ADHD incidence: big increase during COVID
DSM-5 Changes, COVID-19, and ADHD Diagnosis Rates in Individuals Younger Than 30 Years
JAMA Netw Open. 2026 Apr 1;9(4):e265775. doi: 10.1001/jamanetworkopen.2026.5775
Plain language summary
This cohort study of youth in British Columbia examines changes in attention-deficit/hyperactivity disorder diagnosis rates following changes in Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition) and the COVID-19 pandemic.
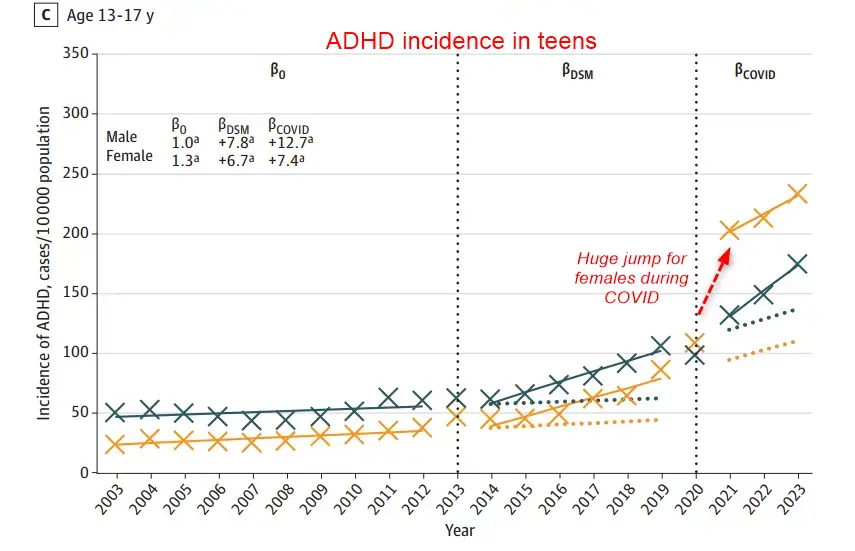
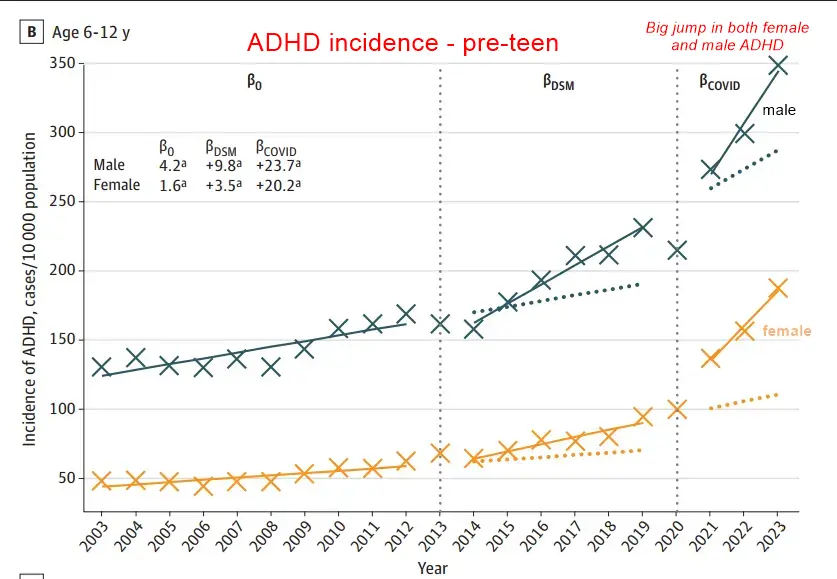
ADHD Explosion: What Really Happened over the Pandemic?
Focal Points review of the study
- "Using a large cohort of nearly 2.75 million individuals aged 3 to 29 years between 2003 and 2023, the authors tracked new (incident) ADHD diagnoses based on physician visits, hospital records, and prescriptions. "
- "Increasing over the years, the estimated cumulative fraction who contracted SARS-CoV-2 was ~90%. Across the full age 3–29 cohort ~75–90% received at least one or more doses of the COVID-19 vaccine. "
VitaminDWiki speculation: Vaccination OR COVID
Asked COVID -AI deep research to speculate about it April 2026
The evidence can be stratified by certainty:
| Claim | Evidence level | Key limitation |
|---|---|---|
| ADHD patients have lower vitamin D | Strong (multiple meta-analyses, >10,000 participants) | Reverse causation and confounding not excluded |
| Vitamin D modulates dopaminergic pathways | Moderate-Strong (cell culture, animal models) | Translation to human ADHD uncertain |
| Supplementation improves ADHD symptoms | Low-Moderate (small RCTs, only in deficient populations) | No large multi-site trial conducted |
| Lockdown vitamin D drops contributed to ADHD surge | Hypothetical (no direct study) | Confounding with screen time, exercise, stress is inextricable |
A Mendelian randomization study — using genetic variants affecting vitamin D metabolism as instruments for ADHD risk — would substantially advance the causal question but has not yet been published. Prospective studies measuring serum 25(OH)D during lockdowns while tracking neurodevelopmental outcomes with adjustment for screen time and physical activity would be the most informative design for testing the pandemic-specific hypothesis.
ADHD diagnoses surged dramatically during 2020–2023, but the vitamin D deficiency hypothesis — while biologically plausible — ranks among the weaker explanations compared to telehealth expansion, social media awareness, and pandemic stress mimicking symptoms. The COVID-19 pandemic triggered a well-documented explosion in ADHD diagnoses and stimulant prescriptions, with U.S. stimulant fills rising from roughly 35.5 million annually in 2019 to over 41 million by 2022. Lockdowns also worsened population-wide vitamin D status by 3–8 ng/mL. Pre-pandemic research consistently links low vitamin D to ADHD through dopaminergic pathways, neuroinflammation, and neurodevelopmental mechanisms. Yet no published study has directly connected pandemic-era vitamin D drops to the ADHD surge, and the magnitude of observed deficiency changes appears insufficient to explain the scale of diagnostic increase. The strongest evidence points to a convergence of structural, technological, and psychosocial forces — with vitamin D as, at most, a minor contributing factor.
The ADHD explosion is real, unprecedented, and concentrated among adults
Multiple independent data sources confirm an extraordinary increase in ADHD diagnoses and treatment during 2020–2023. CDC National Survey of Children's Health data show parent-reported ADHD prevalence in U.S. children aged 3–17 climbing from ~9.8% in 2018/2019 to ~11.4% by 2022 — roughly 7 million children. But the steepest increases occurred in adults. New adult ADHD diagnoses rose 25–30% between 2020 and 2022, with women aged 23–49 seeing diagnosis rates roughly triple. Stimulant prescriptions to women aged 20–44 increased approximately 40–50% over this period.
The international pattern mirrors the U.S. trend. NHS England reported a 7-fold increase in adult ADHD referrals between 2019 and 2023, with waiting lists stretching to 3–5 years. Australian ADHD medication dispensing rose approximately 25% from 2019–20 to 2022–23. Private UK diagnosis services saw demand increase by roughly 400%. A CDC MMWR report using IQVIA prescription data confirmed that stimulant fills among adults aged 20–39 climbed approximately 10–15%, with the increase most pronounced among women (~20%). The FDA acknowledged a formal Adderall shortage in October 2022, driven partly by demand outstripping DEA production quotas — amphetamine salt quotas rose from roughly 43,000 kg in 2020 toward 54,000 kg by 2022–2023.
Telehealth platforms were central to this expansion. ADHD-related telehealth visits increased over 500% between 2019 and 2022 per JAMA Network Open data. Companies like Done Health and Cerebral became massive prescribers through brief virtual assessments — sometimes as short as 15–20 minutes — prompting DOJ investigations and DEA scrutiny. The Ryan Haight Act's temporary suspension during COVID enabled prescribing of Schedule II controlled substances via telehealth without in-person visits, removing a critical regulatory bottleneck.
Pre-pandemic research links vitamin D to ADHD through dopamine and neurodevelopment
The biological case connecting vitamin D deficiency to ADHD rests on convergent evidence across observational studies, mechanistic research, and animal models. Multiple meta-analyses have established that ADHD patients consistently have lower serum 25(OH)D levels than healthy controls. Khoshbakht et al. (2018) pooled data from approximately 4,000 participants and found a standardized mean difference of −0.57 (95% CI: −0.82 to −0.31). Gan et al. (2019), analyzing over 10,000 participants, reported that ADHD patients had vitamin D levels roughly 6.93 ng/mL lower than controls and were 2.57 times more likely to be vitamin D deficient (95% CI: 1.58–4.18). Individual studies from Turkey, Iran, Qatar, and Egypt report even larger differences, with ADHD children sometimes averaging just 11–16 ng/mL compared to 21–23 ng/mL in controls.
The mechanistic pathway is coherent. Vitamin D receptors (VDR) and the activating enzyme CYP27B1 are densely expressed in the prefrontal cortex, hippocampus, substantia nigra, and cerebellum — regions governing attention, executive function, and motor control. Vitamin D upregulates tyrosine hydroxylase, the rate-limiting enzyme in dopamine synthesis, directly connecting it to the dopaminergic dysfunction central to ADHD pathophysiology. Patrick and Ames (2014) demonstrated that vitamin D activates brain-specific tryptophan hydroxylase 2 (TPH2), directing serotonin synthesis toward the brain — relevant because serotonin modulates impulse control and emotional regulation. Vitamin D also suppresses neuroinflammatory cytokines (TNF-α, IL-6, IL-1β) and modulates microglial activation, connecting to emerging evidence that neuroinflammation contributes to ADHD.
Animal models provide the strongest experimental evidence. The developmental vitamin D (DVD) deficiency model developed by the Eyles/McGrath group at the University of Queensland shows that rats born to vitamin D-deficient dams exhibit hyperlocomotion, altered habituation, impaired attention-like behaviors, and altered dopamine turnover in the striatum and nucleus accumbens. These animals also show increased sensitivity to amphetamine — paralleling the pharmacology of ADHD treatment. DVD-deficient rodents display brain morphological changes including lateral ventricle enlargement and reduced cortical thickness, demonstrating that developmental vitamin D deficiency produces a phenotype with both face validity and construct validity for ADHD.
Supplementation trials show modest effects, but only in deficient populations
Randomized controlled trials of vitamin D supplementation in ADHD patients exist but remain limited in scale and quality. Mohammadpour et al. (2018) conducted the most rigorous trial — a double-blind, placebo-controlled RCT of 54 ADHD children in Iran receiving 2,000 IU/day vitamin D₃ as adjunctive therapy to methylphenidate for 8 weeks. The vitamin D group showed significant improvement in evening symptoms on the Conners' Parent Rating Scale, though total ADHD scores did not differ significantly. Dehbokri et al. (2019) found that adjunctive vitamin D improved ADHD symptom scores beyond methylphenidate alone in 96 children. Elshorbagy et al. (2018) documented significant symptom improvement after correcting vitamin D deficiency in 50 ADHD children, though this open-label design limits conclusions.
A critical pattern emerges: positive results cluster in Middle Eastern populations with high baseline vitamin D deficiency, while studies in Northern European populations with moderate vitamin D levels and universal supplementation policies show weaker or null associations. This suggests supplementation may only benefit those who are genuinely deficient — a finding consistent with basic nutritional biology but limiting the generalizability of the intervention. The current Cochrane-grade evidence stands at approximately "low to moderate" certainty, with no large multi-site RCT (n > 500) yet conducted.
The prenatal evidence is somewhat stronger. Sucksdorff et al. (2021), using the Finnish Maternity Cohort with approximately 1,067 ADHD cases and matched controls, found that maternal vitamin D deficiency (<30 nmol/L) during pregnancy was associated with adjusted OR of 1.53 (95% CI: 1.11–2.12) for offspring ADHD. The Spanish INMA cohort (Morales et al., 2015) and Greek Rhea cohort (Daraki et al., 2018) support this association, particularly for inattention symptoms. These prenatal findings represent the most compelling evidence for a causal developmental pathway.
Why lockdown vitamin D drops probably cannot explain the ADHD surge
During COVID-19 lockdowns, population vitamin D levels declined measurably. Zupo et al. (2022) systematically reviewed 14 studies and estimated mean 25(OH)D decreases of approximately 3.9 ng/mL. Barrea et al. (2020) documented 4–5 ng/mL drops during Italy's strict lockdown; Pereira et al. (2020) found a 17% decline in a São Paulo cohort. Lips et al. (2021) estimated drops of 3–8 ng/mL depending on latitude, season, and lockdown stringency. Outdoor physical activity decreased 30–50% based on Fitbit data from 30 million users.
However, these declines are almost certainly too small to be a primary driver of ADHD symptom increases. The difference between ADHD and non-ADHD children in observational studies averages ~7 ng/mL, and the prenatal studies showing elevated risk compare deficient (<20 ng/mL) versus sufficient (>30 ng/mL) categories — a gap exceeding 10 ng/mL. A 3–8 ng/mL population-level decline might push some borderline individuals from "insufficient" to "deficient," but the modest effect sizes in the vitamin D-ADHD literature (ORs of 1.3–1.8) indicate vitamin D is one of many contributing factors even under ideal analytical conditions.
Three additional problems undermine the hypothesis:
Timeline mismatch. The strongest evidence for vitamin D's role involves prenatal and early developmental windows, not acute deficiency in school-age children or adults. Most lockdowns lasted 2–4 months — potentially meaningful for pregnant women during critical gestational periods but unlikely to fundamentally alter neurocognitive function in older children and adults whose brains are already developed.
Inextricable confounding. Children who stayed indoors and became vitamin D deficient simultaneously experienced more screen time, less physical exercise, disrupted sleep, social isolation, and parental stress. These factors have stronger and more consistent evidence for attention effects. It is statistically impossible to isolate the vitamin D signal from these correlated exposures using observational data.
No direct evidence exists. As of current literature, no published study has directly measured whether pandemic-era vitamin D changes mediated increased ADHD symptoms or diagnoses. The connection remains entirely inferential — two well-documented phenomena (lockdown vitamin D drops and ADHD increases) occurring simultaneously without demonstrated causal linkage.
The competing explanations carry substantially stronger evidence
The COVID-era ADHD surge is best understood as a convergence of at least six independent forces, most supported by stronger and more direct evidence than vitamin D:
Telehealth accessibility and diagnostic loosening. The elimination of in-person visit requirements made diagnosis dramatically easier while simultaneously reducing assessment rigor. Some platforms diagnosed ADHD in 15–30 minute sessions without comprehensive neuropsychological testing. Analysis suggests telehealth-originated stimulant prescriptions grew 20–30% faster than traditional prescriptions.
Social media awareness driving self-identification. #ADHD content accumulated over 20 billion TikTok views by 2023, with 52% of top videos classified as misleading (Yeung et al., 2022). Clinician surveys indicate 30–50% of new assessment requests in 2022–2024 were prompted by social media content. This drove both legitimate identification of underdiagnosed individuals and overidentification.
Pandemic stress producing ADHD-mimicking symptoms. Approximately 30–40% of adults presenting for post-pandemic ADHD evaluation did not meet full diagnostic criteria upon comprehensive assessment. The ASRS screening tool produces elevated false-positive rates under conditions of chronic stress and anxiety. Sleep disruption — affecting 40–60% of adults during the pandemic — independently produces ADHD-like sustained attention and working memory deficits.
Unmasking of previously compensated ADHD. The loss of external structure (work routines, school schedules, commute patterns) removed "scaffolding" that had allowed individuals with mild or undiagnosed ADHD to function. Working from home was particularly challenging for adults who relied on office environments to maintain productivity.
Historical underdiagnosis correction. Women, who present predominantly with inattentive-type ADHD rather than hyperactive-impulsive symptoms, were historically underdiagnosed. The pandemic-era surge among women aged 23–49 likely represents partial correction of this longstanding diagnostic gap, compounded by emerging understanding of how estrogen fluctuations modulate dopaminergic pathways.
Screen time effects on attention. Children's recreational screen time approximately doubled during lockdowns (from ~2.5 to ~5+ hours/day). Meta-analyses show a statistically significant but small association with ADHD symptoms (r ≈ 0.10–0.16), explaining only 1–3% of variance. The Lancet Digital Health (2024) concluded screen time likely exacerbates symptoms in predisposed children but is unlikely to be a primary cause.
Where the vitamin D hypothesis fits in the evidence hierarchy
The vitamin D-ADHD connection occupies a specific niche: biologically plausible, observationally supported, but causally undemonstrated — and likely a minor contributor to the pandemic ADHD surge relative to structural and psychosocial factors. The VitaminDWiki database (vitamindwiki.com) catalogs extensive research on both the vitamin D-ADHD link and COVID-related vitamin D deficiency, maintaining a dedicated ADHD and Vitamin D Deficiency category. This resource aggregates the observational and mechanistic literature comprehensively, though it tends toward advocacy for vitamin D's importance rather than neutral synthesis of competing explanations.
Conclusion
The pandemic ADHD surge is real and multifactorial. The vitamin D hypothesis carries genuine biological plausibility — dopaminergic regulation, neurodevelopmental programming, and neuroinflammatory modulation provide coherent mechanistic pathways, and observational data consistently show ADHD patients are vitamin D deficient. However, the hypothesis faces three decisive weaknesses as an explanation for the COVID-era surge specifically: the lockdown-induced vitamin D drops (3–8 ng/mL) fall below the threshold typically associated with clinical ADHD risk; the timeline of acute adult deficiency doesn't match the strongest evidence, which implicates prenatal developmental windows; and the vitamin D signal cannot be separated from far larger concurrent changes in screen time, routine structure, stress, and diagnostic access. The most parsimonious explanation for the surge combines telehealth-enabled diagnostic expansion, social media-driven awareness, stress-mimicking symptoms, and the unmasking of historically underdiagnosed populations — particularly women. Vitamin D deficiency likely operates as a background modifiable risk factor that may worsen symptoms in deficient individuals, rather than as a meaningful driver of the pandemic-era increase. The prenatal pathway remains the most promising avenue for future research: if lockdowns reduced maternal vitamin D during critical gestational windows, effects on "pandemic babies'" neurodevelopment could emerge in coming years as these children reach school age.
Chat GPT AI lengthy answer to this question
- ADHD incidence jumped during COVID. Wondering why. Perhaps a decrease in vitamin D due to COVID or being indoors?
Related in VitaminDWiki
- Children with ADHD again helped by 50,000 IU weekly Vitamin D – RCT
- Vaccinated children had more chronic diseases
- ADHD 2 times more likely if poor Omega-6 to Omega-3 ratio – meta-analysis
- ADHD in Arab countries – vitamin D is the only changeable risk factor – review
Perhaps Vaccine ==> increased ADHD
