
- Reference: Age-adjusted diagnosed diabetes in non-Hispanic White U.S. adults is 6.9% (CDC, NHIS 2019–2021); total (diagnosed + undiagnosed) NHANES estimate is ~9–10%.
- Highest-burden groups (relative to NH whites): Pima Indians of Arizona ~5–7× (≈38–50% of adults), Aboriginal Australians in remote Northern Territory communities ~4–6× (29–40% of adults ≥20 y), Navajo ~3–4× historically, U.S. AI/AN overall ~2.0× (13.6%), African Americans ~1.75× (12.1%), Mongolians ~1.2–1.4× (8–10%), Americans <100% FPL ~2.6× (13.1% vs. 5.1% at ≥500% FPL).
- Confirms the user's framing: Rural sub-Saharan Africa is genuinely low (often 0.5–1.5× white U.S. baseline, i.e. ~1–3%), 2–4× lower than urban African rates, supporting the "epidemiologic transition" hypothesis.
Key Findings — Comparative Table
| Population (year/source) | Absolute T2D prevalence | Ratio vs. NH-White U.S. (6.9%) | Notable variation |
|---|---|---|---|
| Non-Hispanic White U.S. adults (CDC NDSR, NHIS 2019–2021, age-adjusted) | 6.9% diagnosed; ~9–10% total (NHANES Aug 2021–Aug 2023) | 1.0× (reference) | Men 7.7%, women 6.2% |
| All American Indian / Alaska Native (AI/AN) adults (CDC NDSR 2019–2021; IHS NDW) | 13.6% | ~2.0× | IHS 2017: 14.6%; declined from peak 15.4% (2013); regional range 5–25% |
| Navajo adults ≥20 y (Navajo Health & Nutrition Survey, 1997; Brown UJPH 2023 update) | 22.9% age-standardized in 1997; >40% in those ≥45 y; combined diabetes+pre-diabetes ~50% in 2020 | ~3–4× | Women > men; rises sharply with age |
| Pima Indians, Gila River, Arizona (NIDDK / Schulz 2006; Bennett 1971; Pearson 2015) | ~38% age/sex-adjusted adults; ~50% in adults ≥35 y | ~5–7× (highest documented in world) | Mexican Pima, similar genes but traditional lifestyle: only 6.9% — proves environmental dominance |
| Alaska Natives (overall) | ~6–9% (lower than Lower-48 AI/AN; some IHS regions report ~16–17%, others (Alaska) lower) | ~1.0–1.3× | High regional heterogeneity; Yup'ik historically very low (<2%) |
| African Americans (NH Black) U.S. (CDC NDSR 2019–2021; NHANES) | 12.1% diagnosed; total (NHANES 2009–2018) ~14.6% | ~1.75× diagnosed (~1.6× total) | Black women > Black men in some surveys; ESRD from diabetes 2× whites |
| Sub-Saharan Africa — RURAL (Lancet/SSA reviews; Aspray Tanzania; Maher Uganda; Price Malawi 2018; AWI-Gen 2024) | Typical range 1–4%; classic studies: 0.87% rural Tanzania (1989), 0.6–1.4% rural Uganda, ~2% rural Malawi (2013–16), 3.6% rural Ghanaian men/5.5% women (RODAM) | ~0.15–0.6× (well below white U.S.) | Doubles to triples in urban centers; AWI-Gen middle-aged (40–60 y) pooled rural+peri-urban: 5.5% baseline, doubling within 6 y |
| Sub-Saharan Africa — URBAN (for contrast) (same sources) | ~6–18%: urban Senegal 17.9%; urban Ghana ~10%; urban Kenya 12%; urban Malawi 3% | ~1–2.5× | Often 2–4× higher than rural counterparts in same country |
| Aboriginal & Torres Strait Islander Australians (ABS NATSIHMS 2022–24; AIHW; Burrow & Ride; Hare 2022) | Adults ≥18 y: 15.5% (measured); Indigenous Eye Health Survey ≥40 y: ~37% self-report | ~3× (vs. ~6.1% non-Indigenous; AIHW: "2.9× as likely after age-adjustment") | Remote 21.7% vs. non-remote 14.3%; Torres Strait Islander adults 8.6% vs. Aboriginal 7.2% (NATSIHS 2022–23 self-report) |
| Aboriginal Australians, REMOTE Northern Territory (Hare et al., BMJ Open Diabetes 2022) | Adults ≥20 y: 28.6%; Central Australia 39.5%; Top End 24.2% | ~4–6× | Among highest documented worldwide; women > men; very high youth-onset T2D |
| Mongolia (national) (WHO STEPS 2019; Dovepress 2023 OGTT study; Tandfonline 2018) | STEPS 2019 (15–69 y): 8.3%; reanalyzed: 9.5% (urban 8.9%, rural 7.7%); OGTT-confirmed adults ≥30 y: ~10% | ~1.2–1.5× | Tripled since 1999 (3% → ~10%); pre-diabetes 17.4%; men > women; Inner Mongolian Chinese rural: only 3.7% |
| U.S. adults below 100% FPL (CDC NDSR 2019–2021; PCD CDC 2026; NHIS) | 13.1% age-adjusted diagnosed (vs. 5.1% at ≥500% FPL) | ~2.6× vs. high-income; ~1.9× vs. NH-white average | In 2011–2014 the relative increase in DM prevalence vs. high-income was +100% for poor, +74% for near-poor, +40% for middle-income |
1. American Indian : Pima 6X, Navajo 3.5X, other 2X
The CDC National Diabetes Statistics Report (2019–2021 NHIS + IHS data) puts age-adjusted diagnosed diabetes in AI/AN adults at 13.6%, roughly double the non-Hispanic-white rate of 6.9%. The Indian Health Service active-clinical-population analysis (Bullock et al., BMJ Open Diabetes 2020) showed AI/AN prevalence rose from 14.4% (2006) to 15.4% (2013), then fell to 14.6% in 2017 — the first documented decline in this population, attributed in part to the IHS Special Diabetes Program for Indians. The IHS's own communications cite a 2.3× ratio vs. non-Hispanic whites and note that "in some communities prevalence is as high as 60%." Lifetime risk for AI/AN children born around 2000 is estimated at ~50%.
Tribal extremes: - Pima Indians (Gila River, Arizona): The most-studied group in the world. Bennett's 1971 Lancet paper documented ~50% prevalence in those ≥35 years using OGTT — the highest ever recorded. Schulz et al. (Diabetes Care 2006) reported age/sex-adjusted prevalence of 38% in Arizona Pima vs. only 6.9% in genetically similar Mexican Pima living traditionally — one of the strongest natural experiments in diabetes epidemiology. Modern figures: ~50% of Arizona Pima adults aged ≥35 y. - Navajo: The 1997 Navajo Health and Nutrition Survey (Will et al., Journal of Nutrition) found age-standardized prevalence of 22.9% in adults ≥20 y, with >40% of those ≥45 y affected — about 4× the contemporary U.S. rate. More recent reports describe ~50% of Navajo adults having either diabetes or pre-diabetes (2020). - Alaska Natives historically had much lower rates than Lower-48 tribes (e.g., 6% in some Alaska studies vs. 24% in southern Arizona tribes), though they have been catching up.
2. African Americans 1.75X
CDC NDSR (2019–2021): 12.1% diagnosed, vs. 6.9% in non-Hispanic whites → 1.75× higher. NHANES total (diagnosed + undiagnosed) data 2009–2018 gave ~14.6% in Black participants vs. ~10.6% in whites (~1.4× total). The HHS Office of Minority Health (2024) reports Black/African American adults are 24% more likely than U.S. adults overall to have diabetes, 78% more likely to die of it, and >2× more likely to develop diabetic kidney failure. The disparity has widened over time: in 1999–2002 the Black/white ratio for total diabetes was ~1.6×, by 2009–2012 it had grown.
3. Rural Sub-Saharan Africa (continental Africans, rural setting)
The user's framing is well-supported. Multiple high-quality studies show rural prevalence dramatically lower than urban prevalence in the same countries:
- Rural Tanzania (McLarty et al., Lancet 1989; Aspray 2000): 0.87% adults ≥15 y (1989); rural 1.7% vs. urban 5.9% (2000).
- Rural Uganda (Maher 2010; Mayega 2012): 0.6–1.4% — among the lowest rural rates documented.
- Rural Malawi (Price et al., Lancet Diabetes & Endocrinology 2018): 2% men and 2% women in rural Karonga vs. 3% in urban Lilongwe.
- Rural Ghana (RODAM study, 2016): 3.6% men, 5.5% women — vs. 10.3% men, 9.2% women in urban Ghana, and 21.4%/54.2% obesity in Ghanaian migrants in London.
- West Africa pooled (5 countries, WHO STEPS, BMJ Open 2023): age/sex-adjusted T2DM 2.5% rural vs. 6.2% urban (RR urban vs. rural ~2.5).
- Systematic review (Hilawe et al., 2019): Pooled undiagnosed-DM 3.93% rural vs. 8.68% urban — urban roughly double rural across SSA.
- AWI-Gen (Lancet Global Health 2024/2025): Among adults aged 40–60 y in South Africa, Kenya, Ghana, and Burkina Faso, baseline T2D prevalence was 5.5%, but doubled within ~6 years — the rural advantage is closing rapidly.
Implied ratio vs. non-Hispanic whites: Truly rural, traditional-lifestyle SSA populations typically run 0.15–0.6× the U.S. white rate (i.e., 1–4% vs. ~7%). This is the only group in the analysis where the ratio is materially below 1.
4. Aboriginal 5X
- National adult prevalence (ABS NATSIHMS 2022–24, biomarker-confirmed): 15.5% of Indigenous adults ≥18 y — up from 11.1% a decade earlier. Males 16.7%, females 14.3%; remote 21.7%, non-remote 14.3%; ages 55+ 34.6%.
- Self-report (NATSIHS 2022–23): 7.4% overall, with Torres Strait Islanders 8.6% slightly higher than Aboriginal 7.2% — but biomarker surveys show self-report substantially under-reports.
- AIHW summary: Indigenous adults are ~2.9× as likely as non-Indigenous adults to have diabetes after age adjustment (17% vs. 6.1%). The National Indigenous Eye Health Survey (≥40 y) found age-adjusted prevalence of 43.8% in Indigenous vs. 11.5% non-Indigenous (~3.8× ratio).
- Remote Northern Territory (Hare et al., BMJ Open Diabetes 2022, linked-data study of 21 267 Aboriginal residents): Adult (≥20 y) prevalence 28.6% in 2018/19, with Central Australia at 39.5%, comparable to historical Pima rates; type 2 accounted for 99.6% of incident cases.
Ratio vs. non-Indigenous Australians (≈ comparable to NH-white U.S.): 3–4× nationally, ~5–6× in remote NT communities.
5. Mongolia
- WHO STEPS 2019 (ages 15–69): 8.3% by fasting glucose; a reanalysis of the same data covering ages ≥18 reported 9.5% overall (urban 8.9%, rural 7.7%) with pre-diabetes 17.4%.
- OGTT-confirmed national survey (Dovepress 2023, ages ≥30): age-standardized diabetes prevalence ~10% — a 3.2× rise since 1999 (when it was ~3%).
- Ulaanbaatar (Plos One 2025, STEPS-style 2018): 6.27% in adults ≥20, men 7.5% > women 5.3%.
- Inner Mongolian rural Chinese (Han + Mongol mixed, 2009 cross-section): much lower at 3.7%, with high IFG (18.5%), suggesting traditional pastoralist lifestyle still protective.
Ratio vs. NH-white U.S. (6.9% diagnosed; ~9% total): roughly 1.0–1.4× depending on metric. Mongolia is a clear example of a country mid-transition: rural nomadic populations historically had very low rates, urbanization is driving prevalence up rapidly. The IDF Western Pacific 2024 Region (which includes Mongolia) average is 12.4%.
6. Americans Below the Federal Poverty Line (1.9x) 2.6X
This is where data are clearest. From the CDC National Diabetes Statistics Report (NHIS 2019–2021, age-adjusted):
| Family income vs. FPL | Diagnosed diabetes prevalence |
|---|---|
| <100% FPL (poor) | 13.1% |
| 100–299% FPL | ~10% |
| 300–499% FPL | ~7% |
| ≥500% FPL | 5.1% (men 6.3%, women 3.9%) |
- Ratio (poor vs. high-income): 13.1% ÷ 5.1% = 2.57×
- Ratio (poor vs. NH-white average 6.9%): ~1.9×
- A separate CDC PCD analysis (2026) of NHIS 2021–2024 found that among adults below the FPL, rural residents 16.1% vs. urban 13.4% had diabetes — i.e., poverty + rurality is additive.
- The 2016 MMWR report on disparities (1999–2002 vs. 2011–2014) showed that the absolute income-related prevalence gap widened from 4.7 to 7.1 percentage points. CDC/ADA 2021 analysis: between 2011 and 2014, relative diabetes prevalence vs. high-income was +100% for poor, +74% near-poor, +40% middle-income.
The poverty effect is partly mediated by race/ethnicity (minorities are over-represented in low-income groups) and partly independent (operates within every racial/ethnic group through obesity, food insecurity, physical inactivity, and reduced healthcare access).
Recommendations (for VitaminDWiki content use)
- Use the CDC 2019–2021 figures (NDSR) as the primary U.S. comparator (NH-white = 6.9%), and cite NHANES Aug 2021–Aug 2023 for total (diagnosed + undiagnosed) figures (~10–11% adults).
- For the Pima Indian "headline" figure, cite both Bennett 1971 (Lancet — original 50% in adults ≥35 y) and Schulz 2006 (Diabetes Care — 38% age/sex-adjusted; the Mexican-vs-Arizona Pima comparison is the strongest argument that lifestyle, not just genetics, drives the disparity). Avoid using "Pima Indians = 50%" without specifying the age band; for the entire population the figure is closer to 38%.
- For Aboriginal Australians, use two figures — the national ~15.5% (NATSIHMS 2022–24, all adults) and the remote NT ~29% (≥20 y) with Central Australia 39.5% (Hare 2022). Cite AIHW's 2.9× age-adjusted ratio; note the remote rate is higher than for women than men, opposite of the global pattern.
- For rural sub-Saharan Africa, use the systematic review numbers — rural prevalence typically 1–4%, urban often 2–3× higher. Best primary sources: Hilali (BMC Public Health 2011), Price 2018 (Lancet Diabetes Endocrinol), AWI-Gen 2024 (Lancet Global Health), McLarty 1989 (Lancet). State explicitly that rural rates are below the white-U.S. baseline.
- For Mongolia, cite WHO STEPS 2019 (8.3%) as the official figure; the Dovepress 2023 OGTT-confirmed study (~10%, ages ≥30) is more rigorous. Note pre-diabetes ~17%.
- For poverty, the cleanest single statistic is 13.1% (<100% FPL) vs. 5.1% (≥500% FPL) → ~2.6× gradient (CDC NDSR 2019–2021). This is a robust, government-published figure suitable for wiki content.
- Update threshold: If CDC publishes NDSR with 2022–2024 data (likely 2026–2027), figures may shift modestly downward for AI/AN and upward for African Americans/Hispanics — recheck annually.
Caveats
- Definitional variability: Studies use different criteria (self-report, fasting plasma glucose ≥126 mg/dL, OGTT 2-h ≥200 mg/dL, HbA1c ≥6.5%). HbA1c thresholds underestimate diabetes in some African populations (Khosla 2021), so older sub-Saharan Africa figures using HbA1c alone may underestimate. AWI-Gen and Malawi studies used FPG.
- Self-report vs. measured: In remote Aboriginal Australia and rural SSA, self-reported diabetes substantially underestimates true prevalence — the NATSIHMS 2022–24 found 1 in 5 Indigenous Australians with biomarker diabetes had not been previously diagnosed; the SSA review by Hilawe et al. found undiagnosed rates 41–75%.
- Pima Indians "highest in the world" claim: Now contested — Aboriginal Australians in Central Australia (39.5% in those ≥20 y), the Marshall Islands (~31% ≥20 y), and Nauru have approached or exceeded historical Pima rates. The Pima rate has been roughly stable since the 1980s–2000s while others rose.
- Type 2 vs. all-types: Most national surveys (CDC, AIHW, IDF, WHO STEPS) report "diabetes" without subtype. In adults, type 2 is 90–95%+ of cases in all six target groups, so the figures cited are an excellent proxy for type 2; type 1 in AI/AN is rare (~1% of cases in NT Aboriginal data).
- AI/AN data are regional: IHS data come predominantly from on-reservation and clinic users; urban AI/AN populations may be undercounted. Tribe-level prevalence varies from ~6% (some Alaska Native communities) to ~50% (Pima).
- Rural SSA: heterogeneity: Reported rural prevalence ranges from 0.6% (Uganda) to 8.8% (some rural South African studies); the user's "2–3% rural vs. 8–12% urban" framing captures the modal pattern but not the full range. Some rural populations are already mid-transition.
- Mongolia STEPS 2019 included urban-dominant Ulaanbaatar (~half of national population); pure rural-nomadic-herder prevalence is likely lower than the 7.7% reported, but no recent OGTT-confirmed nomadic-only study exists.
- Poverty figures: Cross-sectional, so causality runs in both directions (diabetes worsens income, low income increases diabetes risk). The CDC PCD 2023 analysis shows the diabetes/income gradient narrowed 2001–2011 then widened 2011–2018.
- Some forward-looking figures noted in sources (e.g., IDF projecting Western Pacific +19% prevalence by 2050; AFR Region +142%; ADA 2026 NDSR estimating 40.1 million Americans with diabetes) are projections, not measured prevalence, and should not be presented as established fact.
Related in VitaminDWiki
Americans
- African-Americans at high risk of obesity and diabetes - 2011
- Bibliography of Black-White health disparities linked to vitamin D - Dr. Grant
- The Sunshine Vitamin — restoring what modern life took away from Native Americans
- Last in health, last in funding: Native Americans' double burden
- African American Health Disparities are associated with low Vitamin D - Grant
- Dark Skin Diabetics and Vitamin D - many studies
- 99.7% of people who got Diabetes had been regularly consuming food emulsifiers
In Ultra-processed foods
Diabetes
- Overview Diabetes and vitamin D
- Type 2 Diabetes treated by Vitamin D (often 50,000 IU weekly) – meta-analysis
- Diabetics helped by vitamin D in 5 ways – meta-analysis
- Diabetes and poor Vitamin D genes - many studies
Diabetes and Vitamin D meta-analyses - many studies
Low Vitamin D increases health problems - independent of skin color
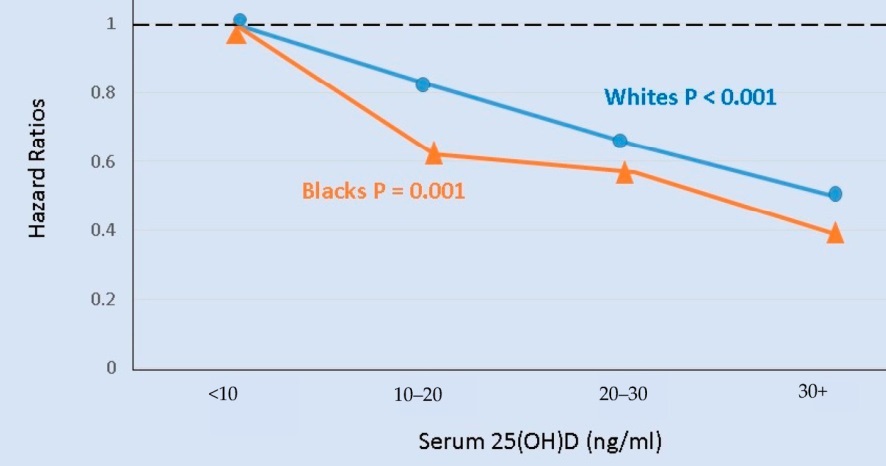
Increase Health Problems for Blacks