COVID vaccines observed to increase Autoimmune diseases in 2022
COVID-19 vaccines and autoimmune disorders: A scoping review
AIMS Medical Science Nov 2025 doi: 10.3934/medsci.2025023
Background Upon the global COVID-19 vaccination campaign, unprecedented in the history of public health, concerns have emerged regarding potential associations between vaccination and autoimmune disorders. Historical research has long identified mechanisms by which vaccines might trigger or unmask autoimmune processes. However, systematic synthesis of evidence concerning COVID-19 vaccines and autoimmunity remains limited.
Objective To review the literature on associations between COVID-19 vaccination and autoimmunity, focusing on six conditions: Graves' disease, Hashimoto's thyroiditis, multiple sclerosis, rheumatoid arthritis, systemic lupus erythematosus, and type 1 diabetes mellitus.
Methods We conducted a scoping review of 109 studies published in 2022, retrieved from PubMed and the WHO COVID-19 databases. Inclusion criteria encompassed English-language articles reporting empirically verifiable clinical manifestations of autoimmune disease associated with any COVID-19 vaccine, without restrictions by population, geography, or study type.
Results Across 109 included studies, relapses or flares in patients with autoimmune disorders were reported in nearly 60% of studies, while about one-quarter described new-onset autoimmune disorders in persons without prior autoimmunity. Several mechanisms of action linking COVID-19 vaccination and autoimmune disorders were reported, such as autoimmune inflammatory syndrome induced by adjuvants, molecular mimicry, bystander immune activation, and interactions with immunosuppressive and disease modifying therapies. Serious adverse events, though less common than mild or moderate ones, were also reported. General and population-specific vaccine efficacy were claimed but empirical support was often lacking.
Conclusions This review highlights the substantial patterns of reported associations of autoimmune disorders following COVID-19 vaccination, in patients with and without prior autoimmunity. The general and population-specific benefits of vaccination are claimed, but evidence for them is lacking. A proper evaluation of risks and benefits is needed to support vaccination recommendations given the reported associations between it and autoimmune disorders.
Autoimmune adverse event following COVID-19 vaccination in Seoul, South Korea - June 2024
Autoimmune disorders reported following COVID-19 vaccination: A disproportionality analysis using the WHO database - Jan 2024
Research European Journal of Clinical Pharmacology 12 January 2024Volume 80, pages 445–453, (2024) PDF behind paywall
Purpose Owing to adverse event following immunization (AEFI) related to autoimmune disorders and coronavirus disease 2019 (COVID-19) vaccines sharing common biological mechanisms, identifying the risk of AEFIs associated with COVID-19 vaccines remains a critical unmet need. We aimed to assess the potential safety signals for 16 AEFIs and explore co-reported adverse events (AEs) and drugs using the global database of the World Health Organization, VigiBase.
Methods We assessed the occurrence of 16 AEFIs following COVID-19 vaccination through the Standardized MedDRA Queries group “Immune-mediated/Autoimmune Disorders” from MedDRA and performed a disproportionality analysis using reporting odds ratio (ROR) and information component (IC) with 95% confidence intervals (CIs).
Results We identified 25,219 events associated with COVID-19 vaccines in VigiBase. Although rare, we detected four potential safety signals related to autoimmune disorders following COVID-19 vaccination, including
ankylosing spondylitis or psoriatic arthritis (ROR 1.86; 95% CI 1.53–2.27),
inflammatory bowel disease (ROR 1.77; 95% CI 1.60–1.96),
polymyalgia rheumatica (ROR 1.42; 95% CI 1.30–1.55), and
thyroiditis (ROR 1.40; 95% CI 1.30–1.50), with positive IC025 values.
The top co-reported AEs were musculoskeletal disorders, and immunosuppressants were the most representative co-reported drugs.
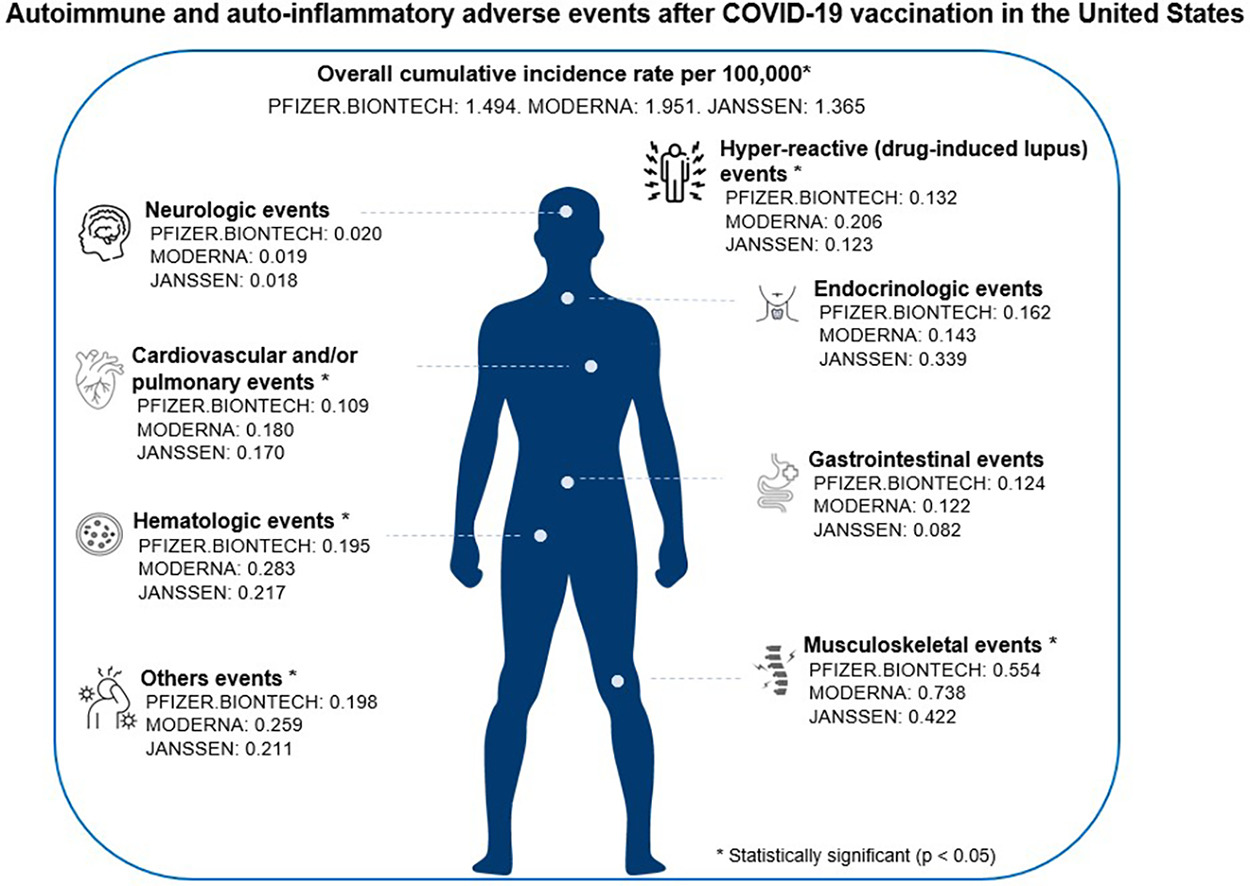
Autoimmune and auto-inflammatory adverse events after COVID-19 vaccination in the United States
Clinical Immunology Volume 259, February 2024, 109882 https://doi.org/10.1016/j.clim.2023.109882 Seong-Jang Kim a b c 1, Taeho Greg Rhee d e f 1, Sung Ryul Shim g h
Abstract
We identified 3620 autoimmune-related adverse events among 223.2 million US residents using Vaccine Adverse Events Reporting System and the COVID-19 Data Tracker. This study is the first to quantify the cumulative incidence of autoimmune and auto-inflammatory adverse events after COVID-19 vaccination. We reported autoimmune and auto-inflammatory adverse events across 6 major classes of medical conditions and stratified by age, sex, and manufacturer.