Worried about Vitamin D toxicity? 7 VDR modulators (and many cheap options) reduce hypercalcemia risk
Synthetic Modulators of the Vitamin D Receptor: From Structural Innovation to Disease-Specific Applications
Biomolecules. 2026 Mar 6;16(3):396. doi: 10.3390/biom16030396
Tram Thi-Ngoc Nguyen 1 2, Tomohiro Kurokawa 1 2 3, Yoshiaki Kanemoto 1 2, Takahiro Sawada 1 2, Shigeaki Kato 1 2 3
Vitamin D signaling via the vitamin D receptor (VDR) regulates calcium-phosphate homeostasis and extensive gene programs controlling cell proliferation, differentiation, immune tone, and metabolism. However, systemic use of the natural agonist 1α,25-dihydroxyvitamin D3 (calcitriol) for extraskeletal indications is limited by dose-limiting hypercalcemia. This review summarizes VDR biology and the structural basis of ligand action, emphasizing how ligand-induced repositioning of helix 12 and altered coregulator recruitment can be exploited to engineer selective VDR modulators. We highlight medicinal chemistry strategies spanning secosteroidal analogs with side-chain or ring modifications and emerging non-seco scaffolds and discuss clinically established agents (e.g., calcipotriol and paricalcitol) alongside experimental "super-agonists", partial agonists, and antagonists designed to widen the therapeutic window.
Finally, we discuss current evidence for VDR targeting across cancer, metabolic disease, fibrosis, and immune-inflammatory disorders, including mechanisms of resistance such as dysregulated vitamin D metabolism and epigenetic repression. Structural and epigenomic insights are positioning next-generation VDR ligands as tissue- and pathway-biased therapeutics that may enable safer, mechanism-guided translation beyond bone and mineral indications.
Claude AI on the Synthetic VDR activators
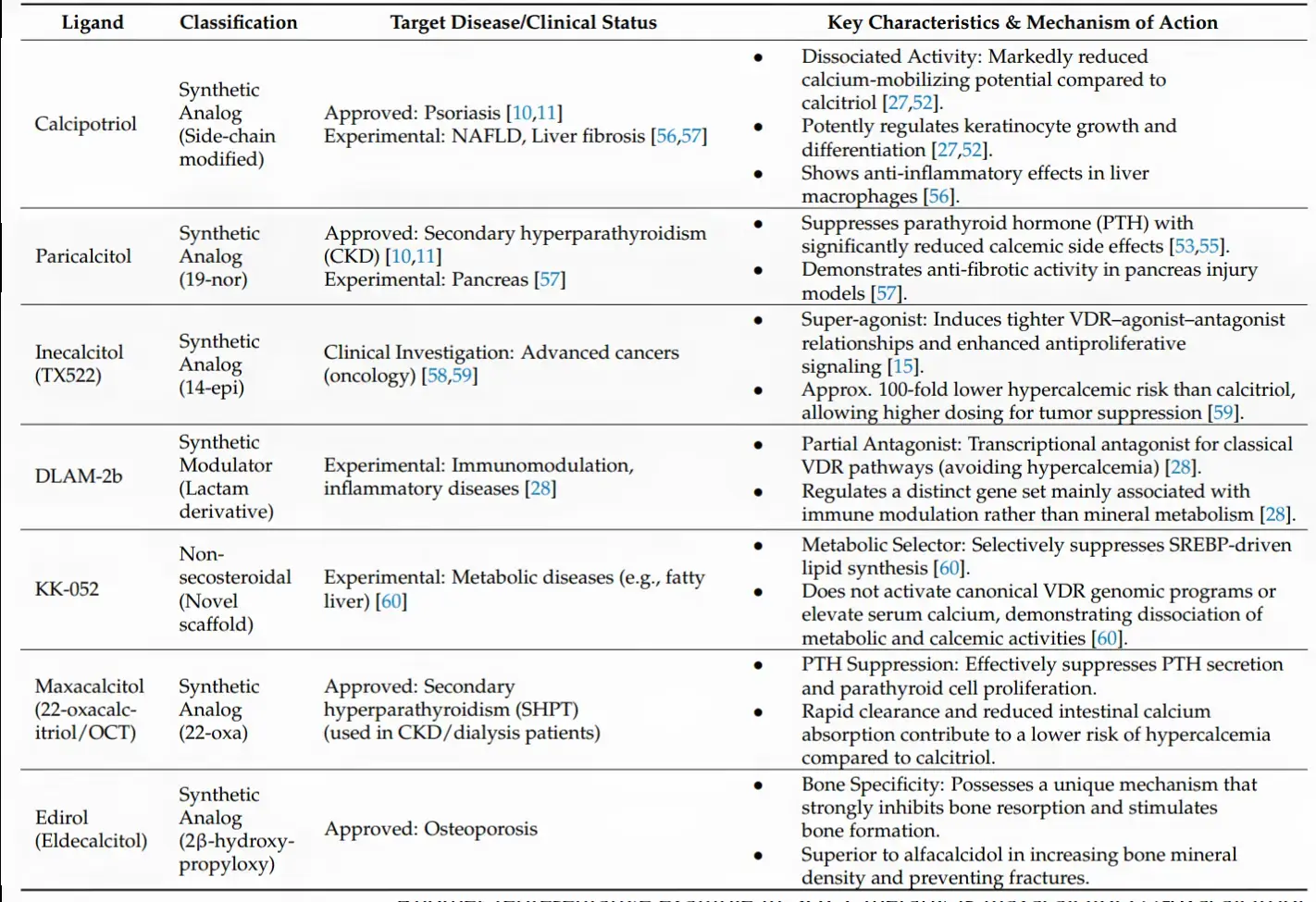
Here's a detailed breakdown of what the research shows for each compound, covering hypercalcemia risk, VDR potency, and clinical applications.
A key framing note: These analogs are not "getting more vitamin D to cells" in the conventional sense — they reshape what the VDR does. Most have similar or even slightly lower VDR binding affinity compared to calcitriol, but achieve tissue selectivity by changing which genes get activated (immune vs. calcium, bone vs. intestine), how long they persist (via VDBP binding), or how the receptor complex folds.Here are the key takeaways for your three questions:
How much hypercalcemia reduction?
The numbers vary dramatically by mechanism. Calcipotriol has comparable VDR binding affinity to calcitriol but is less than 1% as active in regulating calcium metabolism — a near-total decoupling. Inecalcitol has approximately 100-fold lower hypercalcemic activity than calcitriol, though that advantage can compress at equitoxic in vivo doses due to its short half-life. Paricalcitol-treated patients had significantly fewer sustained episodes of hypercalcemia than calcitriol patients (P=0.008), with the mechanism being reduced stimulation of intestinal calcium transport proteins such as calbindin and PMAT compared to calcitriol. KK-052 is a unique outlier — it does not use the vitamin D scaffold at all and elevates serum calcium zero.
How much more VDR activation "getting to cells"?
This framing needs a slight reframe. These aren't delivering "more" vitamin D — they're delivering different VDR signals. Eldecalcitol binds vitamin D binding protein (DBP) with 2.7-fold greater affinity than calcitriol, which extends its half-life and routes it preferentially back to bone. Inecalcitol demonstrates enhanced VDR-mediated transcriptional activity compared to 1,25D₃, with an IC₅₀ value 30 times lower in squamous cell carcinoma cells — a true super-agonist effect in tumor tissue. Paricalcitol's tissue selectivity comes from skipping intestinal VDR upregulation rather than from binding potency.
Which health problems does each address?
| Compound | Primary approved use | Promising experimental targets |
|---|---|---|
| Calcipotriol | Psoriasis, plaque psoriasis | NAFLD, liver fibrosis, alopecia |
| Paricalcitol | Secondary hyperparathyroidism (CKD) | Pancreatic fibrosis, leukemia, proteinuria |
| Inecalcitol | — (trials) | Prostate cancer (CRPC), AML |
| DLAM-2b | — | Autoimmune, inflammatory disease |
| KK-052 | — | Non-alcoholic fatty liver, metabolic syndrome |
| Maxacalcitol | Secondary hyperPTH (dialysis, Japan) | Parathyroid proliferation |
| Eldecalcitol | Osteoporosis (Japan) | Steroid-induced bone loss, fall prevention |
Eldecalcitol reduced new vertebral fractures by 26% compared to alfacalcidol over 3 years, and also reduced wrist fracture risk by 71% — making it the most striking fracture data in this group. In the inecalcitol prostate cancer trial, the maximum tolerated dose was 4,000 µg daily, with dose-limiting hypercalcemia only appearing at 8,000 µg/day, suggesting a wide therapeutic window for tumor-suppressive dosing.
All require a prescription
Related in VitaminDWiki
- Alternate ways of reducing Hypercalcemia:
Reduce Calcium, Increase Water or Magnesium or Vitamin K2
Dr. Coimbra protocol has had 50,000+ people do all 4 - Important for human health: Calcium, Magnesium, their ratio, and Phosphate
- Calcium-Magnesium Balance and How it Relates to Vitamin D - Sunil video Dec 2025
- Click on the Hypercalcemia tag at the bottom of the page to see 10+ studies in VitaminDWiki
