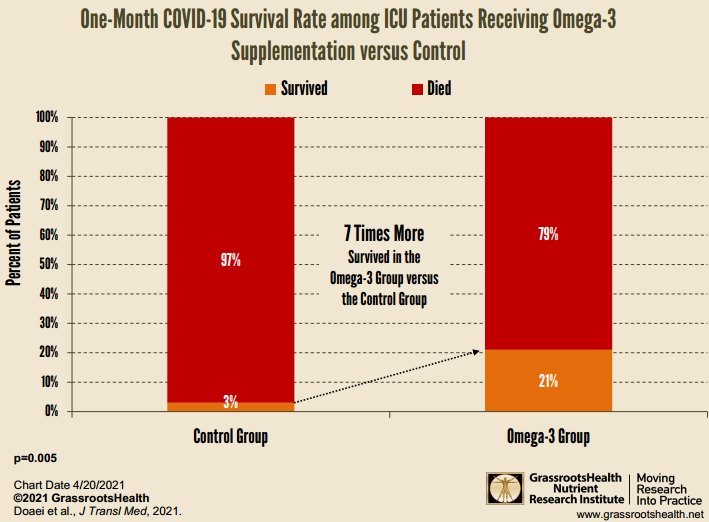
COVID-19 ICU survival rate increased 7X by daily Omega-3 – RCT
The effect of omega-3 fatty acid supplementation on clinical and biochemical parameters of critically ill patients with COVID-19: a randomized clinical trial
Journal of Translational Medicine vol 19, # 128 (2021)
Saeid Doaei, Somayeh Gholami, Samira Rastgoo, Maryam Gholamalizadeh, Fatemeh Bourbour, Seyedeh Elaheh Bagheri, Forough Samipoor, Mohammad Esmail Akbari, Mahdi Shadnoush, Fereshteh Ghorat, Seyed Alireza Mosavi Jarrahi, Narjes Ashouri Mirsadeghi, Azadeh Hajipour, Parvin Joola, Alireza Moslem & Mark O. Goodarzi
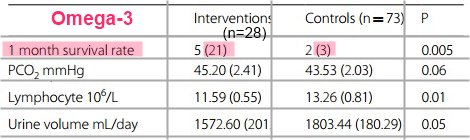
Statistically significant differences
 ---
1. Vitamin D and Omega-3 category starts with
{include}
1. Omega-3 and Virus items
{category}
1. Omega-3 and Mortality items
{category}
1. Omega-3 is very important to health
---
1. Vitamin D and Omega-3 category starts with
{include}
1. Omega-3 and Virus items
{category}
1. Omega-3 and Mortality items
{category}
1. Omega-3 is very important to health
 1. Omega-3 index is very low in many places around the world - 2016
1. Omega-3 index is very low in many places around the world - 2016
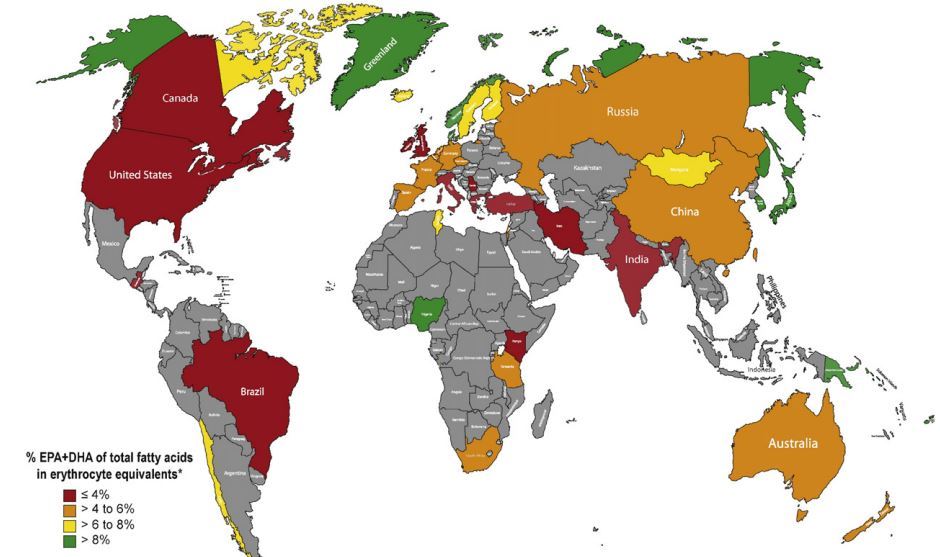 Omega-3 map (most of the world has low levels) – May 2016
Note: This RCT was done in Iran
Omega-3 RDAs around the world: 100 to 2400 mg per day, US still has no RDA – Dec 2018
---
1. Web on Omega-3 and COVID-19
* [12 clinical trials for Omega-3 and COVID-19 as of April 2021](https://www.clinicaltrials.gov/ct2/results?recrs=&cond;=COVID-19&term;=omega-3&cntry;=&state;=&city;=&dist;=)
* Potential benefits and risks of omega-3 fatty acids supplementation to patients with COVID-19 - Aug 2020 FREE PDF
* May omega-3 fatty acid dietary supplementation help reduce severe complications in Covid-19 patients? Dec 2020, FREE PDF
* Omega-3 fatty acids in the psychological and physiological resilience against COVID-19 Oct 2020, FREE PDF
* Omega 3 Fatty Acids and COVID-19: A Comprehensive Review Dec 2020, FREE PDF
---
1. Other than Omega-3
1. COVID-19 risks reduced by Vitamin D, Magnesium, Zinc, Resveratrol, Omega-3, etc. (auto-updated
1. Mortality and Virus studies (mainly Vitamin D)
{category}
1. COVID-19 treated by Vitamin D - studies, reports, videos
* {include}
1. Magnesium and Virus items
{category}
Thanks to Dr. Grant for pointing out this RCT. He got it from Michael Mindlin
Omega-3 map (most of the world has low levels) – May 2016
Note: This RCT was done in Iran
Omega-3 RDAs around the world: 100 to 2400 mg per day, US still has no RDA – Dec 2018
---
1. Web on Omega-3 and COVID-19
* [12 clinical trials for Omega-3 and COVID-19 as of April 2021](https://www.clinicaltrials.gov/ct2/results?recrs=&cond;=COVID-19&term;=omega-3&cntry;=&state;=&city;=&dist;=)
* Potential benefits and risks of omega-3 fatty acids supplementation to patients with COVID-19 - Aug 2020 FREE PDF
* May omega-3 fatty acid dietary supplementation help reduce severe complications in Covid-19 patients? Dec 2020, FREE PDF
* Omega-3 fatty acids in the psychological and physiological resilience against COVID-19 Oct 2020, FREE PDF
* Omega 3 Fatty Acids and COVID-19: A Comprehensive Review Dec 2020, FREE PDF
---
1. Other than Omega-3
1. COVID-19 risks reduced by Vitamin D, Magnesium, Zinc, Resveratrol, Omega-3, etc. (auto-updated
1. Mortality and Virus studies (mainly Vitamin D)
{category}
1. COVID-19 treated by Vitamin D - studies, reports, videos
* {include}
1. Magnesium and Virus items
{category}
Thanks to Dr. Grant for pointing out this RCT. He got it from Michael Mindlin
📄 Download the PDF from VitaminDWiki
Background
Omega-3 polyunsaturated fatty acids (n3-PUFAs) may exert beneficial effects on the immune system of patients with viral infections. This paper aimed to examine the effect of n3-PUFA supplementation on inflammatory and biochemical markers in critically ill patients with COVID-19.
Methods
A double-blind, randomized clinical trial study was conducted on 128 critically ill patients infected with COVID-19 who were randomly assigned to the intervention (fortified formula with n3-PUFA) (n = 42) and control (n = 86) groups. Data on 1 month survival rate, blood glucose, sodium (Na), potassium (K), blood urea nitrogen (BUN), creatinine (Cr), albumin, hematocrit (HCT), calcium (Ca), phosphorus (P), mean arterial pressure (MAP), O2 saturation (O2sat), arterial pH, partial pressure of oxygen (PO2), partial pressure of carbon dioxide (PCO2), bicarbonate (HCO3), base excess (Be), white blood cells (WBCs), Glasgow Coma Scale (GCS), hemoglobin (Hb), platelet (Plt), and the partial thromboplastin time (PTT) were collected at baseline and after 14 days of the intervention.
Results
The intervention group had significantly higher 1-month survival rate and higher levels of arterial pH, HCO3, and Be and lower levels of BUN, Cr, and K compared with the control group after intervention (all P < 0.05). There were no significant differences between blood glucose, Na, HCT, Ca, P, MAP, O2sat, PO2, PCO2, WBCs, GCS, Hb, Plt, PTT, and albumin between two groups.
Conclusion
Omega-3 supplementation improved the levels of several parameters of respiratory and renal function in critically ill patients with COVID-19. Further clinical studies are warranted.
Trial registry: This study was registered in the Iranian Registry of Clinical Trials (IRCT); Trial registration number: IRCT20151226025699N3; Date of registration: 2020.5.20; URL of trial registry record: https://en.irct.ir/trial/48213
Clipped from PDF
The intervention group received one capsule of 1000 mg omega-3 daily (Vita Pharmed, Switzerland) containing 400 mg EPAs and 200 mg DHAs for 14 days through adding the supplement to their enteral formula. Omega-3 fatty acids fortifed formula was administered to the case group by a nurse, who was not a member of the research team, for 2 weeks after the frst 24 h of hospitalization in ICU.
